Recent advances in plant biotechnology have allowed the use of plants and plant cell cultures for the production of pharmaceutical proteins, known as molecular farming. We are now able to exploit the ability of plants to produce, glycosylate, and fold biologically active proteins and other natural products.
One of the most important milestones in this process has been the development of expression systems capable of producing high levels of RNA transcription and protein translation. These systems enable high yield, scalable production of pharmaceuticals and nonpharmaceutical products, making plant-based production systems a commercially viable choice for the development of vaccines, antibodies, and enzymes.
Leaf Expression Systems is a contract development and manufacturing organization (CDMO) specializing in the milligram- to gram-scale production of proteins, vaccines, and complex natural products for research and biomedical applications. The company uses its proprietary, transient viral expression technology, Hypertrans®.
Hypertrans is a plant transient expression system consisting of a disabled virus that was developed by George Lomonossoff, PhD, project leader at Norwich Research Park. It is based on a deconstructed cowpea mosaic virus (CPMV) RNA-2, where most of the coding region is substituted with the gene or genes of interest. The flanking untranslated regions are kept but modified, as the deletion of an AUG codon at position 161 at the 5′ UTR was shown to increase the protein production by 10-fold, reaching 1.2 g/kg GFP of fresh weight (FW) tissue. This viral cassette is inserted into the T-DNA section of a binary vector called pEAQ-HT (from Hypertrans). The addition of a silencing suppressor, p19, within the T-DNA prevents transcriptional silencing and allows accumulation of product over a longer period of time.
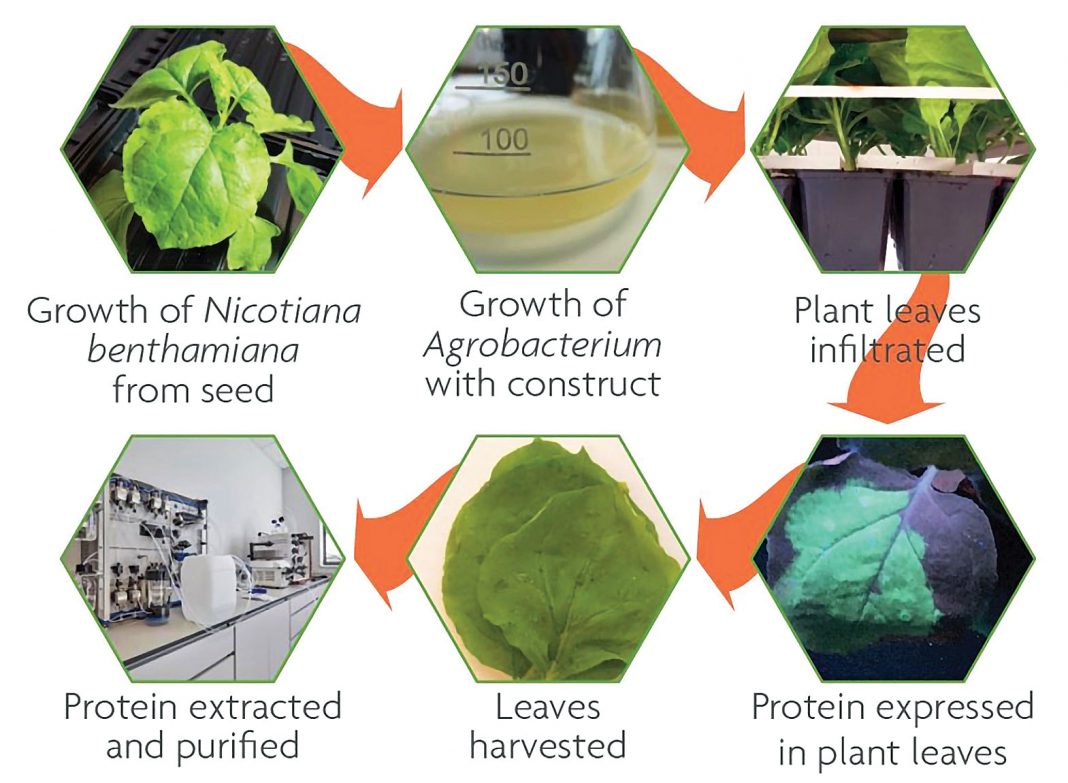
The T-DNA is transformed into the plant cells by a disarmed plant pathogenic bacteria, Agrobacterium tumefaciens. Bacteria are infiltrated into the leaves to transfer the heterologous target gene(s) encoded within the T-DNA section of the pEAQ-HT plasmid to the plant cells, where RNA transcription and protein translation takes place. Only the T-DNA section of the vector will be transferred into the plant. The system benefits from the use of a noncultivated tobacco species, Nicotiana benthamiana, which has a high transformation capability. The Hypertrans system relies on making mRNAs “hypertranslatable” and permitting extremely high-level and rapid production of proteins without viral replication.
