Say freeze! Sitting in a cryogenic studio and waiting to be bombarded with electrons, two proteins, both part of a gene editing complex, were poised to be imaged for posterity. And then, as the cryo-electron microscopy machinery clicked and whirred, a double portrait emerged. It showed the CRISPR/Cascade system associated with a transposition system called TniQ.
Together, CRISPR/Cascade and TniQ form a power couple, one that can insert large genetic payloads into the genome without introducing double-strand breaks in target DNA. The couple made its presence felt just a few months ago, when researchers at Columbia University showed that the couple functions as a fully programmable, RNA-guided integrase. At the time, the researchers speculated about the mechanistic basis of the couple’s integrative functionality. More recently, in a study published December 18 in Nature, the scientists reported that they confirmed their mechanistic ideas by imaging the couple with cryo-electron microscopy.
In this paper (“Structural basis of DNA targeting by a transposon-encoded CRISPR–Cas system”), the researchers recalled, “We proposed that RNA-guided DNA targeting by Cascade could deliver TniQ to DNA in a manner compatible with downstream transpososome formation, and that TniQ might interact with Cascade near the 3′ end of the crRNA, consistent with RNA-guided DNA insertion occurring around 49 bp downstream of the protospacer-adjacent motif (PAM)-distal edge of the target site.”
The paper goes on to describe how the Columbia University scientists—who were led by Sam Sternberg, PhD, and Israel Fernandez, PhD, both assistant professors of biochemistry and molecular biophysics—succeeded in verifying their mechanistic expectations. First, the scientists purified the TniQ–Cascade complex, which they had previously determined to be encoded by the Vibrio cholerae Tn6677 transposon. Second, the scientists loaded the complex with a native CRISPR RNA (crRNA). Third, they determined the structure of the complex by cryo-electron microscopy.
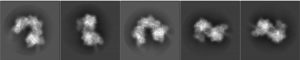
“The cryo-electron microscopy maps enabled de novo modelling and refinement of the transposition protein TniQ, which binds to the Cascade complex as a dimer in a head-to-tail configuration, at the interface formed by Cas6 and Cas7 near the 3′ end of the crRNA,” the authors of the Nature article detailed. “The natural Cas8–Cas5 fusion protein binds the 5′ crRNA handle and contacts the TniQ dimer via a flexible insertion domain.”
The structural model reveals that the complex is made up of two main sections that are arranged in a helical filament. The larger portion, Cascade, winds around and carries a guide RNA that it uses to scan the cell for a matching sequence in DNA. Once it locates and binds the target sequence, it threads the DNA strand through the TniQ “transposition” proteins that sit on the end of the complex and recruit other enzymes that help modify the DNA.
The Columbia scientists hope that their findings will guide efforts to improve the genome engineering capabilities of their Cascade/TNIQ system, which they call INTEGRATE, for “INsert Transposable Elements by Guide RNA-Assisted TargEting.”
“We showed in our first study how to leverage INTEGRATE for targeted DNA insertions in bacterial cells,” said Sternberg. That study, which used genetics and biochemistry to propose how the CRISPR machinery would functionally link to the transposition machinery—the molecules responsible for gene “jumping”—has now been shown to be correct, thanks to the capture of cryo-electron microscopy images, which led to the generation of atomic resolution models of the INTEGRATE system.
“These new images,” Sternberg declared, “explain the biology with incredible molecular detail and will help us improve the system by guiding protein engineering efforts.”
Many researchers around the world now use CRISPR-Cas9 to quickly and cheaply make precise modifications to the genome of a cell. However, most uses of CRISPR involve cutting both strands of the target DNA, and the DNA break must then be repaired by the host cell’s own machinery. Controlling this repair process is still a major challenge in the field, and undesired gene edits are often introduced inadvertently in the genome. Additionally, existing tools often perform poorly at inserting large genetic payloads in a precise fashion. Improving the accuracy of gene editing is a priority for researchers and is critical for ensuring the safety of therapies developed with this technique.
The new INTEGRATE system developed by the Sternberg lab can accurately insert large DNA sequences without relying on the cell’s machinery to repair the strands. As a result, INTEGRATE could prove to be a more accurate and efficient way of making certain gene modifications than the original CRISPR-Cas system that is widely in use. The new tool could also help scientists perform gene editing in cell types with limited DNA repair activity such as neurons, where attempts to use CRISPR have been comparatively less successful.
In addition to informing future engineering efforts, the structures highlight a possible proofreading checkpoint. Existing CRISPR technologies often suffer from so-called “off-target effects,” in which unintended sequences are promiscuously modified. The new structures reveal how Cascade and TniQ work together to ensure that only the correct “on-target” sequences are marked for DNA insertion. The researchers plan to further explore this checkpoint while developing the tool for new therapeutic approaches to disease.


