Drug discovery efforts focused on the treatment of aging is worthwhile,” maintains Janet Lord, PhD, professor of immune cell biology from the University of Birmingham in the U.K. Speaking at the recent European Laboratory Research & Innovation Group (ELRIG) Drug Discovery Conference in Cambridge, U.K., Lord explained: “Aging badly is the biggest risk for many diseases such as heart disease and cancer and costs world economies between four and sixteen percent of their GDP.”
“A recent study suggests that every year that we can increase healthspan [a healthy lifespan] is worth an estimated $38 trillion to the global economy,” added Lorna Harries, PhD, CSO of Senisca.
According to Lord, our current therapeutic model of treating each condition or disease with a single drug can lead to polypharmacy in patients where those with multiple co-morbidities are taking four or more drugs. This can lead to drug interactions, unintended side effects, and potential toxicity which can seriously impact a patient’s life.
“Geroscience, the study of identifying and correlating specific markers and signaling pathways in cells and tissues to diseases of aging, could be the turning point because it will identify the core cause of many diseases,” said Lord. The application of geroscience was debated at ELRIG where a range of practicable technologies and strategies for studying aging was discussed.
In his keynote presentation, Ken Raj, PhD, principal investigator at Altos Labs, discussed how computational science and machine learning applied to epigenetics could help determine the pathways associated with cellular aging.

“In all human organs there are thousands of cytosine sites (CpG sites), whose methylation states change with age,” Raj told the audience. These change in individuals at different rates.” According to Raj, using machine learning, his colleague, Steve Horvath, PhD, professor of human genetics and biostatistics, David Geffen School of Medicine, UCLA, identified 353 CpG sites in DNA whose methylation states, when taken collectively, are best at predicting chronological age and created a model known as Horvath’s Epigenetic Clock.
Although trained on chronological age, this clock captures at least some aspects of biological age which may not necessarily be related to how old a person is in terms of chronological years of their lifespan.
“The CpGs of Horvath’s Clock are merely representations of the aging epigenome, just as a football team would not represent the entire population of a nation so these CpGs do not represent all cytosines comprehensively,” Raj added. “They are only a sampling of the epigenome, and much more will be gained by studying the whole epigenome itself. However, the clock is useful because the outliers that do not fall into the predictive pattern can provide valuable information and there is often a correlation with those that show accelerated biological aging and the onset of different pathologies, as well as the effects of lifestyle and environmental factors.”
As an example of how an epigenetic clock could be used, Raj discussed a recent study of basal cell carcinoma (BCC), published in Nature Aging. According to Raj, the onset of BCC does not align with a person’s chronological age. But using a specifically constructed epigenetic clock to measure DNA methylation (DNAmAge) the group at Altos could accurately measure the biological age of a range of skin cells in culture and track their aging allowing for use with in vitro experiments.
“In the lab epithelial cells in culture age much faster outside the body so we can study age-related hypo and hyper methylations more easily to determine the biological mechanisms of aging and the effects that potential anti-aging drugs such as rapamycin have at the cellular level,” concluded Raj.
Drugs and targets
“An aging population poses major medical, social, and economic challenges. If we could find methods to retard diseases associated with aging such as cancer and neurodegenerative diseases this would have a massive benefit to society,” commented João Pedro de Magalhães, PhD, professor of molecular biogerontology at the University of Birmingham, U.K., and CSO of YouthBio Therapeutics.
In his presentation, de Magalhães discussed tools available to identify genes and potential drug targets for age-related diseases. These included GenAge, a database which details genes related to longevity and/or aging in model organisms and humans, and DrugAge, a database of drugs and compounds that can increase longevity in animal models such as yeast, worms, flies, and mice. “GenAge is freely available to access and could be a useful source of druggable targets,” explained de Magalhães.
According to de Magalhães, there are more than 2,000 genes that modulate longevity and over 1,000 drugs and compounds that are associated with slowing the aging process. He stated that “Resveratrol, rapamycin, and metformin are the most studied longevity drugs, but the lifespan effects are much greater and more consistent for rapamycin than for resveratrol so this is of growing interest. Aging results from the progressive dysregulation of several molecular pathways, including nicotinamide adenine dinucleotide (NAD) metabolism and the mammalian target of rapamycin (mTOR) signaling. These areas are now the focus of study in many biotech companies that are developing anti-aging therapeutics.”
Harries provided a different approach to finding therapeutics to treat aging. According to Harries there are few pathways associated with diseases of aging at the level of total gene expression, and the majority of those are involved with mRNA processing.

“Ninety-eight percent of the mRNA transcriptome can make more than one product, depending on how the mRNA is spliced,” Harries pointed out. “Splicing is decided by regulatory proteins called splicing factors and these decline with age and is one of the reasons why cells become senescent.” To reverse the decline of splicing factors, Senisca is studying the use of oligonucleotide-based therapeutics.
