October 15, 2010 (Vol. 30, No. 18)
Delineating the Roles of Cell- and Protein Analytical Chemistry-Based Assays
One of the major causes of delay in the pharmaceutical development process is inadequate forward planning on the use of analytical methods. Nowhere is this more acute than in the implementation of cell-based assays. It is never too early to develop these methods as the need to identify, quantify, and measure the potency of the drug substance is vital in producing meaningful and interpretable data.
Before going into more detail regarding the development of an analytical package, there are a number of considerations to bear in mind. First, we have found at Covance that it often takes longer than expected to develop, evaluate, validate, and transfer the majority of assays. This is even more of an issue for bioassays that can take six months or longer to transfer and validate.
Second, acceptance criteria should not be fixed too early. During development, the assay may have been carried out over many months by one or two dedicated individuals who gained significant experience and familiarity. This may not be representative of the actual release studies which, due to volume and scale-up, may need to be performed by a greater number of analysts in different locations.
Finally, it is often the detail that is not written into the SOP that can turn out to be the critical parameter that increases variability (e.g., such apparently minor details as angle of pipetting, ambient temperature, and the number of times an incubator door is opened and closed).
In this article, we look at the assays themselves and provide an overview of the key areas to manage to avoid unnecessary delays in the establishment of a robust and validated analytical package for large molecules (Figure).
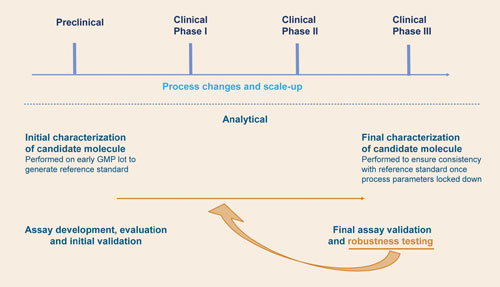
The development pathway of a biopharmaceutical, highlighting the key stages of assay development and validation: Robustness testing of the assay is often included in the final validation, but significant time-saving is possible and simplification of the validation can be achieved when robustness testing (analysts, analyst days, reagent equipment) continues during transfer and development and when lockdown of acceptance criteria (final validation) does not occur until after transfer and re-establishment.
Protein Chemistry Techniques
Protein chemistry techniques are used throughout the drug-development pathway to provide characterization, batch release, and stability-indicating assays together with pharmacopoeial methods.
These assays confirm the size (SDS-PAGE, SE-HPLC), charge distribution (IEX-HPLC, IEF), concentration (UVA280), physical characteristics (pH, appearance, osmolality, Karl Fisher, sub visible particulates), glycoproteins (O-and N-glycans, sialic acid), and impurities (deamidation, methionine oxidation, host-cell proteins) of the protein therapeutic and also any excipients.
At early stages of development, characterization of a protein therapeutic is essential for determining the methods that will be required to support toxicology, clinical, and stability studies as well as batch-to-batch consistency and comparability. In our experience, the main focus needs to be on developing appropriate assays that may require adaptation and on defining appropriate system suitability criteria.
Complete protein characterization provides essential information that can ease this process and pinpoint those assays that may need further development to support impurity analysis.
Problems can occur during the transition to Phase III, which requires validated assays and the need for impurity and excipient assays, which may not have been anticipated. Again, this process is eased by assessing assay robustness as early as possible and prior to validation. At this stage, the development of host-cell protein assays also becomes a priority as the manufacturing process is locked down and the reference host-cell protein can be identified. Both these processes require additional time and ideally should be planned up to a year before they are required.
There is always a balance between the ideal development plan and cost considerations. However, good characterization at an early stage, robustness testing, and planning provide useful information and experience to draw upon.
The Table gives an indication of the methodologies frequently employed during the drug-development life cycle within protein chemistry. While not exhaustive, the techniques reviewed in this article reflect those typically used to support characterization, batch release, and stability. Where appropriate, assays have been identified that can be used in a later phase to give additional information in support of batch release and/or stability testing.
