June 1, 2012 (Vol. 32, No. 11)
The electrifying possibility of reprogramming human cells to different fates has huge ramifications for human therapeutics. The technology is gaining momentum as scientists make headway to better understand the molecular and cellular aspects of reprogramming.
Some of these new and emerging strategies were showcased at Gordon Conference’s recent “Reprogramming Cell Fate” meeting. For example, cutting-edge studies described how only a handful of key transcription factors were needed to entirely reprogram cells.
Pioneer transcription factors help untangle and pry open targeted chromatin sites. Equally important is delineating how to remove inhibitory mechanisms. Understanding the complex maze of reprogramming may ultimately provide new targeted therapies for a host of human diseases.
The intrinsic cellular programming that primordial germ cells (PGCs) undergo can be difficult to study. PGCs represent the common origins of spermatozoa and oocytes, which are specified from the ectoderm (epiblast) of human embryos in the second week of development. They eventually migrate into the gonads where sexual differentiation begins about six weeks after conception, ending with development of egg or sperm.
The fate of a cell is considered to be “specific” when it is capable of following an autonomous program following closure of other options. Specification of PGCs is linked with extensive epigenetic reprogramming that is critical for establishing totipotency, the ability to give rise to a whole new organism.
M. Azim Surani, Ph.D., Marshall-Walton professor at the Gurdon Institute, University of Cambridge, U.K., is examining cellular reprogramming in a mouse model. He said his group focused on establishing a cell culture-based system able to provide mechanistic insights into how PGCs induce reprogramming in germ cells.
“We use murine-derived epiblast stem cells (epiSCs) to investigate aspects of epigenetic reprogramming and the roles of key genes in early germ cells. Epiblast stem cells are derived from the early-stage embryonic stage after implantation of blastocysts, about six days into development, and retain the potential to undergo reversion to embryonic stem cells (ESCs) or to PGCs.”
Dr. Surani and colleagues developed two critical reporter epiSC lines. “There are two major attributes of epiSCs that can be exploited to generate reporter lines for exploring epigenetic reprogramming. One is monitoring the re-activation of the X chromosome.
“Although there are two X chromosomes in females, the inactivation of one is necessary for cell differentiation. Only after epigenetic reprogramming of the X chromosome can pluripotency be acquired. Pluripotent stem cells can generate any fetal or adult cell type but are not capable of developing into a complete organism.”
The second read-out is the activation of Oct4, a key transcription factor involved in ESC development. The expression of Oct4 in epiSCs requires its proximal enhancer.
“Using these two systems, we found that transcriptional regulators Prdm14 and Klf2 synergized to accelerate and enhance X chromosome reactivation and DNA demethylation, which are among the most important reprogramming events seen in early PGCs. While neither alone was sufficient to enhance this epigenetic programming, together they provided a potent combination for reprogramming. Further, the same combination provided a fast and efficient enhancement of Oct4 activity.”
Dr. Surani said that their cell-based system demonstrates how a systematic analysis can be performed to analyze how other key genes contribute to the many-faceted events involved in reprogramming the germline.

Researchers have been able to reprogram a patient’s own cells to produce stem cells that exhibit few DNA structural mutations. [Arvid Emtegren/Istockphoto.com]
Reprogramming Expressway
A number of other recent studies have shown the importance of Oct4 for self-renewal of undifferentiated ESCs. It is sufficient to induce pluripotency in neural tissues and somatic cells, among others.
The expression of Oct4 must be tightly regulated to control cellular differentiation. But, Oct4 is much more than a simple regulator of pluripotency, according to Hans R. Schöler, Ph.D., professor in the department of cell and developmental biology at the Max Planck Institute for Molecular Biomedicine.
One example Dr. Schöler cites is that Oct4 is downregulated in all three germ layers during gastrulation of the embryo.
“This suggests that Oct4 has a critical role in committing pluripotent cells into the somatic cellular pathway. When embryonic stem cells overexpress Oct4, they undergo rapid differentiation and then lose their ability for pluripotency. Other studies have shown that Oct4 expression in somatic cells reprograms them for transformation into a particular germ cell layer and also gives rise to induced pluripotent stem cells (iPSCs) under specific culture conditions.
“These studies indicate that Oct4 is not simply a reprogramming factor,” continued Dr. Schöler. “Instead it is becoming evident that it is the gatekeeper into and out of the reprogramming expressway. By modifying experimental conditions, Oct4 plus additional factors can induce formation of iPSCs, epiblast stem cells, neural cells, or cardiac cells. It is likely these results will be soon extended to include many other lineages, making Oct4 a potentially key factor not only for inducing iPSCs but also for transdifferention.”
Dr. Schöler is now concentrating on reprogramming cells for multipotency, the potential to give rise to a defined set of cell lineages.
“Therapeutic applications might eventually focus less on pluripotency and more on multipotency, especially if one can dedifferentiate cells within the same lineage. Although fibroblasts are from a different germ layer, we recently showed that adding a cocktail of transcription factors induces mouse fibroblasts to directly acquire a neural stem cell identity. This approach demonstrates that differentiated cells can be directly programmed into specific types of somatic stem cells. The next step will be to push back cells to stem cells of the same tissue.”
Pioneer Transcription Factors
Pioneer transcription factors sound as if they should be riding in a covered wagon blazing a trail across the Wild West. While that’s not their role, they do, however, take the lead in facilitating cellular reprogramming and responses to environmental cues. Multicellular organisms consist of functionally distinct cellular types produced by differential activation of gene expression.
