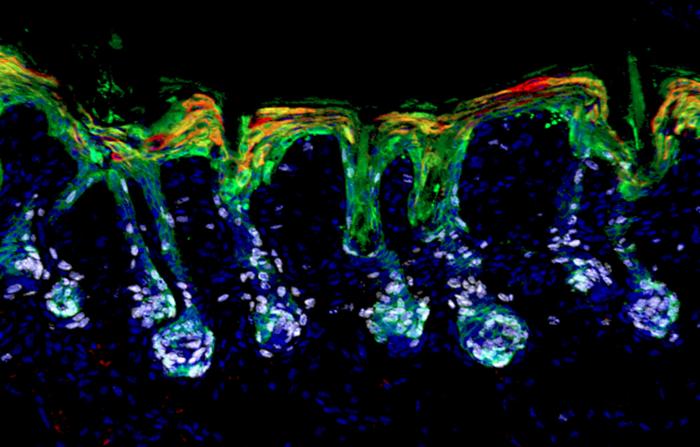
Tissue regeneration is regulated, in part, through specialized niches that hold stem cells. When perturbed, for example by scratches on the skin, the stem cells can move to areas that need repair. In order to do so, the cells enter a state of lineage plasticity.
Lineage plasticity “can act as a double-edged sword,” explained Matthew Tierney, PhD, a postdoc in the lab of Elaine Fuchs, PhD, professor investigator at the Rockefeller University. “The process is necessary to redirect stem cells to parts of the tissue most in need but, if left unchecked, it can leave those same tissues vulnerable to chronic states of repair and even some types of cancer.”
In a screen to identify key regulators of this process, researchers in the Fuchs lab found all-trans retinoic acid (atRA) as a vitamin A metabolite essential to restoring the cells’ physiological identity in culture. The findings shed light on lineage plasticity, with potential clinical implications.
This work is published in Science in the paper, “Vitamin A resolves lineage plasticity to orchestrate stem cell lineage choices.”
“Our goal was to understand this state well enough to learn how to dial it up or down,” said Fuchs. “We now have a better understanding of skin and hair disorders, as well as a path toward preventing lineage plasticity from contributing to tumor growth.”
Fuchs and her team screened small molecules for their ability to resolve lineage plasticity in mouse hair follicle stem cells (HFSCs), under conditions that mimicked a wound state. They found that retinoic acid was essential for stem cells to exit lineage plasticity and differentiate into hair cells or epidermal cells in vitro.
More specifically, the authors noted that by “layering transcriptomic and chromatin landscaping data with retinoid-activated nuclear retinoic acid receptor–DNA interactions, we identified targets that affect this outcome. Included in this downstream regulatory network was pioneer factor SOX9, which is known to directly up-regulate key hair follicle genes while silencing epidermal fates.”
“Through our studies, first in vitro and then in vivo, we discovered a previously unknown function for vitamin A, a molecule that has long been known to have potent but often puzzling effects on skin and many other organs,” Fuchs said. The team found that genetic, dietary, and topical interventions that boosted or removed retinoic acid from mice all confirmed its role in balancing how stem cells respond to skin injuries and hair regrowth. Retinoids did not operate on their own: their interplay with BMP and WNT signaling molecules influenced whether the stem cells should maintain quiescence or actively engage in regrowing hair.
The team also demonstrated that retinoic acid levels must fall for hair follicle stem cells to participate in wound repair—if levels are too high, they fail to enter lineage plasticity and can’t repair wounds—but if the levels are too low, the stem cells focus too heavily on wound repair, to the expense of hair regeneration.
“This may be why vitamin A’s effects on tissue biology have been so elusive,” Fuchs said.
One result of retinol biology remaining obscure for so long is that retinoid and vitamin A applications have long produced confusing results. Topical retinoids are known to stimulate hair growth in wounds, but excessive retinoids have been shown to prevent hair cycling and cause alopecia. The present study presents retinoids in a more central role—regulating both hair follicle and epidermal stem cells.
“By defining the minimal requirements needed to form mature hair cell types from stem cells outside the body, this work has the potential to transform the way we approach the study of hair biology,” Tierney said.
How retinoids impact other tissues remains to be seen. “When you eat a carrot, vitamin A gets stored in the liver as retinol where it is sent to various tissues,” Fuchs said. “Many tissues that receive retinol and convert it to retinoic acid need wound repair and use lineage plasticity, so it will be interesting to see how broad the implications of our findings in skin will be.”
The Fuchs lab is also interested in how retinoids impact lineage plasticity in cancer, particularly squamous and basal cell carcinoma. “Cancer stem cells never make the right choice—they are always doing something off-beat,” Fuchs said. “As we were studying this state in many types of stem cells, we began to realize that, when lineage plasticity goes unchecked, it’s a key contributor to cancer.”
Basal cell carcinomas have relatively little lineage plasticity and are far less aggressive than squamous cell carcinomas. If future studies demonstrate that suppressing lineage plasticity is key to controlling tumor growth and improving outcomes, retinoids may have a key role to play in treating these cancers.
“It’s possible that suppressing lineage plasticity can improve prognoses,” Fuchs said. “This hasn’t been on the radar until now. It’s an exciting front to now investigate.”