June 15, 2013 (Vol. 33, No. 12)
Richard A. A. Stein M.D., Ph.D.
Rooted in mythology, legends, and folklore, the prospect of re-growing body parts has fascinated humanity since ancient times. One mythical creature from ancient Greece, the nine-headed Hydra, was able to grow two heads in place of one.
And Prometheus, whose liver regenerated overnight, often prompts questions on whether early societies were aware of tissue repair and regeneration.
The field of stem cell biology opened the attractive possibility to use cells that can be obtained from adult organisms to generate any cell type. However, translating basic research advancements into clinical benefits presents challenges at multiple levels.
“In any kind of cell replacement therapy, I believe the integration of the cells made in the laboratory into a patient is going to be essential, and one of the most challenging problems to be solved is to find out how to get laboratory-made cells to integrate into a patient,” says Sir John B. Gurdon, Ph.D., emeritus professor at the University of Cambridge, distinguished group leader in the Wellcome Trust/CRUK Gurdon Institute, and co-recipient of the 2012 Nobel Prize in Physiology or Medicine.
Research efforts in Dr. Gurdon’s group are focusing on understanding cellular and molecular mechanisms that allow cells to reach and maintain the differentiated state, and on unveiling the intricacies of embryonic gene reactivation. While regenerative medicine has the potential to impact virtually every clinical area, its promises are particularly important for diseases that have therapeutic options that are currently limited, ineffective, or nonexistent.
“Clearly, any advances in the field of neurodegeneration would be of maximum importance, but any other field that looks really promising, like the restoration of vision, would greatly improve the lives of very many people,” says Dr. Gurdon.
Advances in stem cell biology not only brought therapeutic promises through innovative approaches, but have also created the need to revisit long-standing concepts.
A New Paradigm
“For years, we have defined bone marrow stem cells in terms of their ability to differentiate along a specific lineage, but we missed the possibility that the bone marrow could be a resource to generate adult tissues,” says Richard A. Lerner, M.D., professor of immunochemistry at The Scripps Research Institute.
Investigators in Dr. Lerner’s lab recently reported the possibility to transdifferentiate CD34+ human myeloid bone marrow stem cells into neural progenitor cells using antibodies, in an approach different from those employed to date. To identify a large number of agonist antibodies that each can bind to a different region of the G-CSF receptor (G-CSFR) molecule, Dr. Lerner and colleagues designed a two-step selection strategy.
First, antibodies derived from a phage combinatorial library, which had been converted to a plasma membrane-binding format, are selected based on their ability to bind a membrane-bound version of G-CSFR. Second, based both on binding and function, selected agonists are enriched for those possessing unusual or rare functions.
This strategy favors the selection of antibodies that are generally not identified in solution and provides the opportunity to explore a large number of agonists, each of them binding to a different region of the receptor. Additional strengths of this approach are the presentation of the receptor in its natural environment, where it assumes physiologically relevant conformations, and its ability to test for direct binding.
Among the antibodies they generated, Dr. Lerner and his colleagues identified one that, unlike the natural ligand G-CSF, was also able to activate CD34+ stem cells and initiate neurogenesis.
“What we found is a special antibody to a known receptor whose activation generates more white blood cells but, in this case, the antibody stimulated differentiation into brain cells,” Dr. Lerner explains.
This revealed a new paradigm, an agonist antibody binding, in a population of identical cells, the same receptor as the ligand, but inducing a distinct cell fate, by biasing signaling to specific downstream pathways. While antibodies that can generate other cell types, such as red blood cells, platelets, or dendritic cells, have previously been described, in each of those examples the respective differentiated cells represented a known potential of that specific cell lineage. “In this case, what we observed is not a known potential, and the broad question that emerges is whether we have missed the overall capacity of the bone marrow,” he says.