After infiltration, the plant leaves are harvested and blended, and the proteins are extracted and purified based on their biochemical properties using traditional chromatographic techniques.
Benefits of using transient plant expression, with the Hypertrans system, include:
- rapid throughput (in which the process of producing proteins can be reduced to two weeks from the point of infiltration)
- low cost and directly scalable production capabilities
- animal-product free (thus reducing to a great extent the chance of human-threat contaminations)
- complete eukaryotic processing machinery (including complex post translational modifications).
Furthermore, the system does not have size limitations on the gene of interest, allowing the co-expression of large heteromultimeric protein complexes in one pEAQ-HT plasmid, unlike other viral vectors such as those based in monopartite viruses (i.e., tobacco mosaic virus [TMV]) and mammalian expression systems.
Plants have been successfully used to generate virus-like particles (VLPs) for vaccines against influenza, polio, and papillomaviruses. In addition, VLPs such as those produced from CPMV virus have become excellent platforms for the delivery of a range of different cargoes, including peptides, fluorophores, and therapeutic chemical compounds. For producing these CPMV VLPs, the Hypertrans construct containing the large and small coat proteins is vacuum infiltrated into N. benthamiana plants, which are cultivated within a controlled-environment facility to ensure optimum conditions for plant health and biomass production.
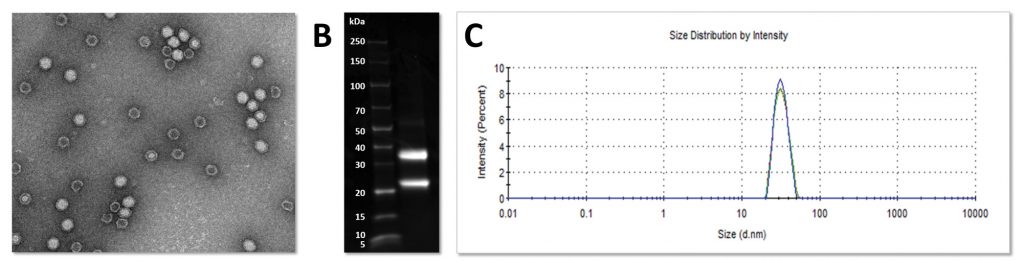
After five to seven days following infiltration, the leaves are harvested and the VLPs are extracted and purified to homogeneity using a bespoke, scalable, multistep process. Using the Hypertrans system we have produced up to 400 mg of CPMV VLPs per kg FW leaf
tissue, with 99% purity (see Figure 1).
In addition, antibody production in plants has been used in the treatment of diseases including Ebola, HIV, and Streptococcus infection. We are currently evaluating our own biosimilar versions of commercial antibodies, like bevacizumab. Bevacizumab is a recombinant humanized monoclonal immunoglobulin G1 antibody (IgG1) sold under the commercial name Avastin® and produced by Genentech.
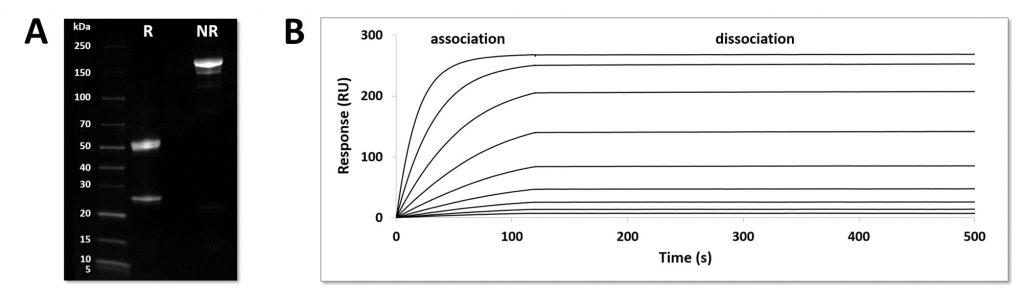
Our pEAQ-HT construct for the antibody contains both heavy and light chains in the same plasmid and after developing a plant specific purification process we are able to produce up to 200 mg/kg FW with the same bioaffinity for its target protein, VEGF-A, as the mammalian cell produced antibody (see Figure 2). Due to the inherent scalability of plant-based protein production, combined with the transient nature of the Hypertrans system, this platform can be used for antibody and vaccine generation where speed and batch-to-batch consistency are essential.
The Hypertrans system can provide a number of benefits for producing therapeutic and nontherapeutic protein products due to its reliability, high yield, low production costs, scalability, and feasibility of producing biosimilar products with excellent biocompatibility.
Albor Dobon Alonso, PhD, is senior microbiology scientist at Leaf Expression Systems.

