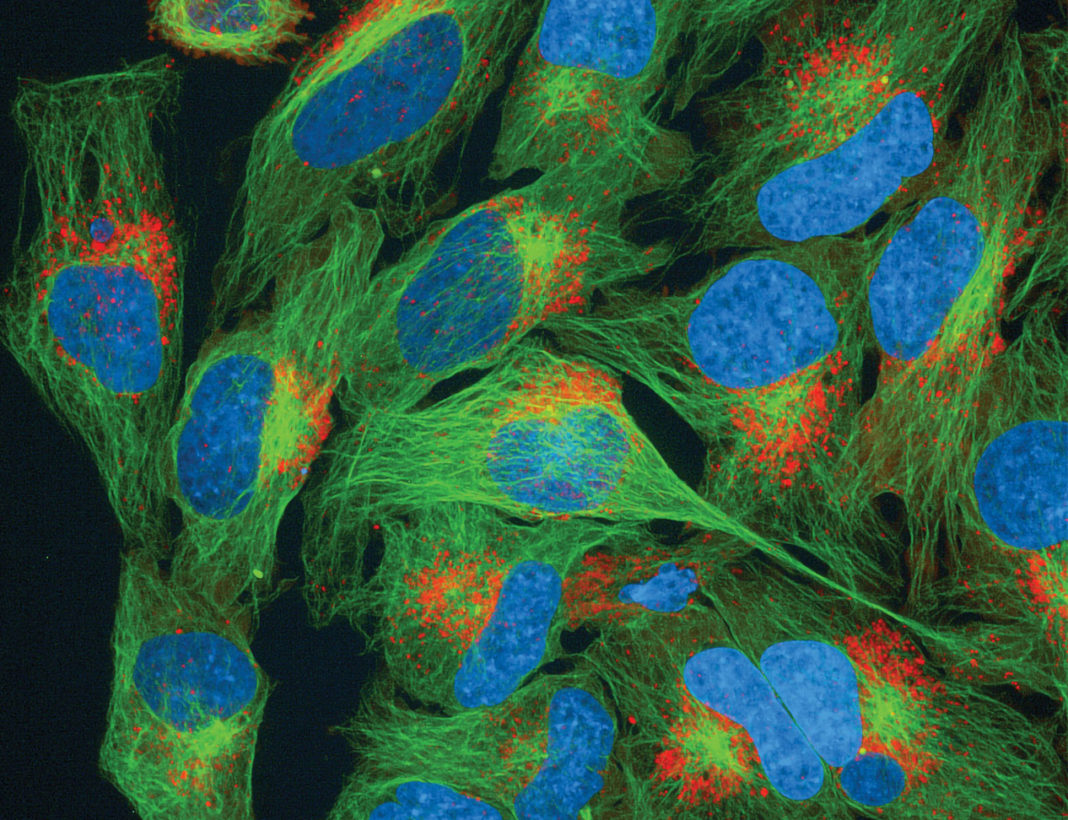
Current studies involving cellular models of disease confront two challenges: recapitulate what happens in the human body, and access longitudinal information. These challenges, which often limit the accuracy and value of cellular models, are being addressed by ExpressCells, a company that has developed a new gene modification technology to expedite the production of custom, knock-in cell lines.
“When disease models use conventional gene reporter systems, researchers can see whether a protein is promoted, activated, or inhibited, but the model may not accurately replicate what actually happens in the human body,” explains ExpressCells CEO Matthew Handel. “Fixing and staining cells to study endogenous protein targets is another option, but then information isn’t longitudinal. Fixed cells show only snapshots in time.”
Performing gene knock-ins with a CRISPR-based gene editing system improves the options to study endogenous proteins, but conventional CRISPR technology has problems, too. “It takes a long time to create the donor vectors,” Handel says. “The process isn’t efficient.” Another problem is that unedited cells must be culled. “It could take months to get a pure cell line,” he notes.
ExpressCells, a young, largely undiscovered company, has a technology to address the general challenges facing cellular models, as well as the specific problems currently limiting the effectiveness of CRISPR systems. The company’s technology dramatically speeds vector construction and reduces the time required to isolate successfully modified clones following transfection. For example, it can insert up to three reporter genes, tagging endogenous proteins of interest with fluorescent proteins, in the time it takes older technology to insert just one gene.
