Sarah Beckman Ph.D. Principal Scientist BioTek Instruments
Combining the convenience and throughput of multiwell assays with the benefits of a fully functioning organism
Standard toxicity experiments carried out in typical cell culture experiments lack the complexity found in small model organisms such as zebrafish. Zebrafish combine the convenience and throughput of assays carried out in a multiwell plate with the benefits of performing assays in a fully functioning organism.
Zebrafish Development
Zebrafish are an excellent vertebrate model for investigating normal and abnormal development. They share many molecular, biochemical, cellular, and physiological characteristics with higher vertebrates. In fact, about 71% of human proteins have an ortholog in zebrafish.1 Unlike other small model organisms, zebrafish possess recognizable organ systems, such as liver, heart, kidney, and pancreas.
The necessary molecular transitions required for development take place in similar sequences but occur much more rapidly in zebrafish compared to humans.2 Zebrafish embryos develop much more rapidly than mammals, and thus using zebrafish embryos as a model organism is efficient for toxicological and other studies.
Zebrafish are close enough to humans on a molecular level to be relevant, and their development is fast enough to make it a convenient model to employ for investigation.
Unlike many other model organisms, one of the major advantages of zebrafish is that the embryos develop externally and are optically transparent. This allows for simple noninvasive microscopic observations of individual cells and systems across a broad range of developmental stages, as well as for visual inspection of organ-specific toxicological effects.
As small vertebrate animals, zebrafish are amenable to high throughput screening and require only small amounts of compounds in testing scenarios. In addition to being small, adult zebrafish are easy to maintain compared to mammalian models, such as mice and rats. Also, adult zebrafish can produce hundreds of embryos per day. Thus, embryos are readily available. Because of their rapid development, organogenesis is completed within 48 hours post-fertilization.
Furthermore, there are readily available disease gene mutant strains, as well as advanced molecular, biochemical, and genetic tools. Consequently, the zebrafish is an efficient and convenient model for many developmental and toxicological studies.2
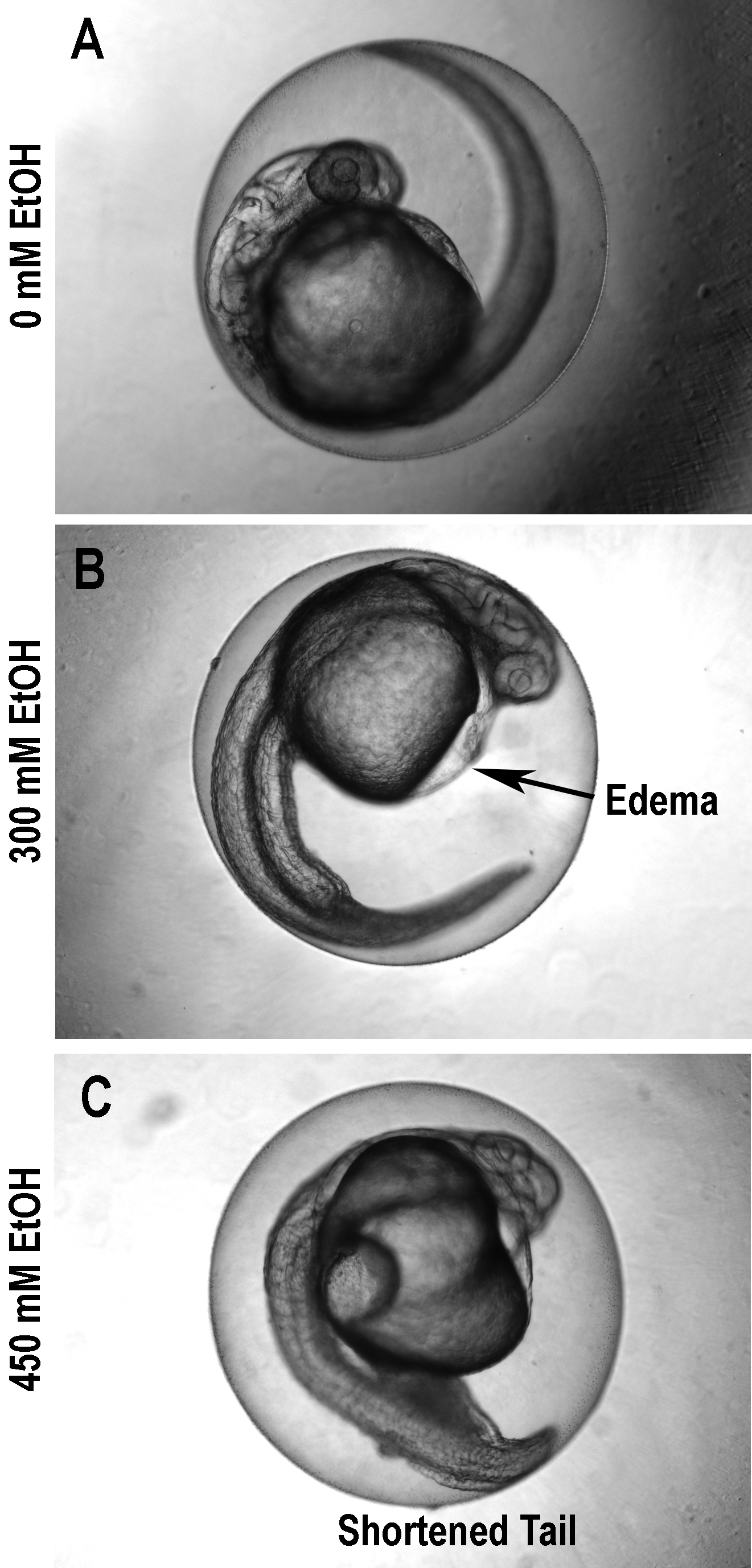
