May 15, 2011 (Vol. 31, No. 10)
Octet and Multi-Analyte Software Tool Help Facilitate Development of Quantitation Assay
Scientific projects are driven by the delivery of quality analytical data at the right time. The lack of robust analytical methods frequently hinders early progress due to the historical lag time that exists between project conception and assay development. For example, modern cell-line development methodologies are able to transition from transfection to selection in 30 days, allowing just a few weeks for development of a quantitation screening assay for clone selection.
While this lack of synchronicity cannot be eliminated, its minimization through advancements in analytical technologies represents an opportunity for increased efficiency, speed, and dexterity. This tutorial describes the rapid evaluation (15 minutes) of biosensor chemistries for a quantitation assay on the ForteBio Octet instrument using a multi-analyte workflow and demonstrating an accelerated pace of assay development.
ForteBio’s Octet systems offer a dip-and-read platform that provides easy, fast, and high-quality quantitation and kinetic data in a 96- or 384-well plate where existing methods have limitations in throughput, performance, and cost. The Octet instrument can read 16 quantitation samples in as little as 2 minutes and 384 quantitation samples in as little as 60 minutes, accelerating both assay development and delivery of decision-making data.
The systems utilize a BioLayer Interferometry (BLI)-based technology to detect molecular interactions on the tip of a fiber optic biosensor. BLI is an optical technique that analyzes the interference pattern of white light reflected from two surfaces: a layer of immobilized protein on the biosensor tip and an internal reference layer.
The instrument may be used for quantitation by measuring the rate of association of an analyte, such as a human IgG1 with a ligand immobilized on the biosensor, such as protein A. Higher antibody concentrations cause faster binding rates and analyte concentration is calculated from the binding rate using a standard curve. With highly specific ligands, such as the immunoglobulin-binding protein A, biosensors can directly quantitate antibody analytes in conditioned media and complex matrices.
When developing a quantitation assay on the Octet platform, the first question is, “which biosensor chemistry should I use?” ForteBio currently offers six out-of-the-box biosensors for quantitation: protein A, protein G, protein L, anti-human IgG Fc, antimurine IgG Fv, and anti-penta-His. As new biosensor chemistries are regularly developed, refer to www.fortebio.com/bio?sensor_types.html for the most current list. Custom quantitation biosensors can be developed using streptavidin and amine reactive biosensors.
By employing a multi-analyte workflow, this experiment evaluates the potential use of proteins A, G, and L biosensors for two antibody analytes using the Octet platform. Proteins A, G, and L are immunoglobulin-binding proteins originally sourced from microorganisms and have been widely adapted as protein analytical tools due to their high affinity and selectivity.
Proteins A and G bind to the Fc domain of antibodies while protein L binds to the kappa light chain. Proteins A, G, and L do not bind universally to all immunoglobulins and tables have been prepared as guidelines for their specificity. However, because each antibody possesses a unique set of binding properties, and because sometimes the isotype of the antibody is unknown, experimental evaluation against a target analyte is the most definitive means of choosing a biosensor chemistry for subsequent assay development.
Experimental Setup
100 µg/mL stock solutions of two purified antibodies (Ab1 and Ab2) were serially diluted in wells A1-G1 and A2-G2, respectively, of a 96-well plate (threefold dilution factor, six steps). 1X kinetics buffer with no analyte was pipetted into wells H1 and H2 as reference measurements. A sample plate map was constructed and sample information was entered (Figure 1A). The assay time was set at 120 seconds, and the shake speed of the sample plate was set at 400 rpm.
A biosensor tray was loaded with proteins A, G, and L biosensors and a biosensor plate map was prepared using the multi-analyte software feature (software version 7.0 and higher; Figure 1B). Biosensor assignment is semi-automated to facilitate scale-up to higher-throughput applications. Data collection was initiated and monitored in real time.
