December 1, 2012 (Vol. 32, No. 21)
New Modality Offers Super-Resolution, 3D Multicolor Imaging
Fluorescence light microscopy techniques offer several advantages when imaging biological samples, including high image contrast, good labeling specificity, multicolor, and three dimensional (3D) imaging, as well as the capability to image living specimens. The primary disadvantage, however, has been the diffraction-limited 200 nm and 500 nm lateral and axial resolutions, respectively, obtainable due to the wave nature of light.
Image resolution is defined as the smallest separation distance between two objects whereby they can be distinguished as being separate. Electron microscopy (EM) has often been used to obtain resolutions beyond that obtained by light microscopy, down to the nanoscale; however, EM of living samples is not typically feasible, and 3D imaging is cumbersome. The advent of super-resolution light microscopy techniques in the last decade has the capability to bridge the gap between traditional light microscopy and EM.
Super-resolution imaging can be broadly divided into three categories: point spread function engineering (used in stimulated emission depletion [STED] microscopy), structured illumination (SIM), and single molecule localization (SML). The principle behind SML microscopy is illustrated in Figure 1. The resolution limit in conventional light microscopy arises due to diffraction, thereby causing the point spread functions of nearby individual molecules to overlap, resulting in a “blurred” image.
SML techniques utilize the principle of “localization” to obtain higher resolution. It has been shown that one can infer the nanoscale position of a single, isolated molecule by computing the centroid of its point spread function. Importantly, in order to obtain the higher resolution offered by localization, it is essential to have only a small subset of molecules visible at a time within the field of view.
To accomplish this, SML microscopy relies on the ability to convert nearly all molecules in the sample to a dark state with only a few molecules visible (active) at a time. The positions of the visible molecules are subsequently determined by localization of the center of the molecules. The activation and localization cycles are then repeated several thousands of times to obtain a higher resolution image of nearly every molecule in the sample.
A number of SML super-resolution microscopy modalities have been demonstrated that utilize various strategies to image single, isolated molecules in a dense sample. Among others, these include fluorescence photoactivation localization microscopy (FPALM), photo-activated localization microscopy (PALM), stochastic optical reconstruction microscopy (STORM), direct STORM (dSTORM), and PALM with independent running acquisition (PALMIRA).
Each of these techniques is based on the principles outlined previously, but varies based on the fluorophores used and the protocol for obtaining sparse subsets of isolated molecules. While SML may not be amenable to every available fluorophore, a variety of genetically encoded and exogenous dyes can be used; these include photoswitchable fluorescent proteins such as Dendra2 and mEos, and organic molecules such as Alexa Fluor and ATTO dyes to list a few.
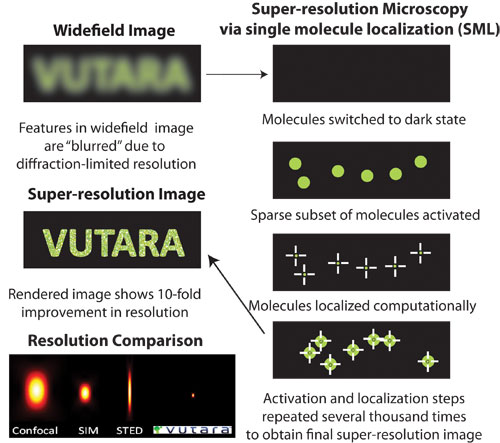
Figure 1. Principle of single molecule localization (SML) microscopy: Repeated localization of sparse subset of molecules over many acquisitions leads to enhanced resolution over the diffraction-limited widefield image. The bottom left panel compares the 3D resolution obtained with confocal, SIM, STED, and the Vutara SML microscope.
Imaging in 3D
Vutara’s flagship product, the Vutara SR-200 series SML 3D super-resolution microscope, is capable of achieving 20 nm lateral and 50 nm axial resolutions. The 3D imaging is made possible by a patented biplane technology that is based on a modified detection path, which allows simultaneous detection from two axially separated image focal planes.
The focal planes are separated by ~500 nm, which enables 3D localization throughout a ~1 µm depth of field, all in a single acquisition.
