Patricia F. Fitzpatrick Dimond Ph.D. Technical Editor of Clinical OMICs President of BioInsight Communications
Key DNA Elements for Replication but Tricky Drug Targets
Recent progress in telomere research through new tools and assays may allow investigators to delve more deeply into the complicated mechanisms that control these key chromosomal structures.
As described by Xin et al., in Genome Biology in 2008, telomeres are nucleoprotein structures at the ends of eukaryotic chromosomes. These structures help maintain eukaryotic genome integrity by preventing chromosomal rearrangements or chromosomes fusing to each other, and by enabling complete replication of the ends of the linear DNA molecules.
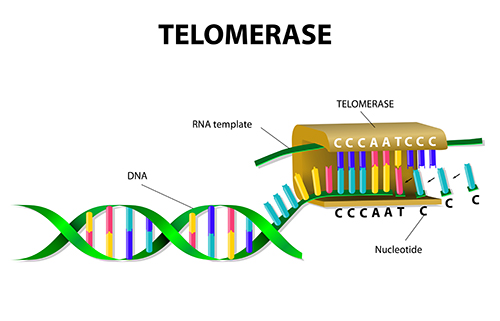
Telomeric DNA is composed of a series of sequence repeats and terminates in a 3′ single-stranded (ss) DNA overhang. At each round of DNA replication the telomeric DNA becomes shorter, but can be regenerated by the enzyme telomerase, an RNA-containing DNA polymerase. Telomeres and telomerase play key roles in maintaining chromosome integrity in the aging process, and in cancer.
Telomerase balances the telomere shortening that occurs during cell division by adding telomere repeats onto chromosome ends. While the enzyme exists at very low or undetectable levels in normal somatic cells, it is continuously expressed in 80-95 % of tumors, acting to continuously elongate telomeres and conferring a sort of immortality on the rapidly dividing cells.
In normal cells, a series of feedback mechanisms maintains the balance between shortening and lengthening established dynamic telomere length equilibrium in humans.
In recognition of this structure’s critical functions, Elizabeth Blackburn, Ph.D., University of California, San Francisco, and colleagues Jack Szostak Ph.D., of Howard Hughes Medical Institute, and Carol Greider, Ph.D., of Johns Hopkins University, received the 2009 Nobel Prize in Physiology and Medicine.
The award cited the scientists’ work on telomeres and for identifying the telomerase enzyme, in addition to the “discovery of a fundamental mechanism in the cell—a discovery that has “stimulated the development of new therapeutic strategies”, according to the Nobel Prize committee.
Now, recent research by Dr. Greider and her colleagues at Johns Hopkins University, reported in the November 24, 2015, issue of journal Cell Reports, details the development of the ADDIT assay to measure new telomere addition at a single telomere in vivo. This discovery has greatly facilitated the detection and study of telomeric length, which prior to the development of this novel assay took months of work.
Because of their roles in human aging and their association with malignancy, telomeres and telomerase have become the focus of drug discovery research to potentially intervene in the aging process, and to treat diseases caused by dysregulated telomeric shortening and cancer.
In commenting on the field in general in an interview in Nature, Dr. Greider’s co-Nobel laureate Dr. Blackburn said “We have a parts list, so we know what (the telomere) does in a static sense. But in live cells, telomeres are extraordinarily dynamic… complex little ecosystems that constantly have proteins arriving and leaving every second. I think we could learn a huge amount by studying telomeres in action, rather like researchers do by watching active ribosomes assemble proteins, in addition to knowing their structure.”
How Telomeres Work
Telomeres consist of DNA characterized by noncoding repetitive sequences and multiple protein components. Repeat sequences vary from one species to the other, the six-nucleotide repeat TTAGGG being characteristic of humans and other vertebrates. At the 3′ end of telomeres, a single DNA strand of about 300 nucleotides loops back with some of the double-stranded DNA forming the “T loop” that plays a protective role by sequestering the overhang terminal inside the double strand. A six-protein complex, the shelterin complex, functions to maintain telomere length, promote T loop formation, recruit telomerase to telomeric ends, and protect chromosome ends from being detected as DNA damage.
