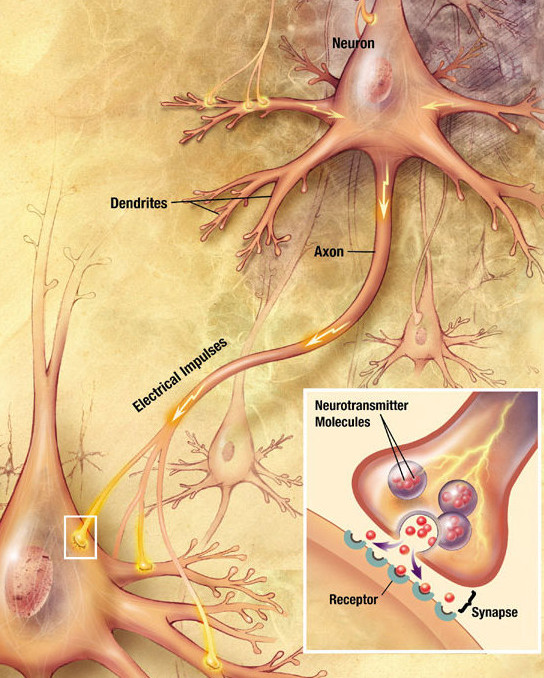
A new model of the molecular architecture at points of neuron-to-neuron contact in the brain, based on measuring the location of individual protein molecules at the sites where cell contact is made. Amazingly, proteins in the two cells align with each other to extremely high accuracy, suggesting a protein column spanning the two cells, and assuring that neurotransmitter release occurs with highest probability near the receptors that sense it.
![New research by scientists at the University of Maryland School of Medicine has for the first time elucidated details about the architecture of neuronal transmission. [WikiCommons]](https://genengnews.com/wp-content/uploads/2018/08/Chemical_synapse_schema_cropped7412818721-1.jpg)
New research by scientists at the University of Maryland School of Medicine has for the first time elucidated details about the architecture of neuronal transmission. [WikiCommons]
The study of the synaptic transmission of neurotransmitters is a field of scientific research that has seen its fair share of groundbreaking discoveries over the past century. Yet, the intricate details of the structural mechanisms that allow information to traverse the small gaps between cells rapidly, allowing neurons to communicate with each other, has eluded scientists—due in no small part to the visual limitations of light microscopy.
However now, researchers at the University of Maryland School of Medicine (UMSOM) have for first time elucidated details about the architecture of the synaptic transmission process. The findings from this new study were published recently in Nature in an article entitled “A Trans-Synaptic Nanocolumn Aligns Neurotransmitter Release to Receptors.”
Synapses are extremely complex molecular machines. But they are also incredibly tiny, spanning only a few nanometers across to allow for a massive number of them to be crammed into a finite space. For instance, it has been estimated that the human brain contains around 100 trillion synapses— each individually and precisely tuned to convey stronger or weaker signals between cells.
To visualize features on this submicroscopic scale, the researchers turned to an innovative technology known as single-molecule imaging, which can locate and track the movement of individual protein molecules within the confines of a single synapse, even in living cells.
“Using localization microscopy,” the authors wrote, they showed “that key proteins mediating vesicle priming and fusion are mutually co-enriched within nanometre-scale subregions of the presynaptic active zone. Through development of a new method to map vesicle fusion positions within single synapses in cultured rat hippocampal neurons, we find that action-potential-evoked fusion is guided by this protein gradient and occurs preferentially in confined areas with higher local density of Rab3-interacting molecule (RIM) within the active zones. These presynaptic RIM nanoclusters closely align with concentrated postsynaptic receptors and scaffolding proteins suggesting the existence of a trans-synaptic molecular 'nanocolumn'.”
This approach allowed investigators to identify an unexpected and precise pattern in the process of neurotransmission. The researchers looked at cultured rat synapses, which are very similar to human synapses in overall structure.
“We are seeing things that have never been seen before. This is a totally new area of investigation,” explained senior study author Thomas Blanpied, Ph.D., associate professor in the department of physiology at UMSOM. “For many years, we've had a list of the many types of molecules that are found at synapses, but that didn't get us very far in understanding how these molecules fit together, or how the process really works structurally. Now by using single-molecule imaging to map where many of the key proteins are, we have finally been able to reveal the core architectural structure of the synapse.”
In the new study, the authors describe an unexpected aspect to this architecture that may explain why synapses are so efficient, but also susceptible to disruption during disease. At each synapse, key proteins are organized very precisely across the gap between cells.
“The neurons do a better job than we ever imagined of positioning the release of neurotransmitter molecules near their receptors,” Dr. Blanpied noted. “The proteins in the two different neurons are aligned with incredible precision, almost forming a column stretching between the two cells.”
Understanding this architecture will not only help clarify how communication within the brain works, but also will allow researchers to elucidate how many of them fail to function properly in cases of psychiatric or neurological disease. Dr. Blanpied and his colleagues are focusing on the activity of “adhesion molecules,” which stretch from one cell to the other and may be important pieces of the “nanocolumn.” They suspect that if adhesion molecules are not placed correctly at the synapse, synapse architecture will be disrupted and neurotransmitters won't be able to do their jobs. The researchers hypothesize that in at least some disorders, even though the brain has the right amount of neurotransmitter, the synapses don't transmit these molecules efficiently.