Michael Grauf R&D scientist Sartorius Stedim Cellca
Bertille Lagrange R&D scientist Sartorius Stedim Biotech
Klaus Schone Application Development Manager Sartorius Lab Instruments
Comparison of Clarification Principles: Centrifugation & Diatomaceous Earth
Abstract
In this study, it was tested whether the body-feed filtration method using diatomaceous earth (DE) as filter aid lead to different monoclonal antibody (mAb) and filtrate characteristics, in regards to five parameters analyzed compared to the well-established centrifugation method. The parameters investigated were reduction of turbidity, recovery of mAbs, mAb molecular weight, charge heterogeneity, glycosylation pattern, and over all work efficiency.
The heterogeneous sample pool comprised of various mAb products (two IgG1, IgG2, fc fusion protein, and bispecific antibody) and different cultivation conditions with cell densities between 38.3 x 105 and 163.6 x 105 cells/mL, turbidities between 557 and 1431 NTU and protein titers between 0.17 and 8.84 mg/mL.
All samples were harvested by both the body-feed filtration method using the Sartoclear® Dynamics Lab P15 kit and by the traditional centrifugation method, followed by sterile filtration. As a result, it could be shown that both cell culture harvest methods lead to similar results in all parameters but one. While leading to high recoveries of unaltered mAbs as well as sufficient turbidity reductions of the cell culture fluid (CCF), the DE method reduced the processing time by more than half.
Introduction
mAbs have been used successfully for years as therapeutic agents for different pathologies, e.g., various types of cancer and autoimmune diseases. They have become one of the main growth drivers of the pharmaceutical industry with a market size of $56.4 billion in 2012 and which is expected to reach $122.6 billion by 2019.1
However, antibody treatments are significantly more cost-intensive compared to drug therapies with chemically defined small molecules. This is due to the elaborate development and production processes associated with mAbs. The reduction of costs per treatment course is becoming increasingly important because of the continuous addition of new therapeutic antibodies while the budgets of national healthcare systems are simultaneously being limited.2
As a result, attempts are being made both to increase the yield of antibodies per production volume and to reduce the use of capital investments through shortened development periods.3 Cost reduction can often be achieved by continuous optimization of the individual steps.
The current study focuses on harvesting CHO cell cultures as well as on the quality of the yielded filtrate for analytical purposes. We present the use of DE as filter aid within a body feed clarification. The sample volumes considered are up to 50 mL, a typical scale for devices like spinner tubes, shaking flasks, and micro-bioreactors such as ambr15, a common setting for cell-line development, early process development, and media optimization.5,6
The method of harvesting plays an important role with regard to the examination of antibody-specific quality attributes because cellular material has to be removed adequately and antibodies as analytes can be influenced by the clarification process, e.g., by adsorption to filter material.
The conventional method of harvesting consists of a centrifugation step in which particles (cells, debris, etc.) with high density or large size are separated. A subsequent microfiltration step, where suspended particles are removed from the cell culture fluid (hereinafter referred to as “centrifugation”), is carried out.
The body-feed filtration method presented here enables the separation of complete cells and coarse debris by using DE while suspended particles are removed by microfiltration at the same time.
In the following, we demonstrate that the centrifugation and the body-feed filtration method are comparable with regard to particle depletion, mAb recovery, charge heterogeneity, and glycosylation pattern. Additionally, this work shows the efficiency of work for both methods. A heterogeneous sample pool with different types of mAb and cultivation methods was compiled to obtain a comprehensive statement.
Body-Feed Filtration by Diatomaceous Earth
Sartoclear Dynamics Lab products base their filtration principles on body-feed filtration where a filter aid is used to form a porous cake with the suspended solids in a dead-end filter. In a conventional filtration, the cake layer solely formed by biomass is compressible and becomes rapidly impermeable, which leads to filter clogging (Figure 2, left). In contrast to that, the filter aid applied for body-feed filtration creates a nearly incompressible filtration cake, which stays porous and prevents blockage over the complete filtration (Figure 2, right).