In humans, syndromes of telomere shortening cause age-related degenerative diseases including dyskeratosis congenita, pulmonary fibrosis, aplastic anemia, and others. In cells lacking telomerase, telomeres shorten with each cell division due to incomplete replication of the lagging strand, oxidative damage, and telomere processing events. Telomeres also shorten normally with aging
Telomerase activity is not detected in most somatic cells but is nearly universally active in human cancer cell lines and in most primary tumors. Telomerase activity counteracts progressive telomere shortening during cellular replication by synthesizing new telomeric DNA repeats at the chromosomal termini. The human telomerase holoenzyme core components include a catalytic reverse transcriptase, hTERT (human telomerase reverse transcriptase), and an associated template RNA, hTERC (human Telomerase RNA). hTERC is ubiquitously expressed in all human cells, and telomerase activity is limited by the expression of hTERT, which is found only in cells with detectable telomerase activity.
Telomeres shorten with each round of cell division, a mechanism that limits proliferation of human cells to a finite number of cell divisions by inducing replicative senescence, differentiation, or apoptosis. Telomere shortening can act as a tumor suppressor, but as Jiang, Z Zhu, and KL Rudolph noted in their article in the October, 2007 issue of Zeitschrift für Gerontologie und Geriatrie, the “downside” of this idea is that accumulating evidence indicates that telomere shortening also limits stem cell function, regeneration, and organ maintenance during aging. And telomere shortening during aging and disease is associated with increasing cancer risk.
David R. Corey, Ph.D., of University of Texas Southwestern Medical Center, writing in Chemical Biology in 2009, said that several obstacles have particularly impeded development of telomerase inhibitors, including lack of purified human telomerase in sufficient quantities to allow inhibitor screening. Until recently, he said, no high-resolution structural information about the enzyme was available, complicating structure-based design strategies. And since telomerase is a polymerase, inhibitors would need to be selective for its inhibition relative to other cellular polymerases.
Because of its pivotal role in aging and cancer, the enzyme telomerase has nonetheless become the focus of drug development efforts for anticancer therapeutics, the goal being inhibition of telomerase, leading to a decrease of telomere length, resulting in cell senescence and apoptosis in telomerase-positive tumors.
Approaches to telomerase inhibition have included small molecule inhibitors, antisense oligonucleotides, immunotherapies, and gene therapies that target the hTERT or the ribonucleoprotein subunit, as well an autologous hTERT-pulsed dendritic cell-based vaccine under development by BioTime subsidiary Asterias.
Asterias’ AST-VAC1 and AST-VAC2 are dendritic cell-based vaccines designed to immunize cancer patients against telomerase. AST-VAC2 differs from AST-VAC1 in that the dendritic cells presenting telomerase to the immune system are produced from human embryonic stem cells instead of being derived from human blood.
Geron’s Imetelstat, a lipid-conjugated 13-mer oligonucleotide sequence complementary to the RNA template of the telomerase, directly inhibits the enzyme’s activity. Geron has partnered with Janssen Biotech to develop and commercialize Imetelstat worldwide for all indications in oncology, including hematologic myeloid malignancies, and all other human therapeutic uses. Under the collaboration agreement, Janssen is responsible for the development, manufacturing, and commercialization of Imetelstat worldwide. On September 16, 2015, Geron announced the dosing of the first patient in a Phase II trial to evaluate Imetelstat in patients with myelofibrosis.
In a recently published New England Journal of Medicine paper by Gabriela M. Baerlocher, M.D. and colleagues, it was reported that Imetelstat showed disease-modifying activity in Phase II studies of myelofibrosis and essential thrombocythemia. Among thrombocythemia patients, patients achieved a hematologic response, with the majority achieving a hematologic complete response.
In the second paper authored by Ayalew Tefferi, M.D., et al., the investigators found that Imetelstat was active in patients with myelofibrosis but also had the potential to cause clinically significant myelosuppression.
In the long run, the therapeutic effects of any drugs than target telomerase will no doubt need to be weighed against their side effects and the full implications of its ubiquitous role during normal cell DNA replication in rapidly renewing cell populations.