May 1, 2011 (Vol. 31, No. 9)
Sue Pearson Ph.D. Freelance Writer GEN
Success of Dendreon’s Provenge Illuminates Possibilities in this Fast-Growing Field
Cell therapy for regenerative medicine is here, “but at the moment what we are offering is first-generation cell products,” said Nagy Habib, M.D., CEO of LiverCyte and head of HPB surgery at London’s Hammersmith Hospital, at the recent “International Business Innovation Zone” (IBIZ) conference. “We have the bike now, but who knows—in five years’ time we may have the Rolls Royce. We have to start here to be able to get these curative therapies to those that really need them.”
“Currently, there are well over 300,000 people worldwide already treated with cell therapy products,” added Chris Mason, Ph.D., professor of regenerative medicine bioprocessing at University College London. “However, many of these products are tissue-engineered skin that are still made in a labor-intensive cottage industry manner in the equivalent of Petri dishes.”
Speakers at the conference presented an interesting selection of promising cell therapies to treat an array of different unmet medical needs. Many of these were autologous cell therapies, yet seemed to represent surprisingly cost-effective alternatives to traditional treatment options for some conditions.
LiverCyte is using adult autologous stem cells as a bridge for cellular recovery of the liver. “Our stem cell therapy is based on omnicytes (the CD34 expressing fraction) from a patient’s own bone marrow, which we believe are producing molecules that prevent apoptosis,” Dr. Habib explained. “These omnicytes are expanded over seven days and then injected into the portal vein to ensure the cells are targeted at the liver.”
The treatment has been tested in a Phase I/II study on two patients, one 64-year-old man with the chronic liver disease primary sclerosing cholangitis, and a second patient with cirrhosis of the liver, both of whom needed a liver transplant to survive. The company plans to carry out a Phase II/III trial of the omnicyte therapy in China because there is a large patient population suffering with hepatitis B and C infections that now have liver damage. Many of these patients require liver transplants but are unable to have them.
“Our first patient is now fit, well, and back to work without needing a liver transplant. The second has improved liver function as indicated by increased levels of bilirubin production, and his condition has improved significantly.
“In the U.K., it costs £500,000 ($816,000) to carry out a liver transplant and ensure the patient does not reject it. There are only around 500 performed in the U.K. every year because of lack of suitable livers. Our omnicyte therapy costs around £10,000 ($16,300) per patient and could prevent patients having to have a liver transplant,” Dr. Habib concluded.
Altrika is developing technology originating from the U.K.’s University of Sheffield. The autologous cell therapy product known as Myskin® is a plasma polymer film coated with the patient’s own skin cells. It is on the market to treat burns or chronic wounds such as venous leg ulcers or diabetic foot ulcers.
“At Altrika we have improved our GMP production significantly, and on average one of the operators can now produce enough Myskin to treat approximately 50 patients per year,” said David Haddow, Ph.D., operations director. “This compares to some other cell therapy processes where operator productivity may be only 20 percent of this due to manufacturing complexities or the cell-expansion time frame. Our increases in production per operative could make the use of Myskin a viable addition to the treatment options for chronic leg ulcers.”
“While traditional approaches are low cost in product terms, they represent high staff costs for the healthcare providers. Also, because wounds are managed rather than effectively treated, the long-term socioeconomic costs associated with frequent repeat hospital visits can make this a more costly option than using high-value, active therapies such as Myskin to heal the wound and maintain it in a healed state.”
Cell Medica is working with major research groups in Europe and the U.S. to show that patient-specific cell therapies can be used not just in regenerative medicine but also to treat infectious disease and cancer.
“T cells provide immunity against viral infections, and they can also help control cancer. You see this in patients with weakened immune systems who often get unusual forms of cancer linked to oncogenic viruses,” commented Gregg Sando, CEO. “Our lead product treats bone marrow transplant patients with infections arising from latent viruses residing within the patient prior to transplant. We do this by using an infusion of selected T cells from bone marrow donors who have immunity to the target virus.
“We use a similar approach to treat certain types of cancers that are associated with viral infections, for example Hodgkin lymphoma, which is linked to the Epstein Barr virus (EBV). In this situation, we take the T cells from the patient rather than a donor, and we activate these T cells to kill the cancer cells based upon the recognition of viral antigens. This can have a dramatic effect, even in patients with advanced disease who are not responding to any drug or radiation therapy.”
Cytovir CMV, the company’s lead product, is a T-cell therapy for bone marrow transplant patients. It is currently in both Phase II and III trials to treat patients at risk of life-threatening cytomegalovirus (CMV) infections in different transplant settings. According to Sando, these randomized, controlled trials are the first to undertake a head-to-head comparison of T-cell therapy with conventional drugs in the infectious disease indication.
