October 1, 2010 (Vol. 30, No. 17)
Application of High-Content Analysis System Can Be Beneficial in Toxicity Screening
The induction of micronuclei is a key characteristic of genotoxic compounds; for example, clastrogens produce DNA strand breakage while aneugens interfere with chromosome segregation. As a result, assays that detect micronuclei have become an important component of toxicology screening for new drug candidates.
Traditionally, micronucleus screening was performed manually by highly skilled technicians using optical microscopy and cell counting; with this time-consuming method, however, analysis was subject to operator variance, bias, and error. An improved alternative is now provided with GE Healthcare’s IN Cell Analyzer 2000 high-content analysis system. The sub-cellular resolution of the system enables micronuclei to be easily discerned, while the automated software provides faster analysis and consistent objective scoring.
Guidelines for in vitro micronucleus assays used in genotoxicity testing usually recommend scoring at least 2,000 cells per treatment condition for single samples, or at least 1,000 cells per condition if replicate assays are used. Therefore, a critical factor in establishing robust high-content cell assays is the assurance that sufficient cells are imaged per treatment condition.
This is attained on IN Cell Analyzer 2000 by automatically counting cells “online” as each image is acquired. In this optional acquisition mode, successive fields of view are acquired until a preset cell-count threshold is achieved.
Online cell counting has the additional advantage of reducing plate read times and the data-storage burden as no excess images are acquired once the required number of cells has been imaged.
Variable Cell Counts
For the purposes of this tutorial, CHO-K1 cells were seeded onto 96-well Matriplate microplates and incubated with mitomycin C (clastogen) or etoposide (aneugen) for 48 hours to induce micronuclei formation. The cells were fixed with ethanol and stained with FITC and Hoechst 33342. With the online cell-count threshold set to 1,000 cells/well, plates were imaged on IN Cell Analyzer 2000 with the 20x/0.45 NA objective.
The improved performance of the online cell-counting feature in variable cell-count assays is illustrated (Table 1) compared to results obtained using an offline analysis protocol. For mononucleated micronucleus assays configured with multiple replicates per treatment condition, a minimum cell count of 1,000 cells/well is recommended.
Without online cell counting, 34 fields of view needed to be acquired from each well to ensure a sufficient cell count was achieved (based on the maximum field count obtained when online cell counting was applied).
This resulted in a plate acquisition time of 80 minutes with the standard-chip camera, compared to an acquistion time of 37 minutes when online cell counting was applied under comparable conditions. As summarized in Table 1, the use of online cell counting significantly decreased the plate acquisition times for the assay.
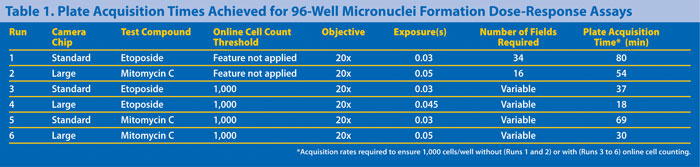
Table 1. Plate acquisition times achieved for 96-well micronuclei formation dose-response assays
Data-Acquisition Rate
To maximize the acquisition rate for micronuclei formation assays, a large-chip CCD camera is preferable to the standard-chip size. With a 2,048 x 2,048 pixel array, the large-chip camera acquires a field of view approximately four times that of the standard-chip camera (Table 1 and Figure 1).
Consequently, fewer fields of view are required to obtain the desired cell count. In addition, capture rates with the large-chip camera configuration are significantly more rapid than with the standard-chip camera in place. Both of these factors contribute to an increased speed of acquisition.
This is illustrated using dose-response assays with etoposide and mitomycin C as test compounds, with the online cell-count threshold set to 1,000. The number of fields imaged per well to achieve the threshold ranged from 1 to 34 using the standard-chip CCD cameras and 1 to 16 using the large-chip CCD camera.