Figure 1. One-day-old zebrafish embryos exposed to ethanol (A: 0 mM; B: 300 mM; C: 450 mM) and evaluated after 24 hours. Notice how edema progresses.
Movie 1. Zebrafish embryo 4 hours post fertilization exposed to 0 mM ethanol for 24 hours.
Toxicology Screening in Whole Organisms
Although conventional in vitro assays using cultured cells are useful for evaluating some aspects of drug toxicities, in vitro results frequently deviate from in vivo results because of the increased complexity found in whole organisms with respect to processes such as drug absorption, distribution, metabolism, and excretion. To produce in vivo phenotypes in zebrafish assays, compounds must exhibit the ability to be absorbed, reach the target tissue, and avoid rapid metabolism and excretion.2 Zebrafish larvae, unlike cells in a dish, possess functional livers, kidneys, and blood-brain barriers, and thus, drugs can be tested in a complete system.
Several studies have found strikingly similar mammalian and zebrafish toxicology profiles.3 General toxicology screens can capture a broad range of phenotypes in wild-type (WT) zebrafish embryos. Pain, sedation, tumor metastasis, vascular tone, and gut motility are all examples of disease-relevant phenotypes that are observable in zebrafish yet inaccessible by modeling in cultured cells.2
Zebrafish are especially amenable to phenotype-based drug discovery (PDD) screens. These consist of identifying compounds of therapeutic value based on the phenotypic perturbations produced regardless of targets or mechanisms of action.4
Movie 2. Zebrafish embryo 4 hours post fertilization exposed to 300 mM ethanol for 24 hours.
As phenotypic screens integrate physiology and multiple signaling mechanisms during the screening process, the identified hits have higher biomedical applicability. PDD evaluates observable phenotypic changes in a cell, tissue, or organism. The changes can then be used to identify small molecules and other modulators for a disorder. A small molecule identified in an in vivo system has a higher chance of having bioactivity and clinical relevance. In fact, PDD screens performed on zebrafish have led to the identification of some lead compounds of therapeutic potential.4
Kinetic Imaging
Many toxicology studies are conducted as endpoint-only studies. In this case, after a certain amount of time after exposure to a drug, phenotypes are quantified. However, intermediate phenotypes are missed when studies are conducted in this manner.
