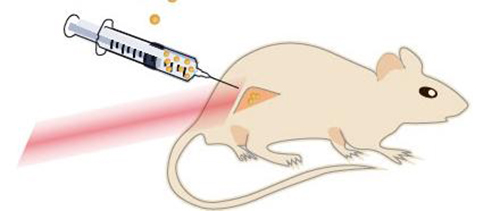
![The drawing shows a mouse with a cancerous tumor on its hind leg. The nanoparticles are injected directly into the tumor, which is then flashed with near-infrared laser light. Near-infrared laser light penetrates through the tissue well and causes no burn damage. [Kamilla Nørregaard/Panum Institute]](https://genengnews.com/wp-content/uploads/2018/08/121152_web4281121110-1.jpg)
The drawing shows a mouse with a cancerous tumor on its hind leg. The nanoparticles are injected directly into the tumor, which is then flashed with near-infrared laser light. Near-infrared laser light penetrates through the tissue well and causes no burn damage. [Kamilla Nørregaard/Panum Institute]
Injecting cancerous tumors with microscopic particles and then zapping them with lasers seems like an idea ripped straight from the pages of science fiction. Yet, that is precisely the treatment strategy which investigators at the Niels Bohr Institute and the Faculty of Health Sciences at the University of Copenhagen have been developing to kill cancer cells.
The researchers report in their findings, which were published recently in Scientific Reports in an article entitled “Single Particle and PET-based Platform for Identifying Optimal Plasmonic Nano-Heaters for Photothermal Cancer Therapy,” that the treatment has been tested on mice—with results demonstrating that tumors become considerably damaged.
“The treatment involves injecting tiny nanoparticles directly into the cancer,” explained senior study author Lene Oddershede, Ph.D., a biophysicist and head of the research group Optical Tweezers at the Niels Bohr Institute within the University of Copenhagen. “Then you heat up the nanoparticles from outside using lasers. It is a strong interaction between the nanoparticles and the laser light, which causes the particles to heat up. What then happens is that the heated particles damage or kill the cancer cells.”
![The experiments were carried out with nanoparticles of different sizes and structures. The first two of the series consisted of solid gold and the last consisted of a core of glass with a surface of gold. The beads were illuminated with near-infrared light with wavelengths of 807 nm and 1064 nm. The most effective nanoparticle was the gold-plated glass bead. [Kamilla Nørregaard/Panum Institute]](https://genengnews.com/wp-content/uploads/2018/08/121151_web5519912621-1.jpg)
The experiments were carried out with nanoparticles of different sizes and structures. The first two of the series consisted of solid gold and the last consisted of a core of glass with a surface of gold. The beads were illuminated with near-infrared light with wavelengths of 807 nm and 1064 nm. The most effective nanoparticle was the gold-plated glass bead. [Kamilla Nørregaard/Panum Institute]
Traditional cancer treatments like radiation and chemotherapy have major side effects because they affect not only the tumors, but also healthy parts of the body. A large interdisciplinary research project between physicists at the Niels Bohr Institute and doctors and biologists at the Panum Institute and Rigshospitalet set out to develop a new treatment that only affects cancer tumors locally and therefore is much gentler on the body. The project is called Laser Activated Nanoparticles for Tumor Elimination (LANTERN).
After experimenting with various biological membranes, the researchers tested the method on living mice. In those experiments, the mice were given cancerous tumors derived from laboratory-cultured human cancer cells. The research team then tested small nanoparticles between 80 and 150 nanometers (nm) in diameter (a nanometer is a millionth of a millimeter). The tested particles consisted of either solid gold or a shell structure consisting of a glass core with a thin shell of gold around it—in an attempt to determine which particles are most effective in reducing tumors.
“As physicists, we have great expertise in the interaction between light and nanoparticles and we can very accurately measure the temperature of the heated nanoparticles,” noted Dr. Oddershede. “The effectiveness depends on the right combination between the structure and material of the particles, their physical size, and the wavelength of the light.”
![The images show PET scans of a mouse with a large tumor (by the white arrow). The tumor is treated with nanoparticles, which are injected directly into the tumor and are then flashed with near-infrared laser light. The laser light heats the nanoparticles, thus damaging or killing the cancer cells (red arrows). [Kamilla Nørregaard and Jesper Tranekjær Jørgensen/Panum Institute]](https://genengnews.com/wp-content/uploads/2018/08/121150_web6656119657-1.jpg)
The images show PET scans of a mouse with a large tumor (by the white arrow). The tumor is treated with nanoparticles, which are injected directly into the tumor and are then flashed with near-infrared laser light. The laser light heats the nanoparticles, thus damaging or killing the cancer cells (red arrows). [Kamilla Nørregaard and Jesper Tranekjær Jørgensen/Panum Institute]
The investigators found that they received the best results with nanoparticles that were 150 nm in size and consisted of a core of glass coated with gold. Furthermore, the nanoparticles were illuminated with near-infrared laser light, which is the best at penetrating through the tissue. In contrast to conventional radiation therapy, the near-infrared laser light causes no burn damage to the tissue that it passes through.
Subsequently, an hour after the treatment, the research team could already directly see with positron emission tomography (PET) scans that the cancer cells had been killed, and the effect continued for at least 2 days after the treatment.
“Now we have proven that the method works,” remarked Dr. Oddershede. “In the longer term, we would like the method to work by injecting the nanoparticles into the bloodstream, where they end up in the tumors that may have metastasized. With the PET scans, we can see where the tumors are and irradiate them with lasers, while also effectively assessing how well the treatment has worked shortly after the irradiation. In addition, we will coat the particles with chemotherapy, which is released by the heat and which will also help kill the cancer cells.”