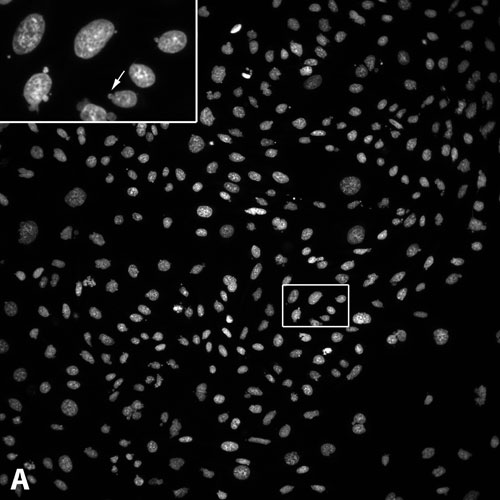
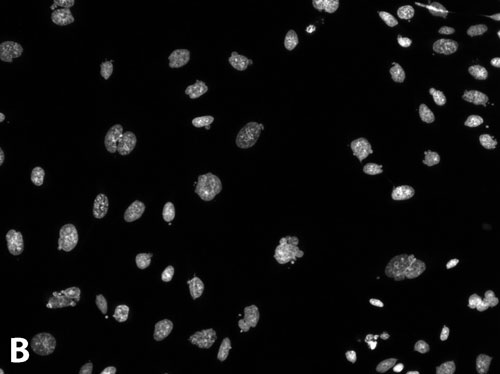
Figure 1. Micronuclei formation assay: Following treatment of CHO-K1 cells with 250 µM etoposide and staining with Hoechst 33342, cells were imaged with (A) the large-chip CCD camera or (B) the standard-chip CCD camera. Relative size of images from the two cameras is as shown. Inset: Enlargement of the region of interest outlined in white in (A). Arrow: micronucleus formed at the periphery of a nucleus. Cell count for the standard-chip camera image is 70 for field 1, compared with a cell count of 386 for field 1 for the large-chip camera.
Use of the large-chip camera reduced plate acquisition times by ~50% compared to those achieved with the standard-chip camera under comparable conditions (Table 1).
Dose-Response Assays
For dose-response assays, percentage maximum cell count and percentage of cells with micronuclei were plotted against drug concentration (Figure 2). Results show that micronuclei count increases with the drug treatment until toxic doses are reached and cells begin to detach from the plate.
Percentage maximum cell count relative to untreated controls can be used as a crude index of cell proliferation (PI), which typically decreases with increasing exposure to genotoxins.
Drug concentration at half maximal proliferation (PI50) was determined to be 127 nM for mitomycin C and 176 nM for etoposide (Table 2). These values are within the 95% confidence intervals of values obtained with the standard-chip camera, and correspond well with those reported in the literature (198 and 216 nM respectively) during validation of an automated micronucleus screening assay using GE Healthcare’s IN Cell Analyzer 1000.
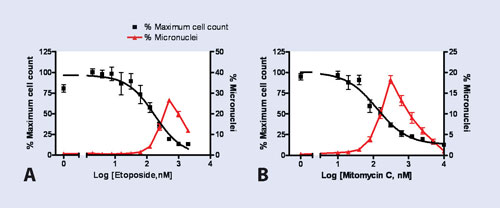
Figure 2. Dose-response micronuclei formation assays: CHO K1 cells were treated with increasing doses of (A) etoposide or (B) mitomycin C and imaged on IN Cell Analyzer 2000 using the large-chip camera. Data points represent mean ±1 SEM, n=8 wells per concentration. Percentage maximum cell count was calculated as [100 x (mean field cell count of treated sample)/(mean field cell count of untreated control sample)].
The percentage of the cells presenting with micronuclei at PI50 was also determined for each assay. Table 2 shows that values obtained with the standard- and large-chip camera were comparable to each other as well as to previously reported values.

Table 2. Dose-response micronuclei formation assays imaged with the large and standard chip cameras
Conclusions
The automatic online cell-counting facility of the IN Cell Analyzer 2000 enables the minimum number of cells required for statistical relevance to be imaged for each micronucleus assay. With this optional acquisition mode, successive fields of view are acquired until a preset count threshold is achieved.
For micronucleus assays, online cell counting ensures that the minimum cell count is reliably achieved even when test compound toxicity results in a cell-count decrease. Online cell counting has the additional advantage of reducing plate read times and the data-storage burden, since no excess images are acquired once the desired number of cells has been imaged. Furthermore, use of the large-chip camera option enables even faster results, with plate read times reduced by as much as 50%.
Hayley Tinkler ([email protected]) is scientist, cell technologies, at GE Healthcare.



