October 15, 2012 (Vol. 32, No. 18)
Biomarkers—the essential fuel for personalized medicine—play an ever-growing role in drug discovery, clinical development, and, at least for cancer, in the clinic where genomic profiling of individual tumors often informs therapy selection.
Advancing assay technology, improving techniques for working with FFPE tissue samples, and multistakeholder collaborations are all driving factors.
“This is a pivotal time in cancer research,” said Brian Leyland-Jones, Ph.D., institute director at the Edith Sanford Breast Cancer Institute, and a speaker at GTC’s recent “Oncology Biomarkers” conference. “We’re going to see this emergence of targeted therapies against individual genomic drivers, whatever their disease is, and breast cancer will be one of the first areas.”
The Sanford institute uses systems biology approaches to identify biomarkers. “Looking at one level isn’t enough,” says Dr. Leyland-Jones. “We are applying several complementary molecular profiling methods to tumor bank specimens from several international clinical trials that include all of the major breast cancer subtypes.”
These biomarker sets are being used to predict short-term (such as pathologic complete response) and long-term (such as distant disease free survival and overall survival) outcomes, as well as guide treatment strategies, and identify potential therapeutic targets.
Among several key technologies being used are:
- The whole-genome cDNA-mediated annealing, selection, extension, and ligation (WG-DASL) assay, which will enable the expression profiling of >24,000 protein-coding genes, is now available for use with FFPE specimens.
- Genomic instability, a hallmark of tumor progression and poor prognosis, can be assessed by array CGH with ultra–high-density arrays containing 2 million features providing unprecedented genomic resolution. Also, SNP analysis can now be performed on arrays that assay 1 million SNP loci.
- Genome-wide methylation arrays have become available to interrogate 27,578 CpG loci, covering more than 14,000 genes at single-nucleotide resolution.
Sanford also plans an initial study of 25 patients, “We will use several platforms (exome sequencing, RNAseq, various detailed protein arrays (e.g., phosphorylation arrays) to try to work out in 25 sequential patients in metastatic breast cancer what the individual drivers are,” explains Dr. Leland-Jones.
“Use of mouse models, including mouse avatars, will also be important. Past studies have suggested 10–13 pathway drivers including, for example, DNA repair, metabolism, and angiogenesis.
“I think it is going to be more complicated. It’s still anybody’s guess. Mine is that it’s going to be on the order of 30 to 40 pathways. No matter what it end ups being, I think treatment will require combinations of two or three agents for each of these subsets.”
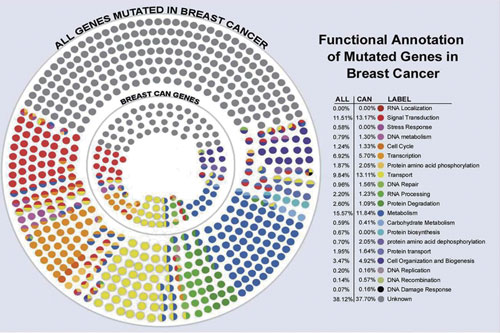
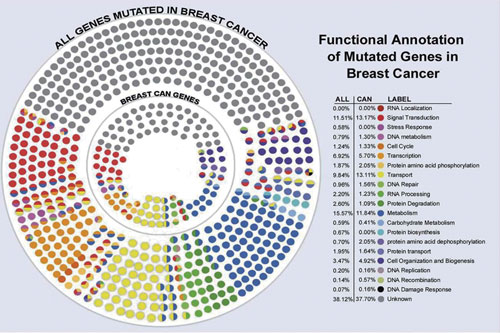
A multidimensional analysis of genes mutated in breast cancer, which will probably end up with 12 to 40 drivers. [Edith Sanford Breast Cancer Institute]
Complex Biosignatures
Fast-moving efforts to identify multiple molecular drivers to divide cancer into more subtypes means biomarkers will become increasingly complex biosignatures that change over time under the influence of drug treatment.
It’s likely that a combination of tests, not just one, will be required to monitor and tailor therapies informed by these changing biosignatures over the course of the disease, said Matthew Morrison, Ph.D., biology manager, medical diagnostics, GE Healthcare.