Inspired by the blood and plasma industry, all Sartoclear Dynamics Lab Products operate in body-feed mode using DE as filter aid. The use of DE as a filter aid in fractionation of human plasma was reported for the first time in the middle of the last century7.
Today DE is well-established as filter aid and in 2015, 75 % of the U.S. diatomite production was produced for filtration application8.
DE is gained from different species of extinct and living diatom algae, where their fossilized skeleton deposited on lakebeds consist of nearly pure amorphous silica (SiO2).9 The wet mixture is dried and clarified via calcination up to 1,000°C or flux-calcination up to 1,200°C to remove organics and partially volatilized metal ions, while the temperature treatment impacts the porosity of the gained particle agglomerates (Figure 1).
For biopharmaceutical applications a high level of purity of the DE is required. The Celpure® DE (Advanced Minerals Corporation) family is highly purified DE, where impurities are removed before calcination, making this purity grade specially designed for cGMP pharmaceutical processing.
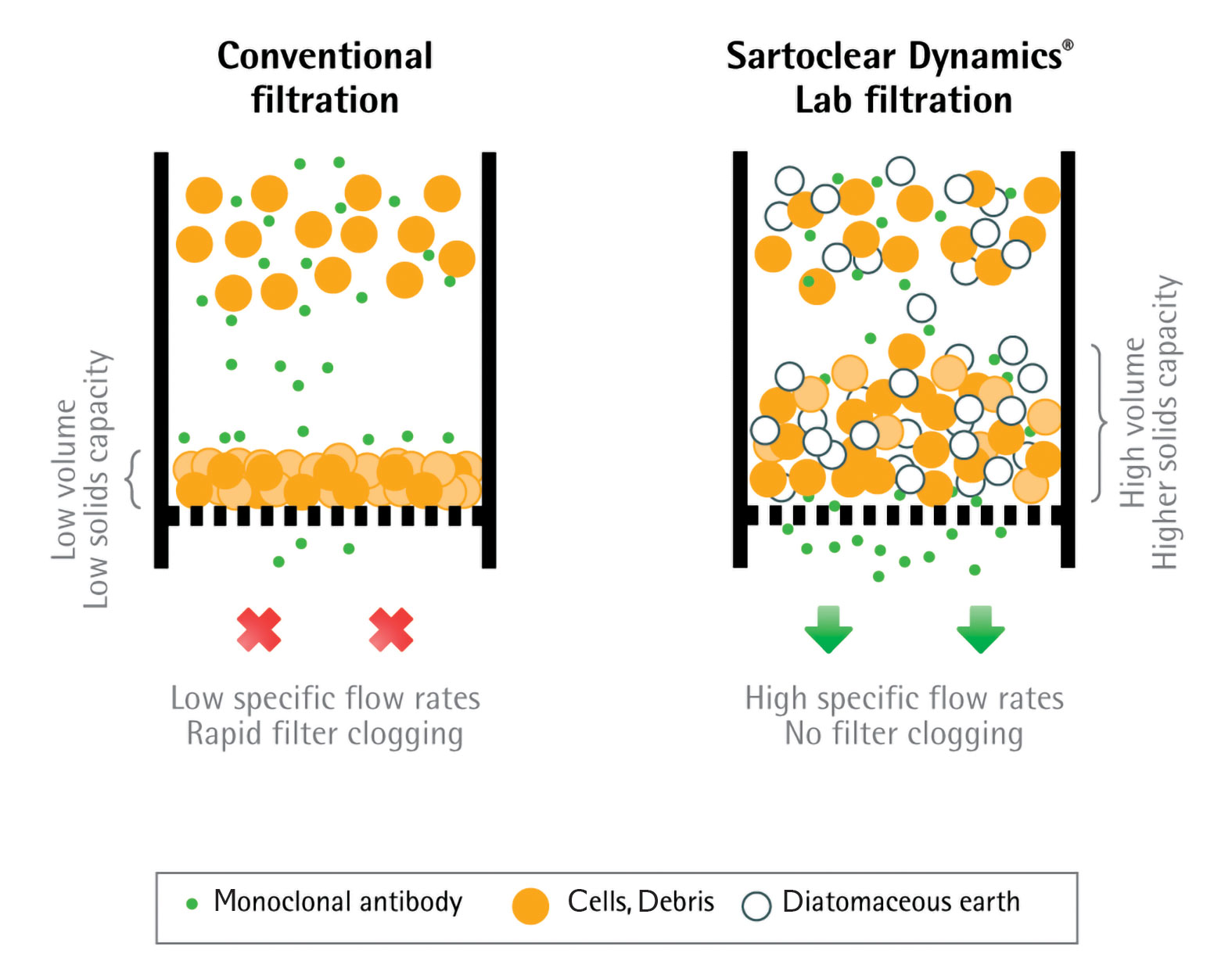
Figure 2. Principles of clarifying cell cultures by using the conventional (centrifugation) and the body-feed filtration (DE) method utilized by Sartoclear Dynamics® Lab.