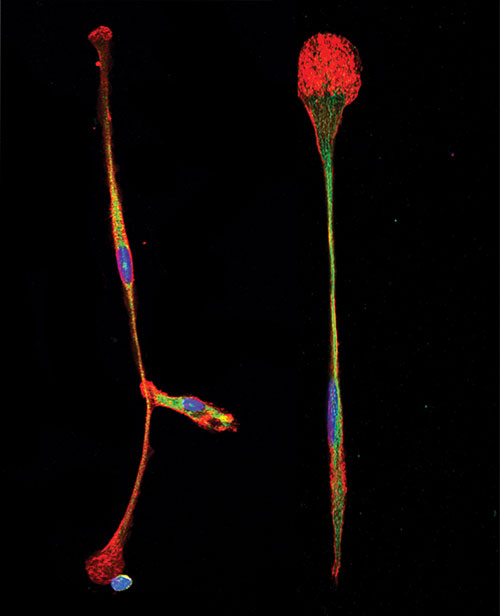
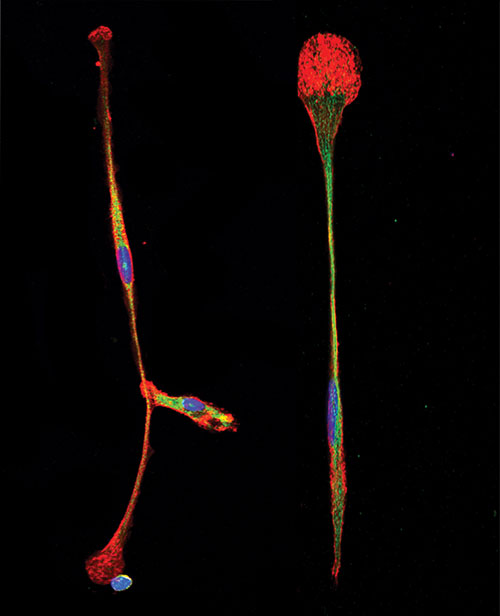
Scientists at The Scripps Research Institute have found a simple way to turn bone marrow stem cells directly into brain precursor cells, such as those shown here. [Richard A. Lerner]
Key Choices
One of the key questions is making choices in terms of the many available sources of stem cells and the different approaches that exist to grow and select for them,” says Edward L. Field, COO at Cytomedix.
Historically, the identification of stem cells has relied on surface markers characteristic of each cell type. “What makes Cytomedix unique is that we select stem cell progenitors based on an intracellular marker, the enzyme aldehyde dehydrogenase,” Field says.
A fluorescent aldehyde dehydrogenase substrate allows the prospective isolation of stem and progenitor cells in which this enzyme is upregulated. “We are using this population of ALDH-bright stem cells that we believe are very potent in tissue repair and regeneration,” he explains.
Another distinguishing feature of the Cytomedix technology is that the ALDH-bright stem cells are a heterogeneous population that include hematopoietic, mesenchymal, neural, and endothelial stem cells. The ability of each of these cell types to participate in tissue repair and regeneration in very specific ways promises advantages over other approaches that rely on homogeneous stem cell populations. “Investigators are increasingly finding that it is the complementary activity of these stem cell types, that provides most benefits,” Field says.
The company recently became part of the first Phase II randomized trial exploring the benefits of autologous stem cell therapy in patients with intermittent claudication, an NIH-funded initiative managed by the Cardiovascular Cell Therapy Research Network.
Technical Issues
“Developing approaches to repair tissue damage is one of the areas of greatest unmet need in multiple sclerosis,” says Jeffrey A. Cohen, M.D., professor at the Cleveland Clinic Lerner College of Medicine of Case Western Reserve University. Dr. Cohen and his colleagues are focusing on developing therapeutic strategies for multiple sclerosis by taking advantage of mesenchymal stem cells, which offer multiple advantages, including the existence of established experimental approaches to isolate and expand them in culture, but their use is also accompanied by certain challenges. “There is a long list of technical issues that need to be addressed in every experiment,” explains Dr. Cohen.
While the experience with mesenchymal stem cells has been promising in terms of safety, certain concerns exist regarding potential unanticipated effects. “The approach in our program is to start with a very meticulous safety study and subsequently build upon that,” says Dr. Cohen. Another challenge stems from the paucity of data tracking the administered cells in humans, despite an abundance of results from animal studies showing that transplanted cells survive and reach their intended locations.
Dr. Cohen and colleagues recently completed the enrollment of 24 participants into a Phase I safety study of autologous mesenchymal stem cells, and no safety concerns were noted. “We plan to proceed with a Phase II study to illustrate the proof of concept for the benefits of this approach, and we also anticipate tracking the administered cells in vivo,” says Dr. Cohen.
To the Clinic
“We have developed a highly differentiated product with multiple biological activities that lead to its therapeutic effects,” says Nick Colangelo, president and CEO of Aastrom Biosciences. Colangelo’s firm has developed an approach that is characterized by several distinguishing features. The proprietary automated system that expands (up to 300 times) key cell types collected from a patient’s bone marrow, and the administration of the expanded cells back to the same patient, removing the risk of rejection and eliminating the need for immunosuppressive therapy, are some of the aspects that make this approach unique.
An additional hallmark of this product lies in its multicellularity. Among several cell types that are incorporated, the CD90+ mesenchymal stromal cells and the CD14+ alternatively activated macrophages are particularly important, due to several sets of activities that account for their therapeutic effects. These cells secrete anti-inflammatory molecules such as IL-10, enzymes involved in extracellular matrix remodeling, and potent angiogenic factors such as VEGF.
The activities of these cytokines and growth factors are complemented by cell-dependent activities, with macrophages, in particular, modulating the immune response by phagocytizing cellular debris and exerting cardioprotective effects that include oxidized LDL uptake and cholesterol efflux.
