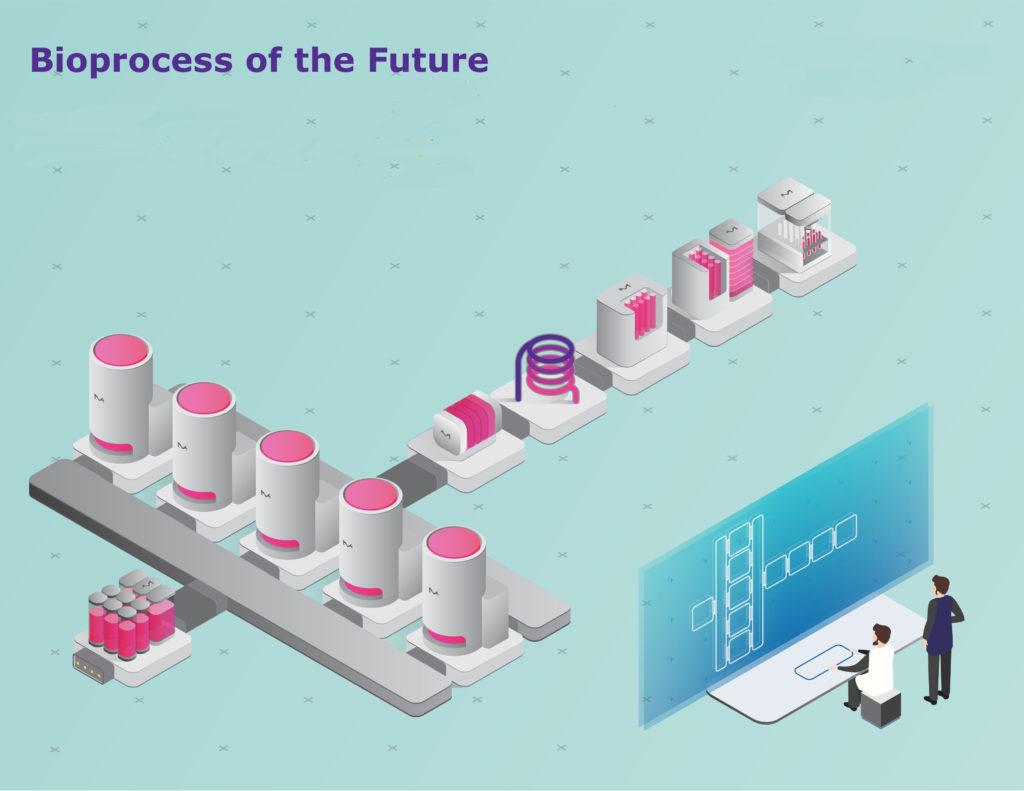
Bioprocessing 4.0 (or Biopharma 4.0) is becoming more tangible every day; according to Darren Verlenden, head of bioprocessing at MilliporeSigma. He adds that the company has multiple initiatives and programs that are defining the application of Industry 4.0 principles for our products, technologies, and services. “The industry is at a convergence of digital and process technologies that offers significant value beyond incremental improvement within individual silos,” he said.
Verlenden described the digital environment as consisting of advanced automation platforms, interconnected bioprocessing systems, operators, and facilities, advanced data and process analytics, and finally, building bridges between end users, manufacturing sites, and suppliers.
“We recently acquired the ProcessPad™ data management and analysis platform for real-time process monitoring and reporting,” he continued. “This browser-based platform provides on-demand access to data and works across a full manufacturing network to visualize and analyze process performance and outcomes.”
Today, manufacturing processes collect vast amounts of data—in some cases beyond one billion data points per batch. The problem is not gathering data but gaining from it the best and most comprehensive understanding. Verlenden sees a critical role for artificial intelligence and machine learning to provide advanced algorithms for pattern matching, anomaly identification, deviation investigation, and application of mechanistic modeling to full bioprocesses. “The Bioprocessing 4.0 principles and follow-on technologies are already being seen; however, there is a trickle-down impact to the bench scale,” he explained.
Verlenden maintains that lab equipment and systems can be connected to centralized data repositories on which advanced analytics tools can be run. The application of hybrid mechanistic/empirical models using machine learning will limit the number of experiments that need to be run during process development and allow in silico scaleup and simulation in order to speed time to clinic and market.
“With Biopharma 4.0, time spent in interpretation of data will decrease, and human-machine interfaces will be more fluid with biometric devices that allow easier capture of information,” predicts Verlenden. “Intensified, convergent processing will allow biomanufacturers to deliver therapies to patients faster and at lower cost.”