Methods and ResultsCHO Cell Culture and Target Proteins
To create stable cell lines, CHO DG44 cells were transfected by electroporation and cultivated under selective conditions for three weeks followed by an amplification step with 30 nM MTX for an additional three weeks. Stable individual cell pools were then expanded and clones were generated by FACS. Clones were analyzed for growth performance and product concentration via fed-batch studies and genetic stability was evaluated during nine-week stability studies, including stability fed-batches (initiated at t = 2 weeks, t = 5 weeks, and t = 9 weeks, respectively).
In 11 cultivation batches, seven combinations of target proteins and cultivation methods were used (Table 1). In addition to 125 mL and 1,000 mL shaking flasks (SF), 5-L stirred bioreactors (UniVessel, Sartorius) were applied. As target proteins, CHO cell lines expressing different types of antibodies or antibody-derived products (IgG1, IgG2, Fc fusion, bispecific antibody) were selected. Cell density and viability were examined with a Vi-CELL XR system from Beckman Coulter.
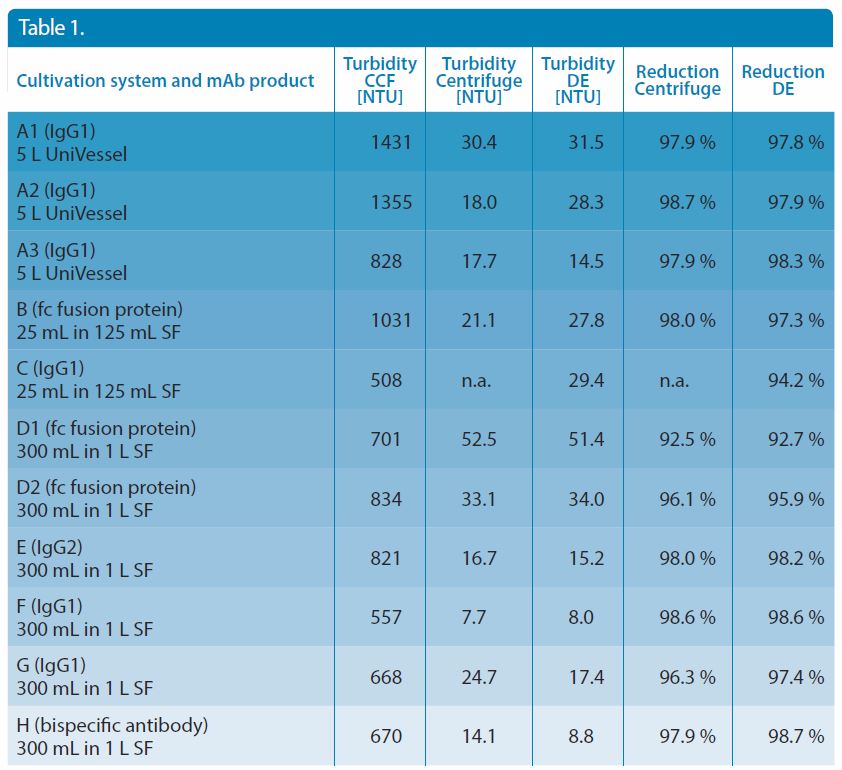
Table 1. Overview of cultivation system, mAb product, turbidity of the cell culture fluid (CCF) and cell culture fluid harvested by centrifugation and by DE, and relative reduction of turbidity by centrifugation and by DE.