At ELRIG, Harries presented in vitro data on how Senisca oligonucleotides could attenuate senescence and markers of fibrosis in human lung fibroblast cells. Harries also showed data to indicate that its oligonucleotides were active in human dermal fibroblasts, chondrocytes, and retinal endothelial cells.
“We currently have 40 oligonucleotides that target 14 novel and proprietary targets which yield up to 50 % less senescence in many cell types, as well as reduce DNA damage and inflammation,” Harries said. “Dysregulation of mRNA processing is now a druggable hallmark of aging. We believe our oligonucleotides can precisely target this and could have potential applications in treating age-related diseases such as idiopathic pulmonary fibrosis, osteoarthritis, and age-related macular degeneration (AMD).”
Challenges of developing anti-aging therapeutics
Speakers at the ELRIG conference agreed that developing drugs to target aging come with a unique set of challenges. According to Raj, determining therapeutic targets is difficult as aging is unlikely to be associated with a single enzyme or receptor. “Drug developers are good at finding enzyme inhibitors, you can guarantee if there is an enzyme, and they will develop a molecule that inhibits its action, as they are highly experienced at single molecule screening,” said Raj. “However, targets for aging will need cell-based assays which are very finicky but are required for measuring epigenetic readouts.”
Another issue noted was that regulatory bodies such as the FDA do not recognize aging as a disease so there is no clear clinical end-point to aim for when developing new drugs. According to Raj, “We cannot do lifespan studies for human clinical trials, or we will die before the results are in. But we can piggyback on pathologies associated with aging and use their development as a primary end-point.”
Measuring in vivo data on aging end-points
David Weinkove, PhD, co-founder and CSO of Magnitude Biosciences. discussed how to accelerate anti-aging drug discovery. “Finding treatments to slow diseases of aging do not fit into our classical pipeline model because there are not any good high-throughput screens,” he explained. “ Although cell-based assays and animal models are better they are expensive and have timelines that are too long for initial screens.”
According to Weinkove, the nematode worm C. elegans can provide an excellent in vivo drug discovery model. “C. elegans has many cell types found in humans, including epithelial, neuronal, and muscle cells, yet it only has a lifespan of two to three weeks and can be easily grown on a lawn of E. coli in Petri dishes. Also, mutations first found in C. elegans to increase their lifespan revealed a pathway that affects aging in mammals. Lifespan is different from healthspan, and we have found that worm movement is a better metric of improved healthspan.”
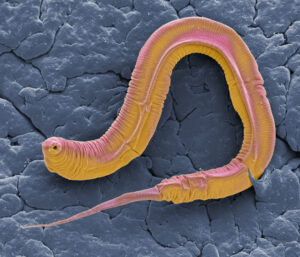
Worm movement is difficult to quantify by eye, noted Weinkove, so Magnitude Biosciences has developed an automated imaging system called WormGazer™. The system continuously monitors and provides data on worm movement noninvasively. It can monitor up to 1,000 worms per week and can be used in assays with different compounds that slow aging to determine efficacy, mode of action, and potential toxicity.
To demonstrate the application of WormGazer assays, Weinkove presented case study data at ELRIG from collaborative work with Gerostate Alpha, a U.S. biotech developing anti-aging compounds. The work showed that using the WormGazer assay, eight out of ten of Gerostate Alpha’s compounds were effective in increasing healthspan. He also presented data from a study with U.K.-based biotech, Five Alarm Bio, which demonstrated that their small molecule (designated PAB001) allowed worms to live more healthily for longer. This positive data helped Five Alarm Bio complete a seed investment round in February 2023, raising over £500K ($600K) to further enable their research.
“Using worm movement is a better functional end-point than lifespan as we believe it could translate to muscle issues and frailty both associated with aging,” said Weinkove. “Using our approach in parallel with cell-based assays means compounds can be assessed at scale and speed to provide solid data to support lead selection before being tested in more expensive in vivo animal models.”
According to de Magalhães, aging is plastic in animals. “Through a single mutation to their insulin signaling pathway, we can increase the lifespan of C. elegans worms by up to 10-fold and with mice we can double their lifespan. Additionally, with mice we can accelerate their aging by inducing DNA damage and we can inhibit growth hormone signaling to make mice grow less but become cancer resistant. These animal models indicate that it should be possible to alter a human gene and increase a human lifespan.”
According to de Magalhães, biotechs are now investing in anti-aging interventions with companies such as Calico, Unity Biotech, Juvenescence, and Altos leading the pack.
“If we can inhibit pathways associated with aging in humans with pharmaceutical interventions rather than gene manipulation it could lead to more effective treatment and a reduction in drug toxicity for older patients,” he concluded. “It would make sense to do this, and it would be game changing for healthcare.”
Sue Pearson, PhD, is a freelance writer based in the U.K.


