October 15, 2010 (Vol. 30, No. 18)
Jim J. J. Zhang, Ph.D.
Commitment from Local Firms and Government to Refine Operations Yielding Results Rapidly
The development of biologics has gradually become a key focus of almost all drug companies worldwide, large or small, major or startup. The global value of the biologics market has been growing rapidly in the past decade and, more importantly, it is poised to continue to grow at an even faster pace in the foreseeable future.
Although the biopharmaceutical industry in China got a late start, it has posted great gains in the last decade as well. With a significant amount of capital being invested by government at various levels, the country’s industrial infrastructure is being transformed at break-neck speed.
Infrastructure improvements, an influx of returnees, readily available funding from various sources, and the entrance of a growing number of experienced multinational biopharma companies all have contributed to the fast growth of China’s biopharmaceutical industry. However, as the Chinese biopharmaceutical industry increases its international presence, the quality of its biologics and the capabilities of its CROs and CMOs are of increasing concern.
There are close to 1,000 bio-related companies in China; they are spread throughout the country and about half of them were established in the last five years. The majority are, thus, small in terms of personnel and sales revenue. About half of these start-ups are R&D-focused companies that were established by Chinese returnees or Western biopharmaceutical companies as their China division or subsidiary.
Among all Chinese bio-companies, about 46% of them are engaged in manufacturing and marketing of biologics, an indication that the industry is still focused on manufacturing. That is changing, however, as more and more major, traditional Chinese pharmaceutical companies enter the biologics field.
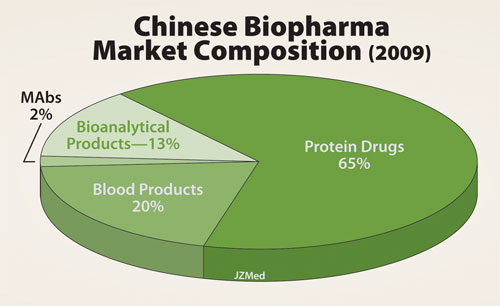
China’s current biologic drug market is about 65% therapeutic proteins. The remainder of the market is made up of blood products, mAbs, and bioanalytical products.
R&D
Most Chinese drug companies that have an eye on international markets have their own dedicated drug R&D teams, usually an internal research institute. In almost all cases, however, these teams are small with few experienced senior scientists. At present, most Chinese biopharmaceutical companies have an annual R&D budget between 5 to 8%. Not surprisingly, many Chinese drug firms collaborate with top-tier Chinese research institutions.
China’s R&D capability is advancing rapidly, and the development of new vaccines is a notable example. China was the first country to approve an H1N1 vaccine and currently, eight companies have gained approval from the SFDA for their H1N1 vaccines.
While Chinese biopharmaceutical companies possess a sophisticated level of knowledge on the development of new recombinant protein drugs, their expertise developing monoclonal antibody (mAb)-based drugs falls far behind their Western counterparts. Currently, only a small number of these firms have R&D programs focusing on mAbs. Almost all of their programs are in very early stages; in fact, over 70% of biologics being developed in China are either in preclinical or Phase I development.
Manufacturing
Among manufacturing-focused companies in China, the largest group is making human recombinant proteins including interferon, interleukin, growth hormones, growth factor receptors, and insulin. Only a small number of companies have the ability to manufacture mAbs on a large scale. Chinese CMOs boast a combined ability to manufacture more than 80 types of biologics.
At present, companies that possess upstream capabilities are largely research focused, such as research institutions. Most strictly, manufacturing-focused companies are still relatively weak in independently developing cell lines suitable for producing desired protein- or antibody-based drugs.
Unlike the widespread and accepted outsourcing of small molecule drug R&D and manufacturing, large-scale outsourcing of biologic operations is just beginning to gain acceptance. Although biologics-focused firms have multiplied in recent years, the number of Chinese service companies in the biopharmaceutical sector at present is much smaller than the number of CROs/CMOs in the traditional small molecule drug sector.
The majority of Chinese biopharmaceutical outsourcing providers are still limited in service scope, capacity, capabilities, and experience. The weakest sector is the large-scale manufacturing of mAbs based on mammalian cell culture.
On a positive note, Chinese CROs/CMOs are improving their capabilities rapidly, largely because of the influence of growing numbers of experienced multinational service companies that are doing business in the country.
Over the past five years, the Chinese biopharmaceutical market has posted an average annual growth rate of about 25%. The current value of the biologics market in China is about $8 billion, which is about 20% of China’s current total drug market value and about 6% of the total global biologics market value.
Within China’s biologics sector, about 65% of the value (or $5.1 billion; see Figure) is contributed by therapeutic proteins. The current market value of vaccines in China is about $900 million; this sector has posted gains of about 22% per year since 2005.
There are numerous factors, both internal and external, that are contributing to China’s burgeoning biologics industry. Support from government entities, growing numbers of foreign and local venture capitalists, and the presence of a large number of multinational biopharmaceutical companies have been helpful in providing a favorable environment for growth.
As the industry grows and new opportunities emerge, nascent firms are entering the arena. And as the Chinese biopharmaceutical industry increases in size and capabilities, the technical gap between China and developed countries is being reduced.
As multinational firms look to outsource more of their operations, the large number of readily available scientists and engineers in China, coupled with the rapid growth of the Chinese biopharmaceutical market, makes China an ideal place for doing business.
In the dynamic global biopharmaceutical industry, China has positioned itself well, particularly in frontier research areas such as genomics and stem cell research. The Chinese government, recognizing the value of these technologies, is providing both financial support and favorable policies to encourage their development.
Although the Chinese biopharmaceutical industry is still young, its momentum has been steady and is building. With continued infrastructure improvement, cooperation of multinational companies, and readily available funding from a variety of resources, the Chinese biopharmaceutical industry is set to develop at an even faster pace in the near future. We believe this market will grow at a CAGR of 28% in the following years, and its value could reach close to $35 billion by 2015.
Jim J. Zhang, Ph.D. ([email protected]), is president and managing director at JZMed. This article is based on the firm’s latest research report, “Chinese Biopharmaceutical Industry: Current State and Future Development”.