Cell Culture Harvest
All cell culture batches (CCF) were harvested after 14 days and clarified in parallel case by centrifugation and DE method. For the clarification, sample volumes between 19 and 31 mL were used.
The cell culture samples (approx. 30 mL, Falcon tubes) were centrifuged for 5 min at 4,500 g and 20°C. Subsequently, the supernatant was filtered with a syringe filter (Minisart Highflow, 16532-GUK, Sartorius) with a pore size of 0.2 μm. Depending on the sample, a varying number of filters (1–3 devices) had to be used due to blockages.
The cell culture samples were clarified with the Sartoclear Dynamics Lab P15 (Sartorius). For this purpose, the cell culture suspension was taken with the syringe prefilled with DE and, after a short resuspension, filtered. The P15 is a kit comprising a 20 mL syringe prefilled with DE powder and a syringe filter equipped with a high-purity quartz microfiber prefilter and a 0.2 μm final filter made of polyethersulfone. The filling tube is useful for harvesting cell-culture suspension from ambr15 bioreactors.
Turbidity and Particle Depletion
The centrifugation and DE method was investigated for its ability to remove cell components from the CCF. For this purpose, we determined the turbidity values before and after clarification (Table 1) by the TurbiCheck WL turbidimeter (Lovibond, white light source). To remain in the linear measuring range between 1 and 1,100 NTU, the samples were diluted with one-fold DPBS before turbidity measurement. Afterwards, the reduction of turbidity was determined by calculation of the rato of values from harvested and unharvested samples.
mAb Protein Titer and Recovery
The mAb titer was determined in the CCF before and after the harvest procedure using the Octet QKe system equipped with the protein A biosensor (ProA) from FortéBio without any interfering sample preparation. Due to the diversity of samples, titers were achieved in a range of 0.17 to 8.84 g/L prior to harvest. Table 2 shows the measured titers and calculated recovery values.
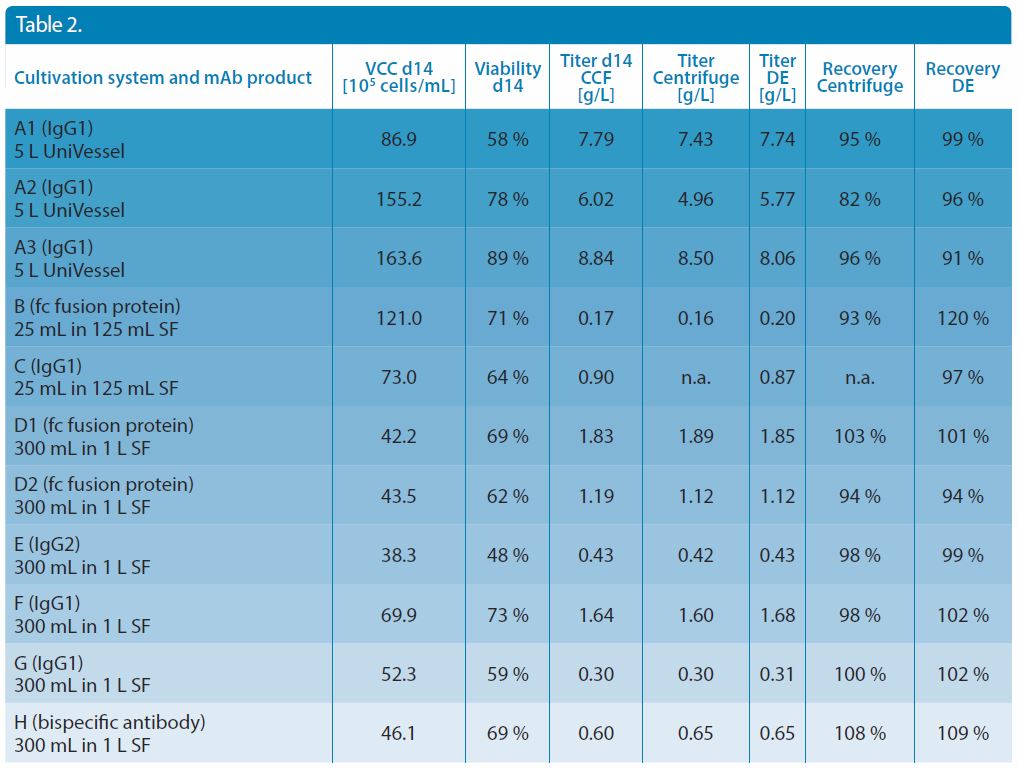
Table 2. Overview of cultivation system, mAb product, viable cell concentration (VCC) after 14 days cultivation, mAb titer for the cell culture fluid (CCF) and the clarified cell culture fluid (centrifugation and DE), and recovery rate (centrifugation and DE).
