November 15, 2012 (Vol. 32, No. 20)
Results from Studies with Two New Reagents Showed Compatibility with Accepted Methods
Automation is used for many applications to reduce variation caused by manual handling and to obtain reproducible results in high-throughput assays. High-throughput applications, such as knockdown studies or target screenings, often include cell transfection. In this study, X-tremeGENE HP and X-tremeGENE 9 Transfection Reagents (Roche) were tested for their compatibility with an automated transfection workstation, the Biomek FX (Beckman Coulter).

Figure 1. Formation of the transfection complex, cell seeding, and transfection were performed in a 96-well format using an automated workstation.
Material and Methods
Automated Transfection
Cell seeding and transfection were performed using the Biomek FX automated workstation. For the transfection, X-tremeGENE HP and X-tremeGENE 9 reagents (Roche) were prediluted in Opti-MEM (dilution factor: 1:50 for X-tremeGENE HP reagent, 1:33 for X-tremeGENE 9 reagent). The EGFP-PC3.1 plasmid was prediluted in Opti-MEM (1 µg/100 µL). The transfection-complex was formed by pipetting the same amount of plasmid dilution into the dilution of transfection reagent, followed by mixing and incubation for 30 min.
Cell Culture and Reverse Transfection
HeLa cells (ATCC® CCL-2™) were cultured in MEM with supplements at +37°C in a 5% CO2 humidified atmosphere in T75 flasks until a confluence of about 70% was reached (Figure 1). Cells were harvested by trypsinization shortly before transfection, washed and resuspended in fresh medium (1.2×105 cells/mL). Reverse transfection was performed by pipetting 100 µL cell suspension to each well already containing the transfection complex.
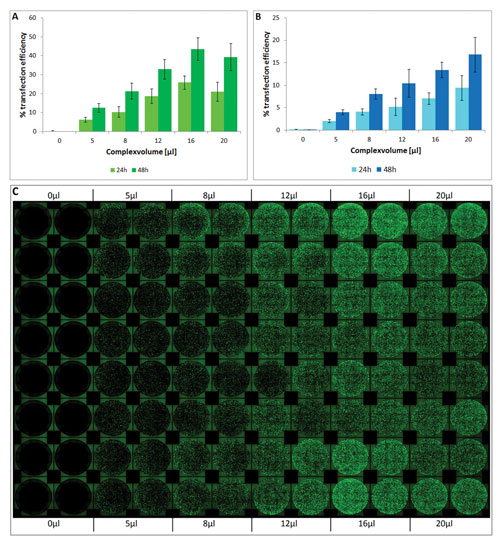
Figure 2. Analysis of transfection efficiency in the automated workflow: The efficiency was measured 24 and 48 hours post transfection and calculated by the ratio of area covered with fluorescing cells compared to total area covered with cells. (A) Transfection using the X-tremeGENE HP Reagent; (B) Transfection using the X-tremeGENE 9 Reagent. (C) Top view showing the fluorescence, as well as the reproducibility (16 replicates) of the automated workflow using the X-tremeGENE HP Reagent.
Results
- Formation of the transfection complex, cell seeding, and transfection were successfully performed in 96-well format using an automated workstation. Increasing volumes of transfection complex from 0 to 20 µL were added to each well in replicates of s16 for each volume.
- The efficiency of transfection was found to be directly dependent on the added volume of transfection complex. As expected, higher transfection efficiency was measured at higher complex volumes unless a plateau phase was reached (Figure 2).
- Both transfection reagents showed very low cytotoxicity for all volumes tested (not shown).
- X-tremeGENE HP and 9 Transfection Reagents are fully compatible with an automated transfection method and show a high rate of reproducibility.
Georg Hannig ([email protected]) is research scientist, Bärbel Rueger is research manager, and Cordula Jany is marketing manager at Roche Diagnostics. For life science research use only. Not for use in diagnostic procedures. X-TREMEGENE is a trademark of Roche. All other product names and trademarks are the property of their respective owners.