“The vision as we see it is to bring together our know-how in molecular imaging (PET and SPECT) and combine this with in vitro diagnostics, bringing in the recent acquisition of Clarient and SeqWright to add pathology and next-generation sequencing platforms,” said Dr. Morrison.
He also discussed two experimental GE imaging agents, one targeted toward angiogenesis imaging and the other toward c-met expression, to gauge patient response to therapy.
“These experimental imaging agents, Fluciclatide (the GE RGD PET tracer) and GE-212 (the GE cMet PET tracer), are both being developed as tools to allow drug therapy selection and drug therapy monitoring. This is important at a number of levels,” he said.
Targeted cancer therapies are costly and work only in sub-populations. “New agents that allow better use of limited healthcare funds to give the treatment to the right patient are important, and in addition stopping treatment when it is not beneficial or even potentially harmful to the patient is important,” added Dr. Morrison.
AGP’s Theranostic Strategy
Roughly 40% of ER+ breast cancer patients treated with anti-estrogen agents become resistant to the therapy. A.G. Pharmaceutical (AGP) has shown that GP88 (progranulin) holds promise both as a marker for resistance and as a target for therapeutic intervention.
In a paper published earlier this year in Breast Cancer Research, AGP’s founder and CEO Ginette Serrero, Ph.D., and colleagues wrote, “The survival factor GP88 is a novel prognostic biomarker, predictive of recurrence risk and increased mortality for nonmetastatic ER+ IDC patients. Of importance, our data show that GP88 continues to be a prognostic factor even after five years.
“These results also provide evidence that GP88 provides prognostic information independent of tumor and clinical characteristics and would support prospective study to examine whether GP88 expression could help stratify patients with ER+ tumors for adjuvant therapy.”
AGP has developed two tests, one tissue based (IHC) and one for fluids (ELISA) to detect the levels of GP88. Because GP88 is such a strong driver of tumor biology, she said, “those two diagnostics tests can have a life of their own in the current standard of care. That was very important to us because if you develop a test that is only linked to a therapy, and your therapy fails, your test is never going to see the light of day either.
Deciphering Diabetes
HbA1, a broad measure of glucose control, remains the dominant marker used in diabetes despite the disease’s substantial heterogeneity.
“We have a glucose centric picture for type 2 diabetes (T2D),” said Mark Broenstrup, Ph.D., director, biomarkers and diagnostics, diabetes, Sanofi. “We believe this picture is too simplistic and would like to dissect T2D into subtypes that better capture the pathophysiological effects in the individual.”
So far that has proven difficult to do even though diabetes’ many complications (blindness, nephropathy, heart disease, neuropathy, etc.) can be devastating. “We have a good understanding of individual mechanism, but understanding the interplay between mechanisms will require a systems biology view,” said Dr. Broenstrup.
Currently, roughly nine classes of drugs target diabetes. “It’s a challenge to position new drug classes if we only have glucose lowering information; the payers will tell us we have other principles that are generic and cheaper than your new one that also lower glucose. Why should we take yours?”
To manage costs Sanofi is leveraging public-private partnerships such as the European Medicine Initiative, which has three diabetes-related projects, each with a strong biomarker component. “We are leading IMIDIA, a program on beta cells, and DIRECT, a personalized medicine diabetes effort,” said Dr. Broenstrup.
Progress is being made on markers for progression to T2D, said Dr. Broenstrup. New reagents are enabling beta cell imaging (beta cell mass is used as measure of insulin-producing capacity of the pancreas). A few liver proteins also show promise as markers. An interesting one has emerged from GWAS studies and indicates most risk signs are in beta cells and not other affected organs.
Monitoring Autoimmune Responses
Identifying the antigens that elicit an autoimmune response can yield panels of biomarkers used to classify disease, predict patient outcomes, and characterize leads. Researchers from Life Technologies presented data from studies with the firm’s high-content ProtoArray and new ProtoPlex platforms demonstrating assay robustness and offering study design tips.
Niro Ramachandran, Ph.D., R&D manager, discussed work with protein microarrays composed of up to 9,000 purified full-length human proteins to evaluate immunological profiles across panels of serum samples. The goal was to identify biomarkers for SLE diagnosis, mesothelioma, and early detection of colorectal cancer.
Instead of printing antibodies on the slide as is often done, ProtoArray entails printing functional proteins, such as transcriptional factors, on the glass.
