All the information about a person’s health (e.g., their exposure to chemicals, their inherited risks, their current illnesses) lies within their molecules, according to scientists. Thus, a team of Vanderbilt University chemists started decoding that total molecular picture by examining many variations of one class of molecule, lipids, which are vital to cell membrane structure, regulating cell activities, and storing energy.
The role a lipid plays in the body is determined by its shape, and the methods and atlas this team developed, which match individual lipid molecules to their shapes, holds the key ultimately to early diagnosis of many different disorders, say the researchers who published their study (“Ion mobility conformational lipid atlas for high confidence lipidomics”) in Nature Communications.
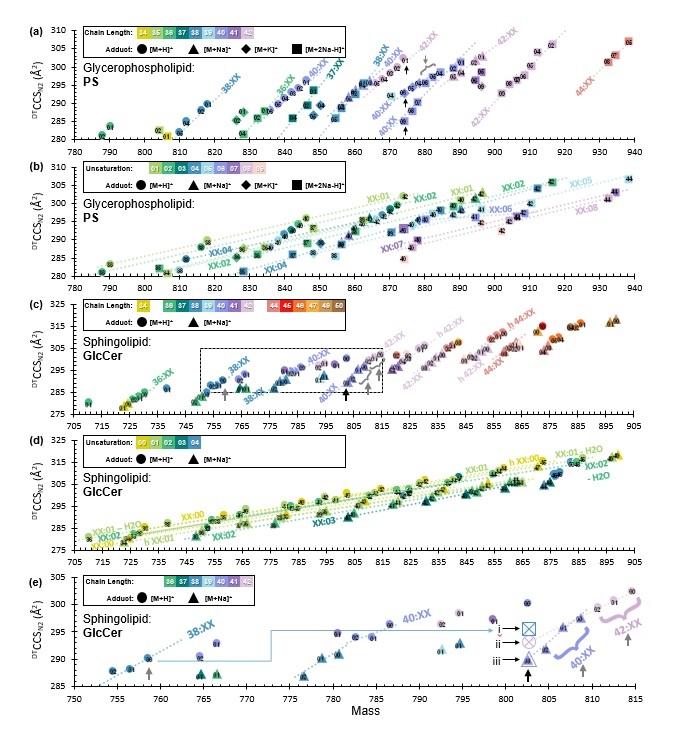
“Lipids are highly structurally diverse molecules involved in a wide variety of biological processes. Here, we use high precision ion mobility-mass spectrometry to compile a structural database of 456 mass-resolved collision cross sections (CCS) of sphingolipid and glycerophospholipid species. Our CCS database comprises sphingomyelin, cerebroside, ceramide, phosphatidylethanolamine, phosphatidylcholine, phosphatidylserine, and phosphatidic acid classes,” the investigators wrote.
“Lipids are well known to be the bellwether of disease,” said John McLean, PhD, Stevenson professor of chemistry at Vanderbilt University. “Dysregulation can mean everything from inflammation to very specific disease states. Because lipids subtly change based on what’s going on in the body, we can use an analytic strategy to map out what shape that lipids adopt as a means to identify them.”
Formerly, a single set of measurements could correspond to many different kinds of lipids, he said, but the Vanderbilt team’s atlas of lipid structures greatly narrows the possibilities. Other researchers, such as those who study lipids and their role in diseases, can go to this atlas for clues in their own work, plus add to it.
Lead author Katrina Leaptrot, PhD, a post-doctoral scholar, said the work was made possible by ion mobility-mass spectrometry, which allows scientists to analyze molecules with more scrutiny. She spent months looking for patterns in her data and learned that a lipid’s shape, and thus its ability to predict dysregulation, was most impacted by the length of its tail and how many double bonds lay in those tails.
A lipid’s tail length is a count of the number of carbon atoms it contains, while double bonds determine how strongly each carbon atom is bound to the one next to it. Double bonds create kinks in the lipids that affect normal lipid functions as well as dysregulations. For example, saturated fats are lipids without double bonds, which raises cholesterol levels in your blood.
“Now that we can better decode how nature assembled these molecules, labs around the world will be able to use this lipid atlas and contribute their own data to uncharted regions,” Leaptrot said.