Multiple z-planes can be imaged by moving the objective focal position using a piezo actuator, thereby permitting the acquisition of super-resolution data many tens of microns away from the sample coverslip. This offers an important distinction from evanescent-wave based imaging (such as TIRF- total internal reflection fluorescence), as it permits imaging away from the plasma membrane to allow investigations of cytoplasmic and even nuclear targets at the nanoscale.
The bottom left panel in Figure 1 compares the effective 3D resolution of the Vutara SR-200 against other imaging modalities. Figure 2 shows an example of a two-color 3D image of microtubules and mitochondria in a BSC1 cell obtained using the Vutara microscope. The Vutara SRX software enables real time localization and visualization during image acquisition, as well as the ability to modify illumination and activation laser intensities “on the fly”.
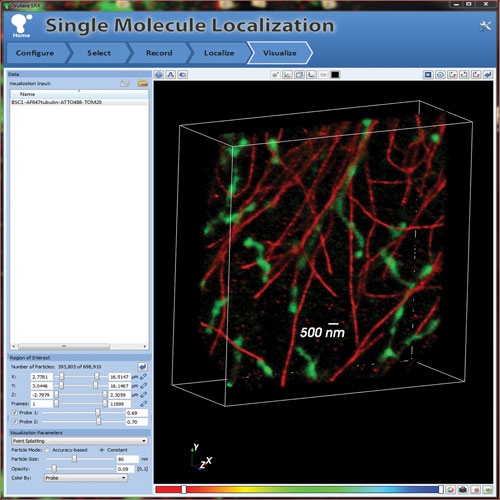
Figure 2. Acquisition and visualization of multicolor super-resolution image in 3D with Vutara microscope: Microtubules (red-labeled with Alexa 647 α-tubulin) and mitochondria (green-labeled with ATTO 488 TOM20) imaged, localized, and visualized in 3D using the Vutara microscope and Vutara SRX software.
Multicolor Imaging
Figure 3 illustrates the comparison between corresponding conventional widefield and super-resolution images. It is clear that features in the cell that were previously blurred due to diffraction are now visible with the enhanced resolution. The three-color super-resolution image shows a fixed BSC1 cell labeled with antibodies against α-tubulin (red), clathrin (blue), and mitochondrial TOM 20 (green). Alexa 647 (α-tubulin), Alexa 568 (clathrin), and ATTO 488 (TOM 20) were imaged sequentially using a 60x/1.2 NA water objective.
Multicolor super-resolution imaging makes experiments such as membrane-protein interactions, direct protein co-localization studies, and host-virus interactions at the nanoscale possible.
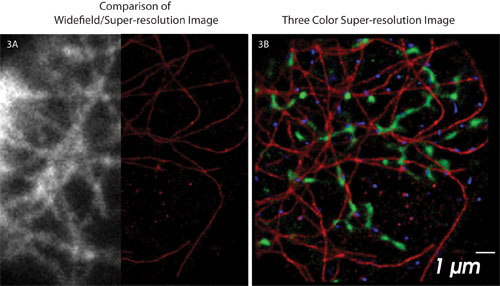
Figure 3. Conventional widefield imaging versus Vutara SML super-resolution imaging: (A) Comparison of a conventional widefield image of Alexa 647 labeled microtubules in a BSC1 cell with the super-resolution image. (B) Three-color super-resolution image of the same BSC1 cell shown in (A): Alexa Fluor 647 α-tubulin (red), Alexa Fluor 568 clathrin (blue), and ATTO 488 TOM 20 mitochondrial protein (green). The scale bar applies to both panels.
Conclusion
Historically, there has been a fundamental mismatch between the optical diffraction limit and the scale at which biochemical processes operate within the cell. Clearly, there is a need for higher resolution when addressing key biological problems. Super-resolution microscopy offers a path toward narrowing that gap by enabling live specimen imaging at the nanoscale in multicolor and 3D (up to 100 µm deep from the coverslip) in a variety of biological systems.
Yeast, Drosophila, C. elegans, tissue sections, and cell culture systems have been successfully imaged on the Vutara microscope. The advent of super-resolution microscopy has revolutionized the biological field by enabling higher-resolution clarity for imaging subcellular features. Virology, molecular biology, neuroscience, and biophysics are a few of the scientific areas that have already benefitted from SML microscopy. New discoveries await at the nanoscale.
Manasa V. Gudheti, Ph.D. ([email protected]), is senior scientist at Vutara.