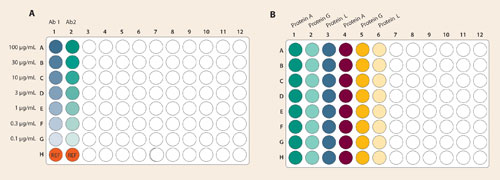
Figure 1. Sample plate map and biosensor tray map for the multi-analyte experiment: (A) Serial dilutions of Ab1 and Ab2 were prepared in columns 1 and 2 of a 96-well plate. Starting concentrations of 100 µg/mL were serially diluted with threefold dilution steps from row A through row G. Row H was reserved for reference controls (buffer with no analyte). (B) Multi-analyte experiments include a heterogeneous mixture of biosensors. Protein A, G, and L biosensors were loaded into columns 1 and 4, 2 and 5, and 3 and 7 respectively.
Results and Discussion
Execution of the multi-analyte experiment acquired data for three different biosensors and two analytes in one 15-minute walk-away experiment, producing 48 individual sensograms (three biosensors x two antibodies x seven dilutions of each antibody + six reference samples).
The raw data, a plot of binding (expressed in nm, the change in distance between the two reflecting surfaces) versus time contains a significant amount of information regarding biosensor-antibody affinity that may be used to select biosensor chemistry for further assay development. Analysis of the data requires consideration of both signal intensity (total nm shift) and the separation between individual traces. Greater signal strength improves signal to noise and the lower limit of detection while greater separation between individual traces improves accuracy and can indicate a greater dynamic range.
Grouping of the raw data by sample and sensor type (grouped view for quantitation, software version 7.0 and higher), enables a clear comparison of biosensor performance for each biosensor-analyte pair (Figure 2). Ab1 bound tightly to protein G, producing large nm shifts of up to 3 nm while smaller responses, due to weaker affinity, were observed with both the protein A and protein L biosensors. The protein G biosensor, therefore, would be selected for further development of a quantitation assay for Ab1.
Ab2 bound to all three biosensors with good affinity, producing maximum nm shifts greater than 4 nm at 100 µg/mL in all examples. At the highest concentration considered, 100 µg/mL, protein L produced the largest signal (approximately 12 nm at 100 µg/mL) with Ab2.
If assay performance at the higher end of the specified concentration range were prioritized, the protein L biosensor would be recommended for further development. However, at lower concentrations of the specified dynamic range, such as between 3.7–11.1 µg/mL, the protein G biosensor produced greater signal strength than the protein L biosensor. Moreover, the protein G biosensor produced greater separation between individual sensograms at lower concentrations than the protein L biosensor. Therefore, if the lower end of the specified dynamic range was prioritized, the protein G biosensor would be recommended for further development of a quantitation assay for Ab2.
In conclusion, the ForteBio Octet instrument and the multi-analyte software tool were used to rapidly evaluate three different biosensor chemistries for development of a quantitation assay of two antibody analytes. Data collection was recorded in less than 15 minutes of instrument time, demonstrating the accelerated pace at which Octet quantitation assay development can proceed. New graphing tools in software version 7.0 enable rapid analysis and visualization of the data. The experimental data highlights the diverse affinities of antibodies-immunoglobulin binding proteins interactions and the benefits of choosing a biosensor empirically.
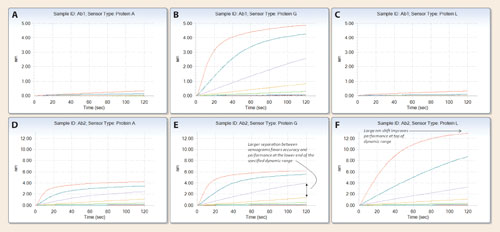
Figure 2. Multi-analyte data for protein A, G, and L biosensors with two analytes, Ab1 (A, B, C) and Ab2 (D,E,F). Ab1 binds with high affinity to protein G only. Ab2 binds to proteins A, G, and L. Protein L produced a higher nm shift (~12 nm) with 100 µg/mL of Ab2, favoring performance at the upper end of the specified dynamic range. Protein G (and protein A to a lesser degree) produced larger separations between individual sensograms at the lower end of the specified dynamic range, favoring performance at the lower end of the specified dynamic range.
Wesley McGinn-Straub ([email protected]) is product manager at ForteBio.