Glycosylation Pattern, Charge-Heterogenity, and Protein Size
The clarified samples were purified by means of Protein A using HP Spin Trap Columns or PreDictor MabSelect SuRe plates from GE Healthcare. For this purification step we used 1 x PBS as binding buffer, 100 mM citric acid, pH 3.0 as elution buffer and 1 M TRIS-HCl pH 9.0 as neutralization buffer.
Subsequently, the buffer was replaced by WFI using an ultrafiltration device (Vivaspin 500, 10 kDa from Sartorius). The NanoPhotometer® N 50 of Implen was used to adjust the protein concentration for the following investigations.
To determine the carbohydrate structure, charge heterogeneity and molecular weight of the targeted mAbs the LabChip GXII Touch 24 from Perkin Elmer was used. The following assays were used for the respective attributes.
Glycosylation Pattern
The relative amount of nine major glycosylation species was examined for six antibody products from the clarified samples. In order to perform the investigation, the Release and Labeling Kit and the HT Glycan LabChip Reagent Kit were used in which the N-glycans were removed enzymatically (PNGase-F), fluorescence-labeled and then analyzed by capillary electrophoreses with laser induced fluorescence (CE-LIF).
The results obtained were compared with regard to the clarification method chosen. Only peaks with a relative area of ≥ 2.5% of the total area were considered.4 Taken together, the composition of the glycosylation patterns are similar regardless of the clarification method. Only slight differences were observed in a few cases such as the presence (at centrifugation) or absence (at DE) of G2 forms for the product IgG1 (Figure 3).
Based on the current data, it can be concluded that the composition of the glycosylation pattern is not significantly influenced by the harvest method.
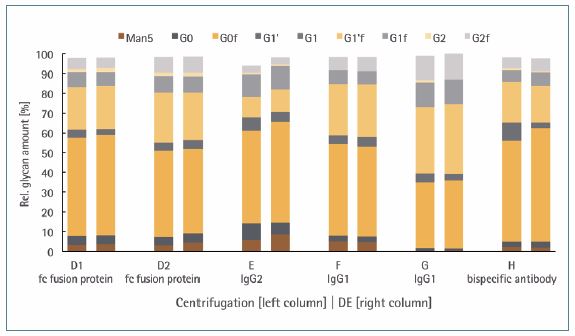
Figure 3. Relative amount [%] of nine main glycan pattern (Man5, G0, G0f, G1’, G1, G1’f, G1f, G2, and G2f) determined for the mAbs fc fusion protein, IgG1 (two products), IgG2, and a bispecific antibody. The antibody samples were taken from the filtrate obtained by centrifugation or DE method.
Charge Heterogeneity
The influence of the clarification method on the compostion of the charge variants was investigated for the target mAbs IgG2 (E) and IgG1 (F) (Figure 4). For this purpose, the prepared samples were adjusted to a protein concentration of 2 μg/μL and the relative distribution of charge variants was determined with the LabChip HT Charge Variant Assay.