Transcription factors help this process as they seek out and bind specific regulatory sequences in DNA. That may be no easy feat to accomplish as DNA is coated with and condensed into a thick fiber of chromatin. In each eukaryotic cell, ~2 meters of DNA is packaged into a nucleus of only several microns in diameter.
“We discovered pioneer transcription factors in 1996 and were surprised to find that when they first attached to chromatin, they did not immediately activate the corresponding genes at that site,” said Kenneth S. Zaret, Ph.D., professor, department of cell and developmental biology, University of Pennsylvania School of Medicine. “By contrast, the pioneer factor endows the competence for gene activity, being among the first transcription factors to engage and pry open the target sites in chromatin.”
Dr. Zaret notes that at any one time, the vast majority of potential DNA-binding sites are not occupied, suggesting that most nuclear DNA isn’t easily accessible. The question prompted is: how do pioneer factors perform their role? An example is the pioneer transcription factor FoxA, a member of the Forkhead box family of transcription factors.
“FoxA factors engage and subsequently help activate silent genes. They are expressed in the foregut endoderm of the mouse and are necessary for induction of the liver program. We looked at the genomic location of FoxA in the adult mouse liver. We found that nearly one-third of the DNA sites bound by FoxA in the adult liver occur near silent genes.”
These studies help explain the progression of cells to cancer. “We found that in sites near where FoxA bound to silent genes, there were motifs for the transcriptional repressors Rfx1 and type II nuclear hormone receptors (HHR-II). We then confirmed protein binding and subsequent repression of adjacent FoxA sites at a novel and otherwise silent enhancer element for Cdx2. Cdx2 mediates differentiation of intestinal epithelial cells and is not normally expressed in esophageal cells. Thus, Rfx1 restricts FoxA1 activity at silent genes.
“We next examined Rfx1 in human esophageal epithelium and in adenocarcinoma and determined that Rfx1 levels decline during esophageal cancer progression, which could allow FoxA to activate Cdx2 in esophageal cells. Overall this suggests that when such networks are perturbed, cancer progression may result. It also suggests that Rfx 1 expression may serve as a biomarker for esophageal cancer progression.
“These studies illustrate how evaluating the binding of pioneer factors to silent genes may reveal the basis for how cell perturbations deregulate gene expression and progress to cancer,” Dr. Zaret concluded. “Thus, understanding how silent genes can be activated provides insights not only for development but also for cellular programming and pathogenesis.”
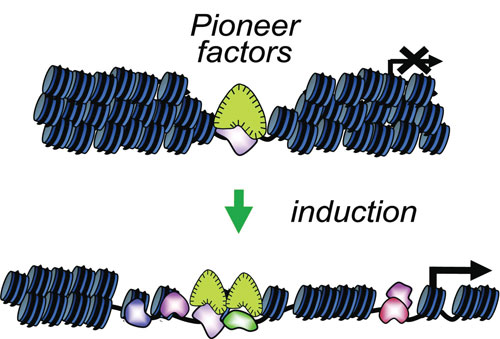
Pioneer factors are distinguished by being able to bind silent genes in chromatin. By binding silent chromatin, pioneer factors can enable the binding of other regulatory factors to the DNA when such factors are induced by different stimuli. Relevant stimuli include those that occur during embryonic development, hormonal induction, and during pathogenic processes that activate genes inappropriately, leading to disease. [University of Pennsylvania School of Medicine]
Germ Cells to Neurons
While transcription factors are critical elements for inducing the identity of specific cell types in multicellular organisms, they also are cell-type specific and can be limited when ectopically expressed in other cells, notes Oliver Hobert, Ph.D., professor, department of biochemistry and molecular biophysics, Columbia University Medical Center. According to Dr. Hobert, the generally accepted paradigm in the field is that transcription factors can exert their activities only within a specific cellular context.
Dr. Hobert indicates that one of the goals in the field is to develop ways to overcome such cellular context dependency. That is, one could envision generating specific cell types using a cocktail of transcription factors to reprogram cells for the creation of in vitro models for specific diseases or to provide a source of material for cellular replacement therapies.
To better elucidate the mechanism for context dependency, Dr. Hobert and colleagues conducted cellular reprogramming studies in the nematode, Caenorhabditis elegans. They initially monitored the effect of expressing the zinc finger transcription factor CHE-1 that programs gustatory neurons called ASE neurons. Next they established from an RNA interference library that the chromatin factor lin-53 controls context dependency of CHE-1.
Dr. Hobert and colleagues determined that to convert germ cells into specific neuron types, mitotic germ cells required not only the addition of the specific transcription factor, but also the removal of inhibitory factors—in this case the histone chaperone lin-53, a component of several histone modifying and remodeling complexes. They also found they could chemically inhibit histone deacetylases to achieve germ cell to neuron conversion.
Dr. Hobert suggests these studies have important ramifications. That is, removing even a single chromatin factor, while also inducing individual transcription factors, dramatically impacts neuronal development. He feels this data is a testament to the simplicity but elegance of programs that control neuronal differentiation.
The emerging picture is that distinct sets of factors orchestrate and coordinate a terminal differentiation regulatory routine. Thus, cellular reprogramming depends not only on expressing the correct transcription factors for inducing a specific neuronal fate, but also removing the inhibitory mechanisms that govern transcription factor activity.