Many of the highly dynamic processes that occur during development can only be studied in full detail in the intact, living embryo, and this is true also of the effects of toxic substances. A kinetic imaging system, such as the Lionheart FX Automated Microscope or Cytation 5 Cell Imaging Multi-Mode Reader (BioTek Instruments, Winooski, VT), enables image capture every five minutes over a 24-hour period and determines intermediate as well as endpoint effects of toxic substances on embryo developmental phenotypes.
An Example of a Kinetic Toxicity Experiment: Ethanol Toxicity
At three hours post-fertilization (hpf), embryos were added to the wells of round bottom 96-well plates. Most of the E3 media was removed, and 1% agarose was added to the wells. Embryos were repositioned, if necessary, to ensure that they remained in the center of the well before the agarose hardened.
To determine the effect of ethanol on zebrafish development, 0-, 300-, or 450-mM ethanol solutions were added to the embryos in E3 media. Embryos were exposed to ethanol from 4 hpf to 27 hpf.
A Lionheart FX or Cytation 5 imager was used to take images at 4× magnification every five minutes for 24 hours in the brightfield channel. These images were then compiled into a movie using Gen5 Microplate Reader and Imager Software, and WT development was compared to development that occurred when zebrafish embryos were exposed to the toxicant ethanol.
Ethanol is a well-established developmental toxicant and is the most commonly consumed and abused drug. Exposure to ethanol during development can lead to multiple disorders such as learning and memory deficits, cell death in the central nervous system, skeletal dysmorphogenesis, and alterations in the startle response.5
Model systems have been used to study ethanol, and consistent with what is found in humans, alcohol causes severe teratogenesis. As embryos are exposed to increased concentrations of ethanol, they present with increased levels of dysmorphogenesis.
Evidence of worsening ethanol-induced dysmorphogenesis in zebrafish embryos is presented in Figure 1, which shows how pericardial edema is absent when the ethanol concentration is 0 mM, evident when the ethanol concentration is 300 mM, and marked when the concentration is 450 mM. In the embryo treated with 450 mM ethanol, there is even evidence of cell death, especially in the tail. Also, in this embryo, axial growth is inhibited compared to that in embryos exposed to 0 mM and 300 mM ethanol.