A pH gradient allows first the elution of the basic, then the neutral and finally the acidic variants of the target mAbs. The coefficient of variation of the method is given by CV < 5% and this limit served as an indicator whether the clarification method influences the distribution of the charge variants. For both target mAbs, no significant difference in the charge distribution has been found as a function of the clarification method.
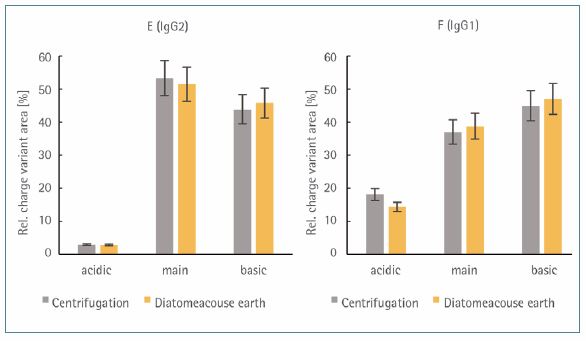
Figure 4. Relative area [%] of acidic, neutral (main), and basic charge variants of IgG2 (E) and IgG1 (F). Error bars show the standard deviation (2s) based on a CV < 5 % of the LabChip HT Charge Variant Assay.
Protein Size
Five samples were examined in regard to the size of the total antibody protein as well as their light and heavy chains by reduced and non-reduced conditions in the presence of SDS. For this purpose, prepared samples were adjusted to a concentration of 0.5 μg/μL and the molecular weight was determined using the Lab Chip “Protein Express Assay Reagent Kit” within a size range of 14 kDa to 200 kDa.
The aim was to get an indication about the identity of the target mAbs and possible impurities in the samples post purification shows the expected molecular weight of the complete mAb protein and their subunits. Both the molecular weight of the total mAb proteins as well as all mAb subunits for all the examined samples were confirmed (Figure 5). Moreover, no indication of impurities was observed.

Figure 5. Molecular weight of fc fusion proteins, IgG2, IgG1 and IgG1 (bispecific antibody) by reduced and non-reduced conditions.
Work Efficiency
To compare both clarification methods regarding their efficiency of work, the total handling time was exemplary measured for processing of one and 48 samples which are obtained from ambr15 bioreactors (Figure 8). In detail, the work steps were categorized as follows: “transfer from bioreactor to centrifugation tubes,” “setup time for centrifugation,” “centrifugation,” “setup time for filtration,” and “filtration” (data not shown).
Discussion
For the comparison of the conventional centrifugation method with the DE, a heterogeneous sample pool was examined. The aim of this study was to evaluate whether the DE cell-harvest method has an effect on the produced mAb compared to the traditional and established centrifugation method.
The investigated parameters of the study were particle depletion, product recovery, charge heterogeneity, glycosylation pattern, and protein size distribution. The main task of harvest is the separation of the cellular components from the CCF with mAbs. During this step, the product titer and the critical quality attributes should be as unaffected as possible.
The removal of cell components, which appear as insoluble particles, was determined by nephelometric turbidity. No differences in turbidity reduction could be observed, irrespective of the turbidity of the CCF ranging between 457 and 1431 NTU prior to harvest (Figure 6b). The turbidity in the filtrate was 8.0–51.4 NTU. These results indicate an equally effective removal of the coarse and fine particles from the cell culture suspension using both methods (Figure 6a) and are considered as suitable for further purification steps.
The recovery of the mAbs was investigated for various antibody types. The values vary between 91% and 109% within an acceptable range. Neither the harvest method nor the sample type, including antibody type and cultivation condition, seemed to exert an influence on product recovery.