“These T-cell therapies are individually manufactured for each patient. We believe that we can use EBV T cells as an adjuvant strategy for about 40 percent of Hodgkin lymphoma patients undergoing autologous bone marrow transplant.
“We can service the entire U.K. with only three hospital support staff, since 80 percent of patients receive treatment in the largest 20 transplant centers. So we could be looking at a potential blockbuster patient-specific therapy that could be produced and delivered relatively inexpensively.”

Altrika’s Myskin® uses patient skin cells grown on a functional polymer membrane to optimize wound healing.
Allogenic Stem Cell Treatments
Michael Hunt, CEO at ReNeuron, discussed his company’s allogenic stem cell lines for treating neurological conditions. “Around 25 percent of U.K. hospital beds are occupied with patients suffering from the complications of a stroke, so the cost of health and social care are staggering. Cell intervention therapy can give hope of recovery.”
ReNeuron has a cell line known as CTX that was selected from 120 cell lines and has been preclinically tested as a therapy for a range of neurological conditions. “In over 30 preclinical studies, we have treated around 500 rats and have shown that CTX is anti-inflammatory, pro-angiogenic, and neurogenic, and it promotes endogenous cell responses that reduce brain scarring.”
The company’s lead product, ReN001, which is based on CTX, is in Phase I trials. ReNeuron aims to treat 12 patients with moderate to severe disability post-stroke by injecting ReN001 cells directly into the brain. The patients will be assessed for two years to determine progress.
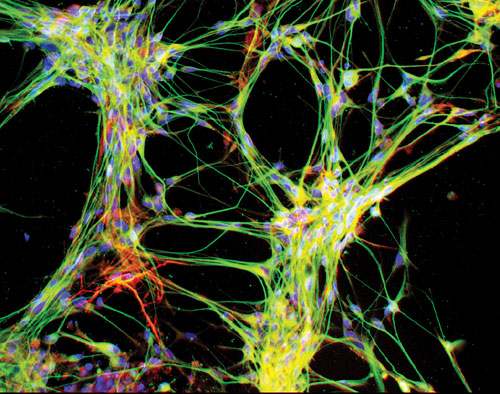
Neuronal differentiation of ReNeuron’s CTX human neural stem cell line. Immunocytochemical staining for BIII tubulin (green) and double cortin (red) expression with a Hoechst 33342 counterstain (blue).
Where Next?
As well as demonstrating the efficacy of many cell therapies, several key barriers to cell therapies becoming widely adopted in the U.K. were discussed. Funding and affordability were cited as problems. “Altrika has already been bankrupt once, but we were fortunate enough to be purchased by Ilika, so now we have the funding to continue our work in the U.K.,” Haddow stated. Dr. Mason added, “There needs to be an urgent step to change the way we calculate reimbursement. Cell therapies are aimed at cure and not conventional symptom control and disease management, therefore a combination of healthcare, employment, and social consequences need to be taken into consideration. “For example, the success of Provenge, Dendreon’s autologous cell therapy for the treatment of advanced prostate cancer, is helping investors to see that an autologous stem cell product that sells for $93,000 is a viable proposition because of its unique ability to significantly extend a patient’s life.” The complexity of the U.K.’s regulatory system was also seen as a potential barrier to success. “We have not suffered because of the science; we have safe cells that have been extensively preclinically tested,” stated Hunt. “They are straightforward to scale and bank, but it is the regulatory process that has slowed down our progress at times.
Analysis & Insight: A Clearer Regulatory Pathway for Cell Therapy May Help Improve Investment
It isn’t just the U.K. that suffers from complicated guidelines for developing these types of treatments. The U.S. sector too is fraught with uncertainty, which has naturally kept most investors away from this field. Government and disease foundations have thus come to act as important funding providers for cell therapy R&D. To get all the details on regulatory and financing activities on both sides of the pond, read our story here.
“We have interacted with 12 licensing and regulatory bodies during the seven-year preclinical development of ReN001,” he continued. “Perhaps our licensing and regulatory bodies need to work more closely together or be consolidated to some extent to ensure that the regulation of cell therapy candidates in the U.K. is as efficient as possible.” Despite these problems, speakers at IBIZ were upbeat about the future of cell therapies in the U.K. As Dr. Habib summarized, “The U.K. is in a prime position, with companies such as Cell Medica showing us how you can use hospital and university centers to produce and distribute more affordable autologous cell therapies. There is also a great resilience seen with companies like Altrika and ReNeuron.



