January 1, 2016 (Vol. 36, No. 1)
Disease Phenotypes in Human iPSC-Derived Cells
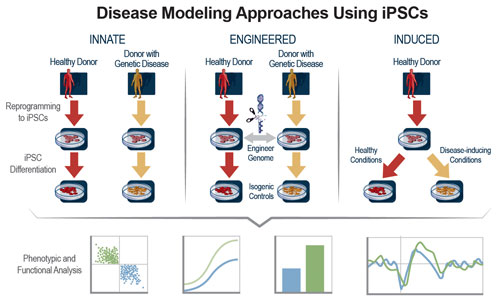
The ability to make induced pluripotent stem cells (iPSCs) from adult human somatic cells and use those iPSCs to produce any cell type in the human body opens new avenues for drug discovery. Human iPSCs can enable phenotypic screening efforts by supplying unlimited amounts of relevant cell types derived from diverse genetic backgrounds (Figure 1).
Newly available biobanks of high-quality iPSCs from hundreds of donors, representing a growing number of disease states, are making it easier to create robust phenotypic screening programs. By 2018, publicly available biobanked iPSC lines are expected to number in the thousands (Novak, 2015).
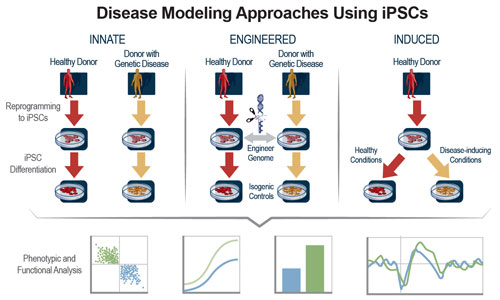
Figure 1. iPSC technology is leveraged to create healthy and disease-associated human cells for use in phenotypic screening assays. Following reprogramming and differentiation steps, iPSC-derived human cells exhibit expected phenotype and function, ensuring their relevancy for screening applications.
Current Model Systems
Traditionally, cell-based high-throughput (HTP) drug screening assays have utilized 1) immortalized cell lines that lack native cellular function intrinsic to normal cells, 2) primary cells isolated from donor tissues that are available in limited cell types and quantities or 3) human cancer cell lines that may not be representative of normal physiology. Human iPSC-derived differentiated cells have been shown to overcome these limitations.
Human iPSC-Derived Cell Models
Studies using Cellular Dynamics International (CDI) iCell® and donor-specific MyCell® differentiated human cells have demonstrated that iPSC-derived cell types overcome the problems associated with traditional cell models used in screening programs for innate, induced, and infectious diseases. Native iPSC-derived differentiated cells are compatible with a variety of HTP screening platforms, with genetic engineering at the iPSC stage or transfection of differentiated cells capable of generating cells that are amenable to the latest HTP readouts.
The intrinsic behaviors of iPSC-derived differentiated cells also facilitate the building of new drug discovery models. For example, cardiac hypertrophic phenotypes can be induced in CDI’s iCell Cardiomyocytes through disease-inducing culture conditions. These same phenotypes are seen in innate models of hypertrophy, such as familial hypertrophic cardiomyopathy (HCM), where disease donor iPSCs cultured under standard conditions result in cardiomyocytes that exhibit expected pathologies.
Importantly for drug discovery efforts, both the induced and innate pathologies can be assayed in phenotypic screening paradigms (Figure 2; Zhi, 2012; Carlson, 2013).
Recently, researchers have used iPSC-based induced and innate approaches to model and screen against diabetic cardiomyopathy—thus moving beyond rare monogenic disorders to more common polygenic diseases (Drawnel, 2014). Culturing iCell Cardiomyocytes in diabetic media induced a variety of structural and functional pathologies that were also observed in cardiomyocytes derived from Type 2 diabetic donor iPSCs cultured under standard conditions.
Similar to the example of hypertrophy shown above, phenotypic screens using both diabetic cardiomyopathy models returned overlapping hits across a variety of cellular pathways thus confirming the relevance of the different modeling paradigms.
The study of hepatic function is also of keen interest for disease modeling and drug development applications, and there is a growing body of literature on the use of iPSC-derived hepatocytes in both of these areas (Sampaziotis, 2015).
CDI’s iCell Hepatocytes, generated from a single donor background in limitless supply, offer superior functional stability over existing cell lines and primary cells (Lu, 2015). Their long term culture potential and the ability of iPSC-derived hepatocytes to assay population-specific responses to disease-causing agents and drugs makes them ideal for discovering therapies to treat infectious diseases of the liver, such as hepatitis and malaria.
