September 15, 2013 (Vol. 33, No. 16)
Andrew Barry Senior Manager PerkinElmer
Seth Cohen, Ph.D. Senior Director PerkinElmer
Alternative to Gel Electrophoresis Overcomes Limitations of Traditional Methods
As the saying goes, “garbage in, garbage out.” When it comes to DNA sample prep, the utility of DNA sequence data is directly dependent on the quality of the input material. As the cost per base pair of DNA sequence data continues to plummet, the breadth of applications for which sequencing is used continues to expand.
With this expansion comes an expanding list of input sample types (for example, formalin-fixed paraffin-embedded specimens), the need to separate target nucleic acids from complex mixtures, and attempts to generate high-quality data from diminishing amounts of material. In many cases, this is confounded by the fact that the laboratory performing the sample preparation and sequencing is not responsible for the extraction of the desired nucleic acid from the source material.
As samples navigate their way to becoming “sequencer-ready,” there is an increased investment of costly reagents, time, and resources with every process step. Thus, the ability to assess the quality of these samples prior to making this investment has become especially critical.
The factors that contribute to a nucleic acid’s viability for downstream applications include amount, purity, and degree of degradation. Nucleic acids often undergo a series of damaging processes as they are harvested from cells or tissue on their way to becoming purified. Improper storage, harsh extraction processes, and fixing processes for cells and tissues are all capable of inducing mechanical and chemical damage to DNA molecules. In the worst case, this degradation can cause irreversible damage and consequently reduce the fidelity of experimental results.

Figure 1. Schematic drawing of the microfluidic chip utilized for the genomic DNA assay showing the location of reagent wells and the path of the nucleic acid to the detection point.
While quantitative measurements for nucleic acid quantity and purity are widely adopted and scalable to high sample numbers, assays used to measure nucleic acid size and degradation still involve the manual and highly technical process of separating DNA using agarose gel electrophoresis, and visualizing through the use of ethidium bromide or other intercalating dyes.
An alternative to traditional gel electrophoresis is a genomic DNA assay developed for the LabChip GX microfluidics platform (PerkinElmer) that has been developed in a microfluidic chip format. The chip contains an interconnected set of microchannels that join the separation channel and buffer wells. One of the microchannels is connected to a short capillary that extends from the bottom of the chip at a 90º angle. The capillary sips sample from the wells of a microplate during the assay, providing automated scalability to high sample numbers (Figure 1).
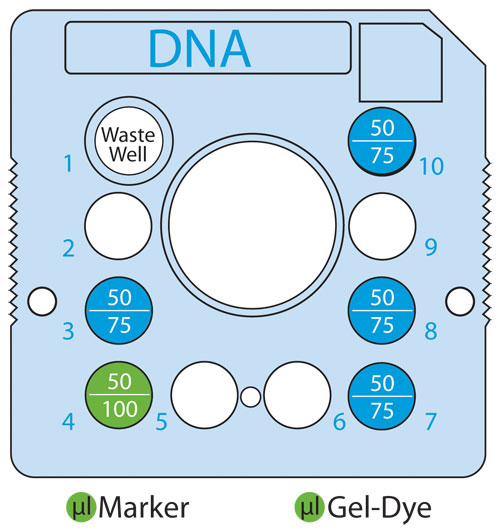
Figure 2. Top view of the microfluidic chip showing the volume (in µL) of Gel-Dye Solution or Marker Solution added for low (1–24) or high-throughput (up to 48) gDNA assays.
Assay Protocol Review
Commercial gDNA was obtained from Biochain and diluted to 50 ng/μL. Degradation was induced through exposure to fragmentase restriction enzyme cocktail (New England Biolabs) according to the manufacturer’s protocol for 10 and 20 minutes as indicated. To prepare the samples, 2 μL of gDNA was diluted with 18 μL of water in a 384-well microplate.
For particularly precious samples, the equivalent dilution may be performed with 1 μL of sample. The plate was spun briefly and placed into the LabChip GX instrument to be analyzed. The microfluidic chip was prepared by adding Gel-Dye and Marker solutions to the chip wells as shown (Figure 2) with top and bottom values corresponding to low-throughput (1–24 samples) and high-throughput (24–48 samples) modes.
Results and Summary
Results are displayed both as an electropherogram (Figure 3A) and also as a virtual gel (Figure 3B). In addition, a gDNA quality score, GQS, is derived from the size distribution of the gDNA; this score and the sample concentration are reported by the software. Figure 2 shows three samples of varying extents of degradation; samples showing greater degradation are given a lower GQS. In addition, data are provided in digital format, allowing for laboratory information management system compatibility, archiving, and distribution.
The reproducibility, compatibility with automation, and throughput of the LabChip GX gDNA assay overcomes the limitations of gel-based methods for qualitative sizing and assessment of degradation of DNA samples.

Figure 3. Electropherogram (A) and Virtual Gel view (B) of commercial gDNA degraded by addition of Fragmentase (New England Biolabs 1 µL enzyme per 1 µg gDNA) for 3 or 20 minutes. The decrease in the GQS score with increasing degradation reflects the shift in size distribution of the material to smaller fragments.
Andrew Barry ([email protected]) is senior manager, biomolecular applications, and Seth Cohen, Ph.D., is senior director, discovery applications at PerkinElmer.



