November 1, 2009 (Vol. 29, No. 19)
Case Study Demonstrates Use of Xevo TQ MS for Bioanalytical Method Development
The inherent sensitivity and specificity of LC/MS/MS has made it the technique of choice for bioanalysis. However, as compounds become more potent and are dosed at lower levels, the resultant circulatory levels also fall. This requires accurate and precise methods capable of analyzing analyte concentrations in the pg/mL range in plasma.
The development of a bioanalytical method at these low levels can be a time-consuming process, requiring the careful balance of mass spectrometry, chromatographic separation, and sample preparation. In early discovery, the development of such a method may not present too much of an issue due to the higher dosing levels and reduced assay-validation needs. As compounds progress into development, however, the need for a more sensitive, robust assay increases and method development becomes critical.
The development of a bioanalytical method is normally an iterative process. It is often necessary to adjust one part of the method to take account or address a finding from other stages. For example, if analyte response in the mass spectrometer is significantly greater at one pH or in one organic solvent than another, then it is appropriate to perform the chromatography using these conditions.
Additionally, if there is an interference that cannot be removed by solid-phase extraction (SPE) or liquid/liquid extraction, then it may be necessary to modify the chromatography conditions or employ a different MS multiple reaction monitoring (MRM) transition.
All of these individual components in a bioanalytical assay have to be evaluated and often require both MRM and full-scan MS data to identify and resolve issues. With a conventional tandem quadrupole mass spectrometer, this often requires two or more separate analytical runs as the collision cell must be emptied for full-scan MS data acquisition and be filled with collision gas for MRM analysis.
Waters’ Xevo™ TQ MS is a tandem quadrupole mass spectrometer equipped with a collision cell that is continuously filled with collision gas, allowing for the simultaneous collection of MRM and full-scan MS data. This capability can be used to simplify the bioanalysis method-development process, allowing both the analyte’s response to changes in methodology to be monitored, and also any background interferences to be detected and identified. The rapid data-acquisition rates of the Xevo TQ MS allow full-scan MS data to be acquired while still collecting a sufficient number of points across the analyte peak, in MRM mode, for accurate quantification.
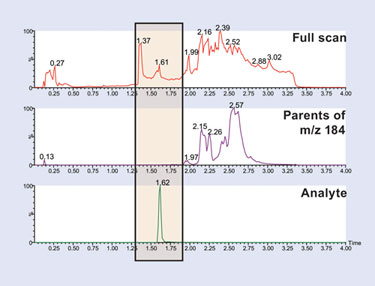
Figure 1. LC/MS/MS chromatogram of alprazolam (bottom) with lipid fraction (middle) and full-scan MS trace of endogenous matrix (top)
Sample Preparation
To demonstrate the efficacy of dual-scan MRM mode for bioanalytical method development we used alprazolam in rat plasma. Alprazolam is a triazolo analog of the 1,4 benzodiazepine class of central nervous system active compounds. A typical 2 mg daily dose results in peak plasma levels of 8 ng/mL, with a half life of 11 hours. The major metabolites formed are the p450 3A mediated 4-hydroxyalprazolam and α-hydroxyalprazolam. Due to the low circulating levels of alprazolam, an assay in the pg/mL range is necessary to accurately define the compound’s pharmacokinetics. Initial evaluation showed that the use of a basic aqueous/methanol gradient gave the maximum MS signal response in positive ion mode using the MRM transition 309→281.
The simplest form of sample preparation is protein precipitation. The resulting MRM and full-scan data obtained from this sample-preparation approach is displayed in Figure 1. In this example, the alprazolam analyte peak is monitored in MRM mode, the plasma phospholipids are monitored using precursors of m/z 184, and the remaining endogenous compounds by full-scan MS. Here we can see that the analyte peak is clearly resolved from the lipid fraction and the phospholipids, which are known to cause ion suppression.
However, investigation of the full-scan MS data can reveal an endogenous peak eluting at the same retention time as alprazolam. In this instance, the analyte had a parent mass of m/z 329. An extracted ion chromatogram of m/z 329 clearly showed that this peak coelutes with the analyte peak. This coelution could well be responsible for ion suppression, limiting the assay sensitivity.