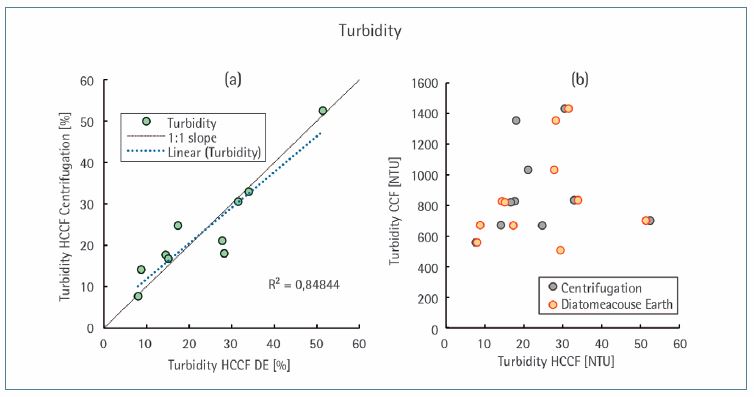
Figure 6. Turbidity of the harvested cell culture fluid (HCCF) clarified by both the centrifugation method and diatomaceous earth (DE). Both clarification methods are compared regarding the turbidity of the HCCF (a) and with the turbidity of the (unharvested) cell culture fluid (CCF) (b).
It should be mentioned, that even though the recovery yielded from the diatomaceous method seemed to be slightly higher, the significance of this observation has to be investigated in further experiments. However, it has been shown that the recovery remains unaffected in the investigated range of 0.17–8.8 g/L (Figure 7). Importantly, a low titer does not lead to a reduced recovery, which suggests relatively low adsorption effects.
During the expression of mAbs by mammalian cells, various post-translational modifications, e.g., glycosylation, can cause the generation of heterogeneous antibody isoforms which are different in their biophysical nature. To be able to analyze the range of variety of antibody isoforms, the clarification method should not affect their relative composition. In particular, differences in net charge could possibly lead to an altered adsorption and consequently to an undesirable depletion of certain antibody isoforms.
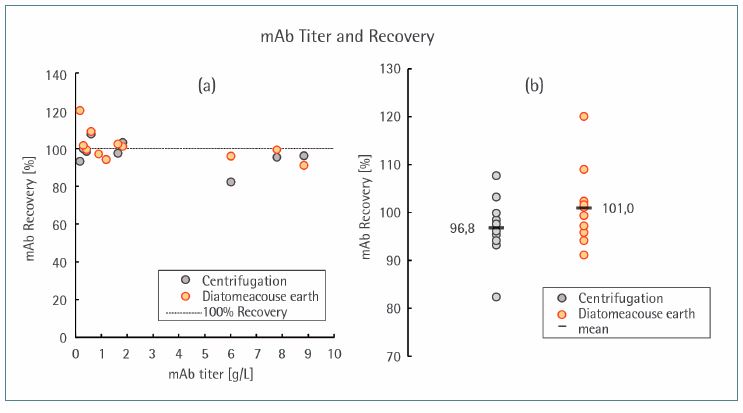
Figure 7. Recovery of mAbs in relation to their CCF titers (a). Samples were obtained from the filtrate after clarification by centrifugation or diatomaceous earth. Distribution of mAb recovery in relation to the clarification method chosen (b).
To get a deeper insight if the harvest method shows an impact on the relative isoform composition, both the charge heterogeneity and the glycosylation patterns were investigated for individual antibodies. For both the diatomaceous and the centrifugation method, we could not determine a selective reduction of antibody isoforms. A different net charge does not cause a significant change in antibody adsorption.
This finding is supported by the comparison of glycan pattern post harvest which shows no serious differences between the methods investigated.
The advantages of the body-feed filtration method are reduction of handling time and equipment, especially since a centrifugation step is not required. This is particularly important for an increased number of samples, especially due to the halved handling time for the final filtration step.
In comparison to the centrifugation method, the use of the body-feed filtration method with the Sartoclear Dynamics Lab P15 resulted in a work saving of 65 % to 48 samples with a volume of 15 mL (Figure 8).
