Drug resistance continues to be a significant problem associated with many cancer therapies. Of the 565,650 cancer deaths projected in the U.S. in 2008, a large percentage will involve patients who develop resistance to chemotherapeutics, radiation therapy, biologic agents, and targeted therapy regimes.
In recent months, the role of the PI3K/Akt/mTOR signaling pathway in chemoresistance and cancer cell development has been the subject of multiple research efforts. As a result, new levels of attention have been focused on this pathway as a target in disease progression and drug development.
These research efforts have yielded novel insights into the role of the PI3K/Akt/mTOR pathway as a potentially promising target to reverse resistance to many leading cancer therapeutics. Recent research also indicates that Akt-activation inhibition may play a role in the development of HIV therapies.
Treatment failure due to chemoresistance can occur for several reasons. One common cause is cell mutation, in which cancer cells not initially killed by chemotherapy mutate and become resistant to the chemotherapeutic. Gene amplification, the replication of a gene that decreases the effectiveness of a particular chemotherapeutic, is another common cause of chemoresistance.
In other cases, cancer cells may counter the influx of a chemotherapeutic by draining the drug from cells, deactivating the protein that transports the drug across cell walls, restoring DNA breaks, or developing some other mechanism to deactivate the drug. Physicians often prescribe chemotherapeutics in combination to avoid development of resistance to one agent. Developed resistance to one drug, however, often leads to resistance to other chemotherapeutics.
Activation of protein kinase B (PKB or Akt), a key component of the PI3K signaling pathway, is known to promote cancer cell growth and survival as well as resistance to therapy. Akt pathway activation is often correlated with loss of PTEN (phosphatase and tensin homolog deleted on chromosome ten) function.
PTEN normally helps promote and regulate cell-division cycles, inhibiting uncontrolled cell growth by signaling cells to stop dividing and die. As part of this regulating function, PTEN suppresses activation of the PI3K/Akt/mTOR pathway. Loss of PTEN function can lead to hyperactivation of Akt, which has been linked to inhibition of apoptosis, a higher risk of cancer relapse, and increased resistance to widely used cancer therapeutics including trastuzumab and cisplatin.
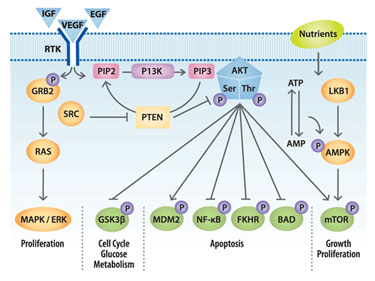
Akt as Master Regulator
Akt appears to be an especially pivotal protein, since when it is activated it regulates the stage of the cell cycle, apoptosis, glucose metabolism, growth translation, angiogenesis, and overall cellular survival. Akt exists as three isoforms, Akt1, Akt2, and Akt3, apparently with common but somewhat different normal functions.
For example, Akt1 seems to have a role in brain development, Akt2 in glucose regulation, and Akt3 in skeletal muscle and liver tissue. This is important because inhibition of Akt may have unwanted side effects.
In cancer, hyperactivation of Akt has been found to correlate with many different cancer types—ranging from glioblastoma multiforme; breast, lung, and prostate cancers; melanoma; leukemias; and multiple myeloma. Activation of specific isoforms is often correlated with specific cancers. For example, enhanced activity of Akt1 is found in breast and ovarian cancers, Akt2 in ovarian and pancreatic cancers, and Akt3 in hormone-insensitive cancers such as breast cancer cell lines deficient in estrogen receptor and prostate cancer cell lines that are androgen insensitive.
It is also interesting that Akt activation alone is usually not sufficient to cause cancer and that other mutations are generally necessary to create the cancer phenotype. Because of the intricate pathways that surround Akt activation, it is not surprising that resistance to standard chemo- and radiation therapies are often associated with activated Akt. This has lead to studies using Akt-targeting drugs in combination with agents that have other specific targets such as the receptor-tyrosine kinase inhibitors.
Research Activities
Recently, new insights on the role of Akt in treatment resistance and cancer cell survival have supported a wide range of preclinical and clinical research activities targeting Akt inhibition. In October, researchers at the department of molecular and cellular oncology at the University of Texas announced that they were successfully able to overcome trastuzumab resistance in breast cancer by combination treatment with drugs that inhibited either Akt or mTOR, including VioQuest’s (www.vioquestpharm.com) Akt inhibitor, VQD-002.
In January, scientists at the H. Lee Moffitt Cancer Research Center in collaboration with VioQuest successfully reversed cisplatin resistance in ovarian cancer, again by the use of drugs that specifically inhibited Akt activation, including VQD-002. Based on promising preclinical data in lung cancer studies, the NCI is planning clinical trials involving Akt pathway inhibitors among lung cancer patients.
Interestingly, Akt-activation inhibition has also been demonstrated to have an effect on HIV disease progression. A study published in Retrovirology in January demonstrated that Akt inhibition significantly reduced HIV-1 production from long-living, virus-infected macrophages.
In the study, inhibitors were shown to hypersensitize infected macrophages to extracellular stresses, leading to cell death of infected macrophages without harming uninfected cells. These findings shed light on how infection uses the Akt pathway to establish the cytoprotective effect of HIV-1 infection and thereby protects a key viral reservoir.


