While surveilling the worldwide spread and evolution of a deadly bacterium, researchers at the Wellcome Trust Sanger Institute uncovered a surprising vulnerability. The bacterium, enterotoxigenic Escherichia coli (ETEC), differs from strain to strain much less than had been expected. In particular, it turns out that strains from relatively poor countries around the world—Latin America, Africa, and Asia—have a limited repertoire of colonization factors, surface proteins that enable the bacteria to attach to human stomach lining.
Each year, ETEC, the most common cause of childhood and travelers’ diarrhea, causes 400 million cases of diarrhea and kills 400,000 people. The lethal bacterium had been feared to vary widely across the world, but a new large-scale study suggests that many of the bacterium’s strains spread globally from a common source. As a result, there may be commonalities among ECTC strains that make it possible to target multiple strains with a single vaccine.
This encouraging result appeared November 10 in the journal Nature Genetics, in an article entitled, “Identification of enterotoxigenic Escherichia coli (ETEC) clades with long-term global distribution.”
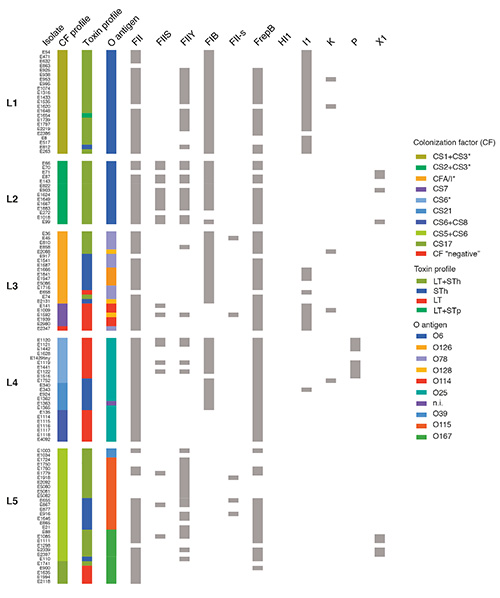
“The genes encoding the enterotoxins and most of the colonization factors are located on plasmids found across diverse E. coli serogroups,” wrote the paper’s authors. “Whole-genome sequencing of a representative collection of ETEC isolated between 1980 and 2011 identified globally distributed lineages characterized by distinct colonization factor and enterotoxin profiles.”
What’s more, the genetic information that helps the lineages infect humans appears to have been acquired by an individual bacterium fairly recently. It was then passed to multiple lineages, which now appear to “harbor chromosome and plasmid combinations that optimize fitness and transmissibility.” The success of the lineages is attributed, in part, to global travel.
According to the researchers’ data, the emergence of the globe-trotting lineages probably took place between 51 and 170 years ago, a finding that suggests that the lineages are stable and unlikely to become quickly resistant to vaccines.
“This research strengthens our belief that it is possible to target a broad range of ETEC groups with one vaccine,” said Professor Gordon Dougan, senior author from the Wellcome Trust Sanger Institute. “By targeting the most prevalent colonization factors in these lineages, we stand a chance of developing a vaccine that will reduce the disease burden caused by this bacterium.”
Detailed genetic information on each lineage has been made publicly available to the research community, enabling scientists to learn more about the bacteria and to track its future spread and evolution. Among other studies, researchers will use this data to learn more about the 130 ETEC samples that had colonization factors that had never been identified before.
“This dataset is exceptional,” asserted Astrid von Mentzer, first author from the University of Gothenburg and the Sanger Institute. “Researchers around the world will be able, from these whole-genome sequences, to draw out data about the virulence profiles and other proteins that may be important for virulence and that these strains could have in common. All of this research helps us to identify the shared weaknesses in ETEC bacteria that we can exploit with vaccines.”