“It’s fundamentally a discovery tool designed to screen one-third of the proteome,” said Dr. Ramachandran.
The first coarse screening is done with ProtoArray followed by ProtoPlex, using Luminex xMAP technology, for high-throughput characterization of promising makers.
“ProtoPlex is similar to an ELISA technology except with ELISA you can test just one reaction in a well, i.e., one antigen or antibody. Luminex offers these color-coded beads that you can put different antigens on and the color can be read so it’s addressable.
“We can take 50 of these different beads and 50 different proteins and put them all in the same well and from one well you can pass serum against 50 antigens,” said Dr. Ramachandran, adding the concordance between the two platforms is around 70%, higher than mass spec.
Companies also use the platform to screen small molecules seeking protein binding information to help reveal mechanisms of action and identify leads and targets.
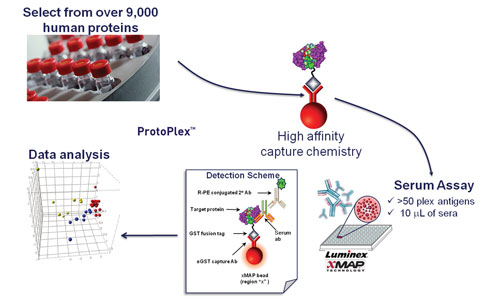
According to Life Technologies, ProtoPlex is similar to an ELISA but it uses Luminex xMAP technology, for high-throughput characterization of promising markers.
MSIA Platform
Roughly one year ago Thermo Scientific acquired Intrinsic Bioprobes, developer of a tip technology for purifying microscale amounts of protein or peptide antigen in complex biological fluid for subsequent mass spec. Now, Thermo is launching a set of products that comprise a complete mass spectrometric immunoassay (MSIA) platform and workflow that begins with sample prep and ends with mass spec detection.
Thermo’s tip immuno-affinity protein purification system works with Thermo’s TSQ Vantage mass spec to deliver protein and peptide quantification. A new liquid handler, Versette Pipetting Workstation, is also being launched.
Currently the platform is only for research although Thermo expects it will eventually be used as an IVT. The platform is Clinical and Laboratory Standards Institute compliant.
“We’ve been translating the tip technology into a more robust, better performing product combined with a liquid-handling systems,” said Urban Kiernan, Ph.D., senior scientist at Thermo.
“We have a couple of liquid-handling systems depending upon the amount of throughput users require.”
MSIA, say advocates, is a simpler way to generate accurate high-content data than competing technologies. Dr. Kiernan also expects the system to be used by companies needing to more accurately characterize their molecules such as in biosimilar development or by original biologics innovators seeking to differentiate their products from potential biosimilars.
“We’ve also been working with clients who use our technology to enrich proteins either from plasma or serum or even intracellular approach in which they are looking for post translational modifications that would essentially be a turn-on turn-off for a particular pathway,” he explained.
Breast Cancer Analysis Identifies Spectrum of Mutations
A major breast cancer analysis project has catalogued the spectrum of mutations that underpin the four basic categories of the disease. The data, published in Nature, represents the most comprehensive molecular blueprint of breast cancer generated to date, and could help researchers design and develop more effective drugs and combination treatment regimens that target each subtype of disease according to its molecular background.
The four categories are:
- Basal-like—About 15 to 20% of breast cancers. Most are “triple negative”—they are estrogen receptor-negative (ER-), progesterone receptor-negative (PR-), and human epidermal growth factor 2-negative (Her2-).
- Her2-enriched—About 7% to 12% of breast cancers. Typically, ER- and PR-, but also Her2+. These tumors have a fairly poor prognosis and are prone to early and frequent recurrence and metastases. Women with Her2+ tumors appear to be diagnosed at a younger age.
- Luminal A—About 40% of all breast cancers. Low or moderate tumor grade, hence they carry the best prognosis of all breast cancer subtypes. Usually are ER+ and/or PR+, but also Her2 negative.
- Luminal B—About 20% of breast cancers, typically ER+ and/or PR+. Also, Her2+ or show high number of cancer cells actively dividing. Prognosis generally poorer than luminal A due to factors including poorer tumor grade, larger tumor size, and p53 gene mutations.