To eliminate this interfering plasma peak, an SPE method was developed using Oasis® MCX with the LC/MS chromatogram produced using this approach displayed in Figure 2. In this data, we can observe that the method is successful in removing the coeluting matrix component, reducing the potential for matrix effects. This improved assay allows for a limit of detection of 5 pg/mL to be achieved.
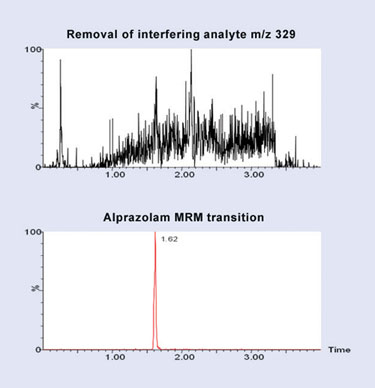
Figure 2. LC/MS/MS chromatogram of alprazolam (bottom) and extracted ion chromatogram m/z 329 (top) following SPE
Resolving Drug Metabolites
Monitoring matrix components is but one factor that needs to be considered when developing an LC/MS/MS method for bioanalytical assays; it is also critical that the assay is capable of resolving the drug metabolites from the analyte requiring quantification. The co-elution of drug metabolites with the analyte could also result in matrix effects and reduced assay performance.
Metabolites such as glucuronides and N-oxides metabolites can undergo thermal degradation in the source of the mass spectrometer and revert to the parent analyte. If the metabolite is not chromatographically resolved from the parent, this conversion will result in an overestimation of the concentration of the parent compound.
To ensure the assay specificity, it is necessary to monitor drug metabolites and ensure that they are resolved from the parent drug. Metabolite detection can be achieved by the use of full-scan MS or common fragment ion detection. The collision cell design of the Xevo TQ MS allows for the simultaneous collection of MRM, full-scan MS, and common fragment ion data. The data displayed in Figure 3 shows the simultaneous acquisition of MRM, full-scan MS, and common fragment ion data for the analysis of alprazolam in plasma.
Using this approach, the α-hydroxy metabolite of alprazolam was clearly detected by the MRM with a retention time of 1.55 minutes. This metabolite was also detected using the common fragment m/z 281. The full-scan MS data was used to confirm the identity of the metabolite. The data in Figure 3 confirms that the metabolite is well-resolved from the parent drug using a generic UPLC gradient, thus eliminating the possibility of an overestimation of drug compound.
In summary, utilizing the Xevo TQ MS’s dual scan MRM mode allowed for the detection of coeluting analytes from the plasma matrix in a single injection, reducing time and the need for additional analytical runs during the method-development process. Also employing Oasis MCX effectively isolated alprazolam from interfering components in the plasma matrix.
Endogenous metabolite information was obtained in a single injection with dual scan MRM mode, and the major metabolite of alprazolam was well resolved from the parent drug utilizing a generic UPLC® gradient. Dual scan MRM mode allowed full-scan data to be acquired simultaneously with MRM, which enabled matrix monitoring for method development as well as discovery of nontargeted compounds.
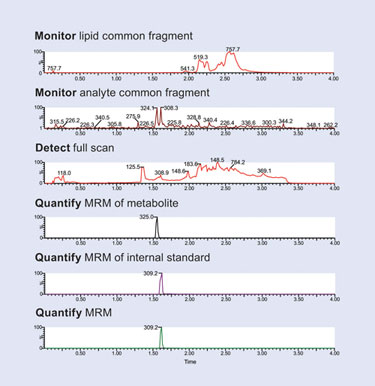
Figure 3. Simultaneous acquisition of MRM, full-scan MS, and common fragment ion data
Joanne Mather ([email protected]) is an applications chemist, Paul D. Rainville ([email protected]) is a principal applications chemist, and Robert S. Plumb, Ph.D. ([email protected]), is the senior applications manager in the pharmaceutical business operations group at Waters. Web: www.waters.com.