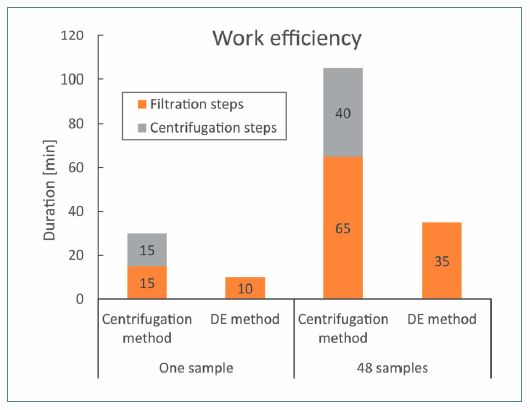
Figure 8. The duration of all centrifugation steps (including 5 min centrifugation time and setup time) and all filtration steps were determined for the clarification methods “Centrifugation” and “DE.” The duration was taken for one sample and for 48 samples.
Conclusion
In this study, we have shown the suitability of DE, as filter aid for the clarification of mAb samples dedicated for analytical purposes, in particular for cell culture samples with a turbidity below 1431 NTU and titers in a range between 0.17 to 8.84 g/L. By comparing the conventional centrifugation method with the DE method prior to membrane microfiltration, we could not find any significant difference with regard to turbidity depletion, mAb recovery, relative composition of charge heterogeneity, glycosylation isoform pattern, and antibody protein size.
In addition, while ensuring the requirements for harvesting mammalian cell cultures—such as high recovery of mAb and sufficient reduction of the turbidity during harvest—we conclude that the DE method utilized by Sartoclear Dynamics Lab can make a paramount contribution to reducing the overall workload and saving time.
Acknowledgement
Special thanks go to Dr. Christoph Zehe, Dr. Dirk Müller, and Dr. Noushin Delmdahl for reviewing the manuscript and for their constructive discussion on this subject.
Michael Grauf (michael.grauf@sartorius. com) is an R&D scientist at Sartorius Stedim Cellca, Bertille Lagrange serves as an R&D scientist at Sartorius Stedim Biotech, and Klaus Schöne is an application development manager at Sartorius Lab Instruments.
References
1. Shalini Shahani Dewan, Antibody Drugs: Technologies and Global Markets, BIO016J January 2015
2. Paul Cornes, The economic pressures for biosimilar drug use in cancer medicine, Targ Oncol (2012) 7 (Suppl 1):p.57–S67
3. Jozef Orpiszewski, Rapid Development of High-Quality, Robust Mammalian Cell Culture Manufacturing Processes, BioProcess International 14(8) September 2016
4. ProfilerPro Glycan Profiling Quick Guide LabChip® GXII Touch
5. Ingrid Lange, Sunil Chhatre, and Barney Zoro, Reducing Timelines in Early Process Development – Using a Multiparametric Clone –selection and Feed-Optimization Strategy, Bioprocess International 12(10) November 2014
6. Delouvroy et al.: ambr™ Mini-bioreactor as a high throughput tool for culture process development to accelerate transfer to stainless steel manufacturing scale: comparability study from process performance to product quality attributes. BMC Proceedings 2015 9 (Suppl 9):P78.
7. Cohn EJ, et al. Preparation and Properties of Serum and Plasma Proteins, IV: A System for Separation into Fraction of the Protein and Lipoprotein Components of Biological Tissue and Fluids. J. Am. Chem. Soc. 68, 1946: 459-4756
8. Crangle Jr. RD. Diatomite: Statistics and Information. US Geological Survey, Mineral Commodity Summaries 2013: 52-53; (version dated: Thursday, 23-Feb-2017 12:44:32 EST)
9. Antonides LE. Diatomite. US Geological Survey, Mineral Commodity Summaries 24, 1997:1-4
Keywords or Phrases
CHO cell culture, mAb, harvest, centrifugation, diatomaceous earth, turbidity, protein recovery, charge variants, glycosylation, protein size, work efficiency
Abbreviations
mAb Monoclonal Antibody
CHO Chinese Hamster Ovary
NTU Nephelometric Turbidity Unit
DE Diatomaceous Earth
cGMP current Good Manufacturing Practice
MTX Methotrexate
FACS Fluorescence-Activated Cell Sorting
PBS Phosphate Buffered Saline
TRIS 2-Amino-2-(hydroxymethyl) propan-1,3-diol
SDS Sodium Dodecyl Sulfate