“It is this combination of secreted factors and direct cell activities that leads to the therapeutic effects that we have seen, which involve resolving inflammation, remodeling damaged tissue, and promoting angiogenesis,” Colangelo explains.
One of Aastrom’s recent efforts is the development of stem cell-based therapeutic approaches for advanced heart failure that develops as a result of ischemic dilated cardiomyopathy (CM). After completing two Phase IIa open-label studies, investigators at Aastrom initiated a 12-month Phase IIb study in 2012.
“We enrolled our first participants in early April 2013, and treated these patients in the second half of the month,” Colangelo says. Ischemic CM represents one of the most frequent forms of heart failure, and with the scarcity of therapeutic options that are currently available, the development of cellular therapies emerges as a lifesaving approach for this patient group.
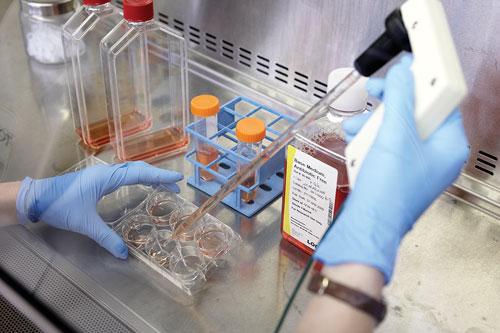
According to Aastrom Biosciences, its cell-processing system selectively expands mesenchymal cells, monocytes, and alternatively activated macrophages, up to several hundred times more than the number found in the patient’s bone marrow, while retaining many of the hematopoietic cells collected from only a small sample of the patient’s bone marrow.
New Pathways
We unveiled the role of a previously unrecognized pathway that is important for maintaining the stem cell state,” says Carla M. Koehler, Ph.D., professor of chemistry and biochemistry at the University of California, Los Angeles. In a chemical screen that proposed to identify inhibitors of the mitochondrial FAD-linked sulfhydryl oxidase Erv1, essential for mitochondrial biogenesis and cell survival, Dr. Koehler and her colleagues identified MitoBloCK-6, a small molecule that selectively inhibits the mitochondrial redox-mediated import of Erv1 substrates.
Despite its ability to inhibit Erv1 and its human homolog ALR in vitro, MitoBloCK-6 did not affect the in vivo growth and function of differentiated cells but, surprisingly, caused apoptosis in pluripotent human embryonic stem cells.
“When we differentiated stem cells they were no longer susceptible, and when we reprogrammed them to the pluripotent state, they again became susceptible to this compound,” she says.
MitoBloCK-6 is an attractive option to remove undifferentiated stem cells from differentiated cell populations, and presents multiple advantages over existing protocols. “We are further exploring this compound, and hope that it can be used at a certain time during the differentiation protocol to allow the removal of cells that failed to differentiate, without affecting the ones that became committed to become specific cell types,” Dr. Koehler adds.
This finding has an additional, more profound significance that extends beyond the field of stem cell biology, as it underscores the cardinal position that basic research plays in advancing translational research.
“There are so many aspects that we do not understand about the function of many cellular pathways at the basic level, and it is difficult to explore the endpoint without performing and trying to understand the basic research. In this case, we are still trying to understand why mitochondria are important for stem cell survival,” Dr. Koehler says.
Basic and translational research in stem cell biology and regenerative medicine are witnessing an exciting time. More than 5,000 clinical trials on stem cell therapies are currently under way globally, promising to revolutionize therapeutic prospects. As our knowledge about differentiation and regeneration is advancing, it is essential to appreciate and capitalize on the interdiscliplinary spirit that has become a defining feature of this vibrant and fascinating field.
Cell-Based Therapies
“Having for several decades been at the forefront of the biomanufacturing and bioprocessing of recombinant proteins and monoclonal antibodies, we envision cell therapies as the next wave that is coming along in this market space,” says Firman Ghouze, Ph.D., director of the cell bioprocessing group at GE Healthcare.
One of the research initiatives at GE Healthcare is directed toward the development of platforms that facilitate the transition toward large-scale applications of such therapies. In the case of cell-based therapies, this involves providing solutions that enable investigators to pursue therapeutic programs in a way that is robust and cost-effective—two important factors to consider, particularly for large clinical trials.
“We are also trying to build on the scalability,” adds Dr. Ghouze. Significant efforts are being focused toward developing automated and closed systems, which promise multiple benefits, including increased yield, decreased contamination risks, maximal effective use of the manufacturing space, a reduction in the amount of manual work, and the possibility for high-throughput, parallel processing.
Over the past few years, these have represented some of the key aspects that turned out to be instrumental in the bioprocessing of recombinant proteins. “I can see these considerations becoming important as we develop solutions for cell-based therapies,” explains Dr. Ghouze.