In recent years, researchers at Roche and Bristol-Myers Squibb have reported the use of iCell Hepatocytes for modeling HCV infection (Mann, 2013; Friborg, 2015). Capitalizing on their long-term stability in culture, iPSC-derived hepatocytes have also been validated for HBV infection models (Shlomai, 2014; Chiao, 2014). In addition, infection of iCell Hepatocytes with the most clinically challenging malaria parasite species, Plasmodium vivax, represents a novel approach for identifying malarial therapeutic strategies and treatments (Ng, 2015). Altogether, these iPSC-derived hepatic models represent previously unavailable, consistent, donor-specific access to human biology.
iPSC models are also important to neurobiology drug discovery and screening efforts. Prior to the development of iPSC platforms, the creation of appropriate cell-based neurologic disease models was hampered by the inability to acquire even limited quantities of human primary cells. When primary cells could be obtained, they proved intractable to modern molecular techniques and failed to recapitulate both the latent and lytic phases of infection.
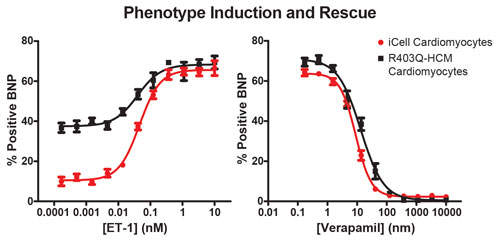
Figure 2. iCell Cardiomyocytes provide an inducible HCM model from “healthy” iPSC-derived cardiomyocytes and an innate model from R403Q-HCM donor cardiomyocytes. While both models can be induced to show cellular pathologies of hypertrophy (increased BNP expression) in response to endothelin (ET-1; left panel), the R403Q-HCM cardiomyocytes have innately elevated levels of BNP expression under control conditions and can be used in screening paradigms to find competitive antagonists of the pathology (right panel).
CDI’s iCell Neurons, derived from a single healthy donor, have been shown to support the lytic and latent phases of the varicella zoster virus (Figure 3) that causes chicken pox and shingles (Yu, 2013; Grose, 2013; Baird, 2015). These iPSC-derived neuronal cells are currently enabling mechanistic studies of viral reactivation as well as development of HTP screening assays.
iCell Neurons have also shown promise in Alzheimer’s disease (ALZ) biomarker analysis and drug screening applications. ALZ pathology can be induced in iCell Neurons by treating them with β-amyloid 1–42 (Ab1–42) aggregates. A phenotypic screen using these ALZ cells identified small molecules that rescued the phenotype. Follow-on assays provided additional evidence for the involvement of a new druggable pathway in ALZ (Xu, 2013).
Additionally, studies of innate ALZ pathologies were conducted using human neurons derived from genetically engineered iPSCs carrying one of two beta amyloid precursor protein (APP) alleles, A674V and A674T. These alleles have been linked with progression of and protection from ALZ, respectively. Results revealed distinct differences in APP processing and neural network activity, criteria which can both be exploited in the development of HTP phenotypic screening assays.
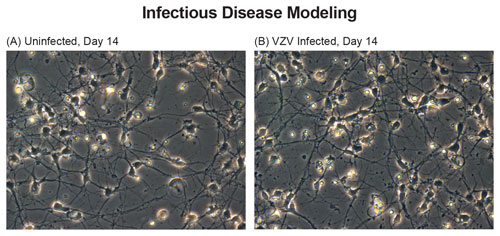
Figure 3. iCell Neurons infected with VZV produce a nonproductive infection, enabling molecular analysis of virus-neuronal interactions and viral reactivation. Phase-contrast of uninfected neurons (A) and VZV-infected neurons (B) are similar, with no cytopathic effect shown (Yu, 2013; Baird, 2015). This response is consistent with in vivo disease biology.
Conclusions
CDI’s iPSC-derived human cells enable more effective modeling and phenotypic screening applications. It is not necessary for a cell model to recapitulate the natural history of a disease in a dish. Instead, it is more important that the model includes a phenotype or biomarker that correlates with the disease state being studied.
CDI’s range of iPSC-derived human cells represents a wide variety of disease backgrounds and overcomes the limitations of conventional cell-based screening assays by delivering a consistent, reproducible and limitless source of tissue that is reflective of native human function. All of these factors combined indicate that the use of CDI’s iPSC-derived differentiated cells for phenotypic screening purposes will increase the likelihood of discovering more efficacious and safer drugs.
Blake Anson, Ph.D. ([email protected]), Susan DeLaura, Eugenia Jones, Ph.D., and David A. Mann, Ph.D., are product managers at Cellular Dynamics International, a FUJIFILM company.