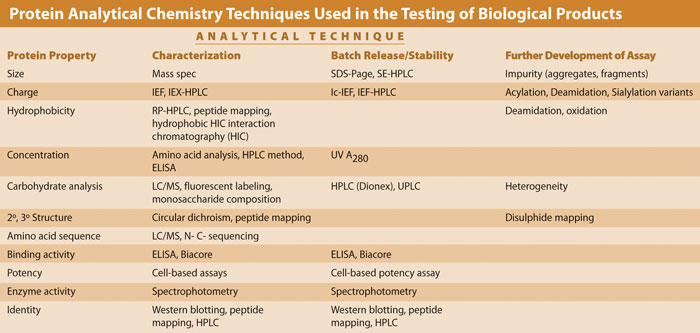
Protein analytical chemistry techniques used in the testing of biological products
Cell-Based Assays
The cell-based assay plays a critical part in the release and stability package for large molecules as it provides data on the biological activity of the therapeutic under investigation. To avoid delays in development of the molecule, the design of the cell-based assay must be considered at an early stage. It needs to be based on the mode of action of the drug, which can be challenging when the mechanism(s) involved are not well defined.
For novel therapeutics, the mode of action is usually unique. As a result, the cell-based assay used for release testing is likely to be unique as well. The design of the assay should take into account the fact that it needs to be easily reproducible as the results will lie on the critical path for release or for stability measurements.
The use of “primary” cell lines, for instance, may initially seem the best option for demonstrating the drug’s mode of action. However, due to the often inherent high variability and reproducibility of these cells, it can be difficult to validate such assays and they should be replaced as soon as possible with more stable cell lines and well before late-stage release.
On the other hand, the use of reporter gene constructs can be the platform of choice for a release assay due to the high sensitivity, tight reproducibility, and, even more importantly, the short time required for the assay (a few hours incubation as opposed to 7–10 days for some assays using primary cell lines).
It is also important to consider the transfer of the cell-based assay to a second quality control laboratory. As a CRO, we are often required to develop and optimize cell-based assays on behalf of our clients prior to validation. However, we are most frequently required to transfer in assays that have been developed externally. On the face of it, this would be expected to be a simple process and it is, so long as it is approached in the correct manner.
Transferring a cell-based assay to a QC lab is a test of the robustness of the assay. It is crucial to think about assay transfer prior to final validation of the method and, in particular, prior to the establishment of acceptance criteria. Assay transfer from development to QC needs to be taken into consideration as a crucial part of assay validation. Without such consideration, assays that already have fixed acceptance critieria during development could result in, at least initially, failure to meet the expected values in the QC laboratory.
As the number of assays being performed grows as the scale increases, then more analysts, greater cell volumes, and more equipment will be involved in carrying out the method. Ideally, the performance of the assay should be evaluated by the external QC lab prior to validation, when the acceptance criteria can still be adjusted and critical parameters can still be optimized.
Specific training of analysts on a particular assay is vital and a GMP requirement. Having sufficiently trained staff requires proper planning as part of assay development and transfer.
Finally, the performance of the assay must be monitored on a regular basis. Data is then used to trend performance over time. Changes in suppliers of reagents, reference standard materials, plates, incubators, equipment, analysts, etc. can significantly affect the performance of cell-based assays and should be continuously evaluated and cross-validated. If necessary, system suitability acceptance criteria should be reviewed and adjusted. In extremis, the assay may need re-validation if significant changes in performance are observed.
Relevant statistical analysis of the assay is essential for performing this continuous monitoring and is a topic in its own right.
Camille Dycke, Ph.D. ([email protected]), is manager, bioassays; Sian Estdale, Ph.D., is manager, protein chemistry; and Carl Martin, Ph.D., serves as vp and CSO in the biotechnology division at Covance.



