October 15, 2008 (Vol. 28, No. 18)
Designing Well-Plate Microfluidics for Applications in Microbiology and Cell Adhesion
Life science research and drug discovery typically favor in vitro assays for cost-efficient screening and in vivo assays for data with higher physiological context. Recent innovations in live-cell imaging have begun to close this gap by offering the convenience, control, and throughput of well-plate based assays that approach the clinical relevance of an animal model.
Fluxion Biosciences developed its well-plate microfluidics technology to emulate in vivo conditions in a well-plate assay format. The company’s BioFlux System incorporates this technology to offer live-cell imaging assays with controllable shear flow.
Numerous biological processes occur in the presence of shear flow, particularly in the vascular, immune, and respiratory systems. Many of these processes behave differently under flow versus static conditions. This severely limits the overall effectiveness of conventional static well-plate assays. The BioFlux platform enables simulation of physiological shear flow rates as well as other microenvironmental factors such as temperature, oxygen, and carbon dioxide concentrations. This yields a high-content, live-cell imaging assay that can be performed in a well-plate format. Some of the key applications will be discussed in this article, including cell adhesion and microbiology.
The BioFlux System comprises a benchtop instrument and microfluidic well-plate consumable (Figure 1). Each BioFlux well plate contains up to 24 individual flow cells that are fully enclosed microfluidic channels for running flow-based experiments. The flow cells are arrayed on the bottom of an SBS-standard well plate with a conduit integrated from the well to the microfluidic path (Figure 2).
This enables the reagents to be introduced directly into the microfluidic path via the well, rather than through tubing or off-chip reservoir. Effective containment of the fluids within the plate obviates the cleaning and assembly steps required with other types of flow cells.
Some BioFlux plate configurations offer two input wells in each experimental channel. This feature provides a software-controlled exchange of compounds, media, or buffer in real time without interruption of flow. Once the reagents have been introduced into the wells, an air-tight interface is placed on the plate with a series of pneumatic connections to each of the wells.
Shear is initiated using a pneumatic controller that delivers bidirectional shear force within the physiological range, and rapidly (
Principal benefits of the system include higher throughput and experimental flexibility. Microfluidic parallelization enables up to 24 unique assays on a single plate, which can be multiplexed up to 96 simultaneous assays using multiple device interfaces.
BioFlux software supports automated shear control and integrated imaging with select CCD and CMOS cameras. This allows the user to concurrently control flow protocols and imaging in an automated sequence. For example, if images are captured during a step-wise shear ramp, the software can be set to acquire in concert with the shear switching. The software integrates analysis modules to quantitate common assays such as cell rolling velocity, cell counts and adhesion, migration, and fluorescence intensity.

Figure 1. The BioFlux 200 System for live-cell assays under controlled shear flow

Figure 2. BioFlux Plate channels as viewed from beneath the well plate.
Cellular Adhesion
Live-cell assays run on the BioFlux platform provide insight into major disease areas such as platelet adhesion, metastasis, and allergy and autoimmune disorders. Many immunology and cancer applications focus on cell adhesion. Often the process of adhesion occurs under physiological shear flow within the blood vessels.
Typical BioFlux assays in this area involve growth of endothelial cell layers, rolling velocity measurements, cell adhesion counts, transmigration, and high-content imaging, all of which can be examined under shear flow conditions (Figure 3). Small molecule and antibody screens can also be performed under flow conditions to enhance the physiological relevance over conventional well-plate assays (Figure 4).
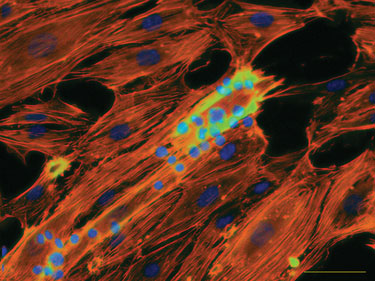
Figure 3. Cell-cell adhesion assay under flow.
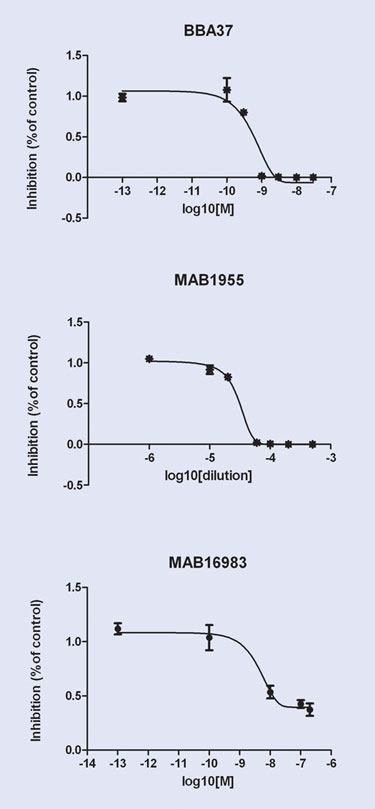
Figure 4A. Adhesion inhibition screen

Figure 4B. Micrograph of Jurkat cells stained with calcein AM in two different experiments.
Microbial Biofilms
Microbiology assays are another focus area requiring shear flow. Many microbial communities, classified as biofilms due to their sticky polysaccharide matrix, grow only in the presence of shear flow. Others can grow in their free-swimming planktonic form but take on a much more drug-resistant configuration when they attach to a surface and produce a biofilm. Biofilm studies benefit from the presence of controlled shear flow by providing a physiologically relevant flow profile equivalent to the sites of biofilm infection.
These sites of biofilm growth include the respiratory tract in cystic fibrosis patients, the surfaces of catheters and implants, burn wounds, and in hospital-derived infections. Quantitative biofilm assays often incorporate growth kinetics, morphology, viability, and cross-comparisons between mutant and wild-type microbial strains. Antimicrobial efficacy and dose-response assays also benefit from the flow approach because a wide disparity often exists between planktonic and biofilm drug efficacy, the latter being more relevant in most human infections (Figure 5).
Other advantages of the BioFlux platform include low media usage (working volumes as low as 20 µL), higher throughput over conventional flow cells, and options to control the microenvironment (i.e., anaerobic, hypoxic). Aerobic and anaerobic biofilms from a wide variety of organisms grown with the BioFlux System include: Pseudomonas, Streptococcus, Porphyromonas, Bacillus, and Acinetobacter.
Beyond immunology and microbiology, the BioFlux shear flow system has applications in stem cell research, imaging cytometry, neuronal cells, and other specialized cell cultures. Further expansion of the system will include higher density well plates, alternate well configurations for greater experimental flexibility, and integrated microscopy systems for complete research/screening solutions.
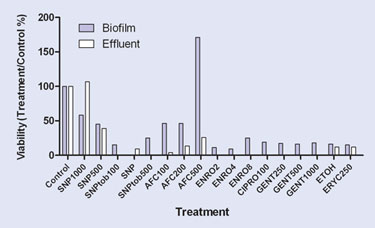
Figure 5. Compound screening for biofilms.
Carolyn G. Conant, Ph.D. ([email protected]), is application scientist, Fluxion Biosciences. Web: www.fluxionbio.com.



