A new graphic from research organization Decision Resources Group illustrates countries with biosimilars pathways, biosimilars in clinical trials, and regions with marketed biosimilars.
The key data points of the above infographic from Decision Resources Group include:
• >280 biosimilars in the pipeline
• 17 biosimilar products approved in EEA (see the Table to the right)
• >20% annual increase in biosimilars in clinical trials
• 32 biologic molecules targeted by biosimilar developers
To download your own copy, click here.
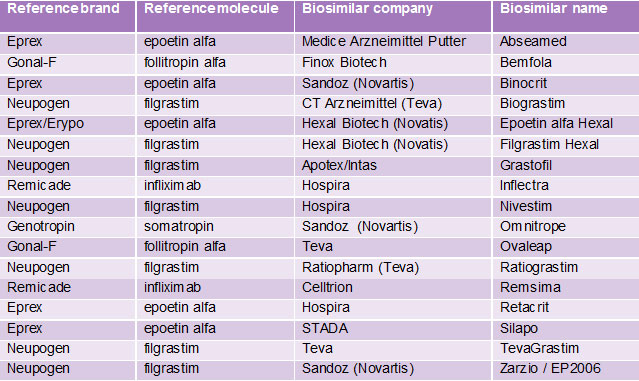
Table. The 17 approvals in Europe that are referenced in the sidebar of the infographic.



