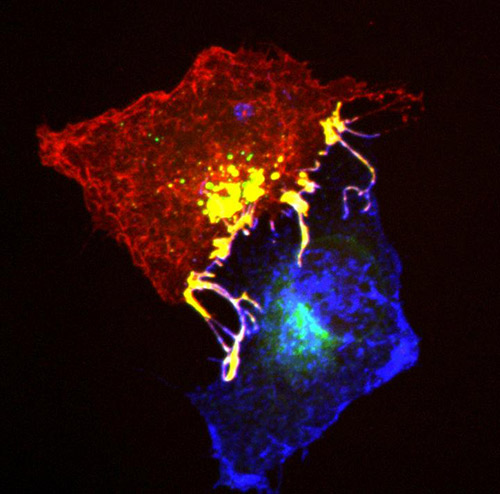
![Ephrins (blue) and Ephs (red) form complexes (yellow) at cell contact points. To enable the cells to separate from each other, they are pulled into one of the cells with the help of the signalling proteins Tiam and Rac. [MPI of Neurobiology/Gaitanos]](https://genengnews.com/wp-content/uploads/2018/08/Sep7_2016_Gaitanos_CellContactPoints1752432171-1.jpg)
Ephrins (blue) and Ephs (red) form complexes (yellow) at cell contact points. To enable the cells to separate from each other, they are pulled into one of the cells with the help of the signalling proteins Tiam and Rac. [MPI of Neurobiology/Gaitanos]
When cells get together, they have a curious way of breaking bread. They take bites out of each other, by means of a process called bidirectional trans-endocytosis. Then they go their separate ways, moving as needed to form or repair the body’s tissues. Although cells have long been known to tear themselves away from each other, the etiquette of the process has been unclear. What codes of behavior do cells observe when they regulate contact-mediated repulsion?
These codes, researchers based at the Max Planck Institute of Neurobiology have discovered, involve signaling proteins, the deployment of which is no less proscribed than the order of silverware in a formal place setting. To start, contact-mediated repulsion requires the formation of complexes between ephrin receptors and membrane-bound ephrin ligands. Next, ephrin/Eph complexes activate Tiam signaling proteins. Then, Rac enzymes become active, causing the engulfment of the ephrin/Eph complexes by the cell membrane through the local restructuring of the actin cytoskeleton.
If one of these components is missing, the engulfing process through endocytosis is blocked and the cells do not repel each other but remain attached.
These findings appeared September 5 in the Journal of Cell Biology, in an article entitled, “Tiam–Rac signaling mediates trans-endocytosis of ephrin receptor EphB2 and is important for cell repulsion.” The article describes how the Max Planck researchers found evidence for a conserved pathway for EphB trans-endocytosis, the process that removes the physical tether between cells and thereby enables cell repulsion.
When cells come into contact with each other during development, repair, or metastasis, information is exchanged by proteins, which are embedded in the cell membranes and form tight lock-and-key complexes with each other. These connections must be severed if the cells want to transmit a repulsion signal. It appears that the fastest way to do this is for the cells to engulf the protein complex from the membrane of the neighboring cell.
Development is an extremely rapid process. Increasing numbers of cells are formed that must find their correct position in the body, clearly demarcate themselves from each other to form tissue, or—as is the case in the nervous system—establish contact with partner cells in remote locations. “The crowding is accompanied by orderly pushing and shoving,” said Rüdiger Klein, Ph.D., a neurobiologist at Max Planck and the senior author of the JCB article. “A popular way for one cell to show another which direction to take is for it to repel the other cell following brief contact.”
A problem arises, however, when a pair of cells needs to separate as quickly as possible. The cells are attached to each other through the tight ephrin/Eph complex. Rather than undo the complex, the cells do something else: they extend their own cell membranes so far over the individual complexes that the complex and the surrounding membrane detaches from the neighboring cell and is fully incorporated into the cell.
“We identified an actin-regulating pathway allowing ephrinB+ cells to trans-endocytose EphB receptors from opposing cells. Live imaging revealed Rac-dependent F-actin enrichment at sites of EphB2 internalization, but not during vesicle trafficking,” wrote the JBC artice’s authors. “Systematic depletion of Rho family GTPases and their regulatory proteins identified the Rac subfamily and the Rac-specific guanine nucleotide exchange factor Tiam2 as key components of EphB2 trans-endocytosis, a pathway previously implicated in Eph forward signaling, in which ephrins act as in trans ligands of Eph receptors.”
The authors added that the Tiam-Rac signal pathway, in contrast to the Eph signal pathway, is not required for uptake of soluble ligands in ephrinB+ cells.
The clarification of the signaling pathway is important, as it provides a better understanding of the development of neuronal networks and other organ systems. The findings are also of considerable interest for cancer research: thanks to their ability to control cell repulsion, ephrin and Eph receptors play a major role in the penetration of tissue by cancer cells and in the formation of metastases. For this reason, receptors and their connection partners are the focus of current medical research. Better understanding of this signaling pathway, through which cell repulsion is controlled, could enable the development of new drugs to combat cancer.