How it works
ExpressCells’ technology is a plasma vector system called FAST-HDR. “FAST” refers to the system’s emphasis on speeding vector construction, and “HDR” refers to homology-directed repair, the DNA repair mechanism that cells can use during cell division if a donor template of homologous DNA is present. “We use the cell’s own repair mechanism to insert the genes,” Handel emphasizes.
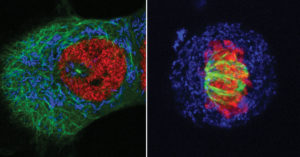
FAST-HDR uses modular plasmid backbones in combination with CRISPR-Cas9 to quickly promote HDR and the insertion of the desired labeling tag. The process, from plasmid backbone assembly to the isolation of modified cells, takes as few as two weeks.
The FAST-HDR plasmid vector system causes minimal disruption to the cell’s natural biology, Handel says, which makes it more predictive. It also expedites the insertion of multiple genes in the same cell line, advancing timelines for researchers eager to perform assays with live cells and conduct more complex experiments. For example, a FAST-HDR-enabled multigene model could help researchers quantify one protein while simultaneously determining changes in localization over time. (These changes could be made evident if the model were to use organelle-specific proteins as reference points.)
The time savings for cell line developers is substantial. ExpressCells notes that conventional genetic modification technologies require four to eight months to deliver a cell line with a single knock-in. In contrast, this new technology delivers a cell line with a single knock-in in as little as one to two months —a quarter of the time it takes traditional technology to deliver a line with only one knock-in. The company can knock-in three genes in about the same amount of time it takes to knock-in a single gene using older technology. “That wasn’t on people’s radar,” Handel declares.
Initial focus: common needs
ExpressCells focuses on two categories: tagged proteins for visualization of common targets and common cell lines (like HEK293T), and complex models for studying cellular processes, such as autophagy, that is, the process that cells use to remove their damaged components.
Recently, the company announced catalog cell lines with up to three pretagged proteins of interest, as well as what it calls “backbone” cell lines that feature one or two pretagged proteins and a third user-specified protein. It also develops custom cell lines with up to three inserted proteins of interest.
Within the next five years, Handel predicts, ExpressCells will have a robust catalog of ready-to-use products and greater capacity to undertake custom projects. The company intends to create multiple cellular options for drug discovery.
“We’re really excited about proposals for custom projects,” Handel says. He expects that ExpressCells’ work with additional targets, genes, and services will help the company broaden its capabilities.
Customer-centric innovation
Some may consider the FAST-HDR plasmid vector system a disruptive technology, but Handel prefers “customer centric.” “The word ‘disruption’ is overused,” Handel says. “It implies that traditional technology is dead. We’re not saying that. We’re not telling researchers existing models are wrong.
“Instead, we’re offering a way to make existing models better. We believe our technology is the next logical step in the work researchers perform. We’re trying to solve needs researchers already have, to enhance what they are doing.”
This evolutionary approach reflects the experience of ExpressCells’ scientific founder, Oscar Perez-Leal, MD, an assistant professor of pharmaceutical sciences at Temple University. He developed the plasmid vector system to address his own lab challenges. Before long, the system attracted interest from his colleagues and other researchers. At Temple University and the Howard Hughes Medical Institute, beta users showed that the plasmid vector system could generate successful cell lines.
“We started in mid-2018 as two people with a plan,” Handel recalls. He and Perez-Leal had created a therapeutic company previously, so teaming up again to create ExpressCells was natural.
“It took us one year to raise funding,” he continues. “We had to find investors who believed in us and our technology, and who were ready to move forward.” The young company raised $1.2 million after a Series A funding round—$800,000 after the first close (July 1), and $400,000 after the second (September 2019). The total includes a 20% oversubscription.
At this point, ExpressCells is still a small, growing company. It recently added a senior scientist to the staff, as well as multiple board members. Product promotions are just getting underway.
Now ExpressCells’ biggest challenge is building awareness among the thousands of researchers in academia and in industry labs, from tiny startups to big multinationals. “We have a great technology,” Handel insists, “and we are now focused on ensuring people are aware of it.”
ExpressCells
Location: 3675 Market Street Suite 200, Philadelphia, PA 19104
Phone: (484) 483-6759
Website: www.xpresscells.com
Principal: Matthew Handel, CEO
Number of Employees: 6
Focus: ExpressCells has developed a new gene modification technology that expedites the knock-in of multiple genes of interest in a cell line. Each knock-in is done in is little as one-quarter of the time required by traditional technologies, speeding drug development for basic researchers.