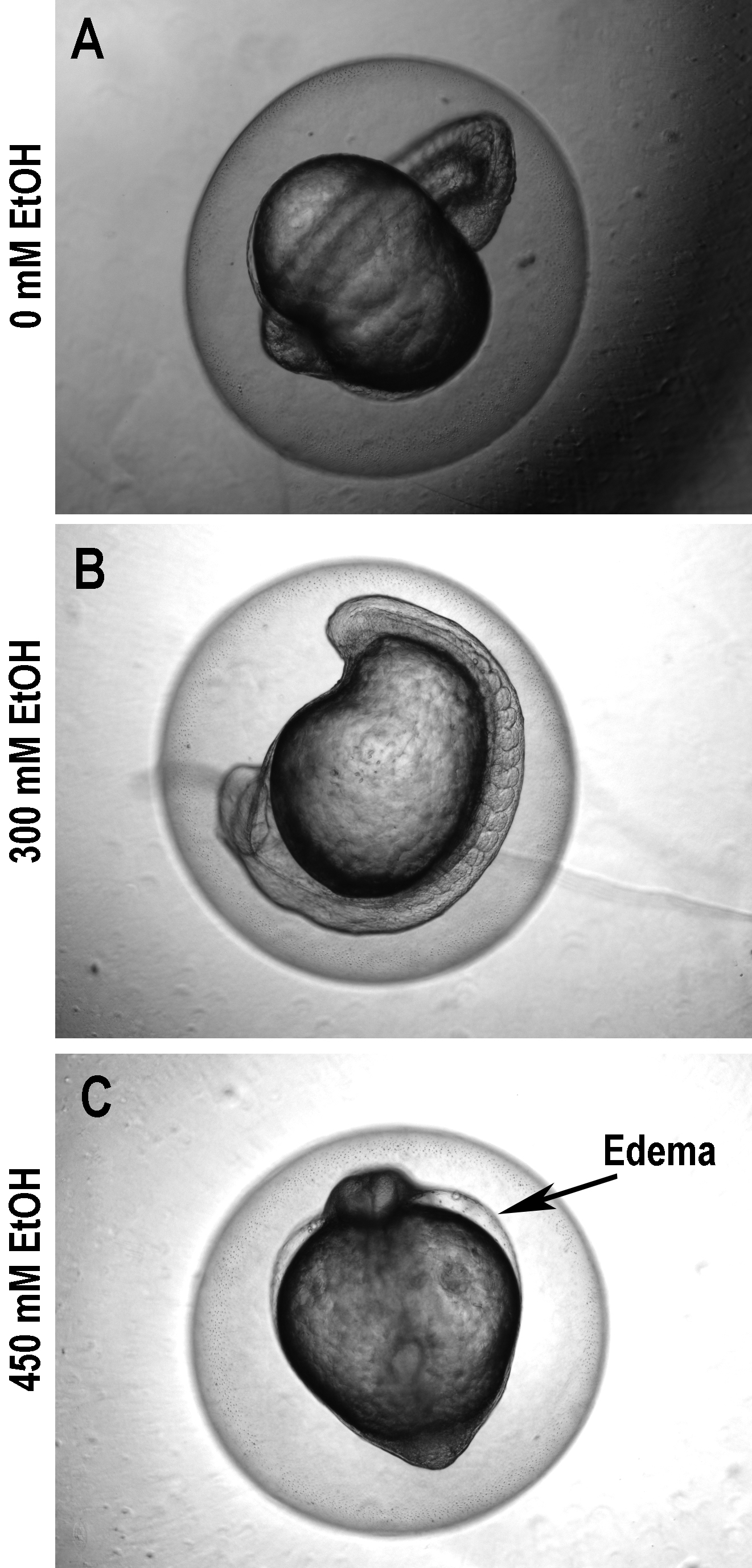
Figure 2. Zebrafish embryos exposed to ethanol (A: 0 mM; B: 300 mM; C: 450 mM) and evaluated after 12 hours. Notice how edema progresses and development is increasingly delayed.
Movie 3. Zebrafish embryo 4 hours post fertilization exposed to 450 mM ethanol for 24 hours.
Ethanol exposure also suppresses embryo activity. An untreated embryo (0 mM ethanol, Movie 1) is more active than treated embryos (300 mM ethanol, Movie 2; 450 mM ethanol, Movie 3). The treated embryos are also delayed in development, as indicated by less pigmented retinal cells.
This delay in development is further highlighted in Figure 2 where the embryo treated with 300 mM ethanol shows a delay in development as demonstrated by the decreased length of the yolk extension as compared to the wild-type embryo. The embryo treated with 450 mM ethanol has a delay in development as well as the beginnings of edema.
Conclusions
Zebrafish are an excellent model for carrying out kinetic toxicology experiments. Their small size, ease of use, and complexity as an organism make them ideal for these types of studies. Performing automated, kinetic toxicity studies on zebrafish using the Lionheart FX or Cytation 5 enables phenotypic evaluation of the effects of toxic substances.
1. Howe K, Clark MD, Torroja CF, et al. The zebrafish reference genome sequence and its relationship to the human genome. Nature 2013. 496: 498–503.
2. MacRae C.A, Peterson, RT. Zebrafish as tools for drug discovery. Nat. Rev. Drug Discov. 2015. 14: 721–731.
3. Zhang Y, Wang X., Yin X., et al. Toxicity assessment of combined fluoroquinolone and tetracycline exposure in zebrafish (Danio rerio). Environ. Toxicol. 2016. 31: 736–750.
4. Szabo M., Svensson Akusjärvi S., Saxena A., et al. Cell and small animal models for phenotypic drug discovery. Drug Des. Devel. Ther. 2017. 11: 1957–1967.
5. Carvan MJ, Loucks E, Weber, DN, Williams, FE. Ethanol effects on the developing zebrafish: neurobehavior and skeletal morphogenesis. Neurotoxicol. Teratol. 2004. 26: 757–768.
Sarah Beckman, Ph.D., is a principal scientist at BioTek Instruments. The company designs, manufactures, and distributes life science instrumentation such as cell imaging systems, microplate readers, washers, dispensers, automated incubators, stackers, and pipetting systems.



