March 1, 2012 (Vol. 32, No. 5)
Bioanalysis of drugs in development is shifting toward high-sensitivity and high-throughput methodologies based on LC/MS platforms capable of drug and metabolite identification and quantification, with the ability to detect low-volume analytes and to discriminate between closely related molecules in complex biological samples. At the upcoming “Pittcon” conference, several presentations will focus on strategies for optimizing the use of LC/MS for analyzing small molecule and biological drugs in development. The tools and techniques described all aim for a simplified and automated workflow, increased accuracy and reproducibility, and optimal detection of the analyte of interest with minimal interferences and background noise.
Robert Ellis, Ph.D., R&D director at AB Sciex, will focus his talk on four main trends in LC-MS/MS analysis of small molecule analytes: improvements in selectivity, capacity, confidence, and sensitivity.
LC-MS/MS provides time- and chemistry-based (on LC separation) and mass-based (on MS detection) selectivity. However, background interference and the presence of isobaric molecules can still compromise the ability to discriminate and quantify the target analyte.
Dr. Ellis describes method development work at AB Sciex that exploits differences in ion mobility to design an orthogonal sample- prep method that makes it possible to separate out isobaric or background interferences and to distinguish between isomeric forms of a compound. By passing samples through an asymmetric waveform field before they enter the mass spectrometer, this technique differentiates analytes based on their trajectory, which depends on their size and shape.
For the second trend described by Dr. Ellis, enhancing capacity of LC-MS/MS analysis, strategies may include increasing the throughput of sample processing or extracting more information from each experiment. He will discuss the benefits of the SWATH™ technique, in which groups of ions are scanned sequentially across defined time periods and across the full mass range.
With SWATH, instead of focusing MS analysis on the detection of a particular target analyte, all of the ions are detected, generating a complete dataset that can then be processed and analyzed to obtain various types of information and to test multiple hypotheses without the need to rerun a sample. The main challenge with this approach is the need for complex algorithms and a high-capacity data-management system to collect, store, and analyze the data.
Improving confidence in bioanalytical results will require optimizing the speed and accuracy of existing systems, doing multiple reaction monitoring (MRM) more efficiently, and collecting more data points per peak.
“In R&D you have to hit the sweetspot,” says Dr. Ellis. “You need to recognize that optimizing one parameter might compromise another.”
The goal of increased sensitivity depends mainly on improving the signal-to-noise ratio and being able to achieve accurate analyte detection in a smaller sample.
On the LC side, improvements in sensitivity are being achieved using techniques such as microflow-LC, which enables separation of small molecules at rates of 10–15 µL/min, according to Dr. Ellis. As both sampling efficiency and the efficiency of MS ion sources improve, more ions will be generated in a shorter amount of time, yielding a better signal.
Ichiro Hirano, manager of the mass spectrometry business unit at Shimadzu, will present data related to the development of an automated LC/MS method for assessing the purity of the active pharmaceutical ingredient (API) atorvastatin calcium hydrate, sold under the trade name Lipitor (Pfizer), which could be applied in the development of a generic form of the drug.
Conventional methods development for purity analysis of small molecule drugs tends to rely on nonvolatile buffers. However, as the bioanalytical workflow shifts toward the use of LC/MS, a volatile buffer is needed for the mobile phase, which introduces uncertainties as the same chromatographic pattern cannot be reproduced.
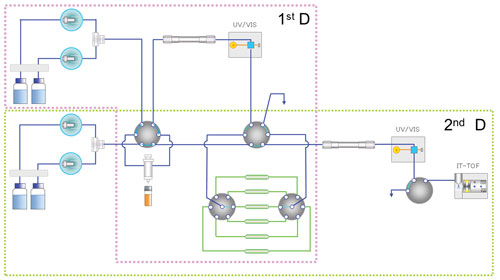
Hirano described a 2D-LC/MS-IT-TOF approach in which a nonvolatile buffer is used in the first-dimension LC process to separate the impurities in an atorvastatin sample. The impurity peaks are collected and fractionated by passing them through a loop module controlled by a high-pressure valve system. The impurity fractions are automatically captured based on the known retention times of each impurity, which are input into the system by the operator.
The system uses the retention time information to create a time program for fractionating the peaks. The fractionated impurities are then injected onto the second-dimension LC/MS system for accurate mass analysis using a volatile buffer.
In the study presented, the atorvastatin impurities are analyzed using ion trap (IT) time-of-flight (TOF) mass spectrometry, using both positive and negative ion polarity. Hirano provides spectral data showing that the 2D-LC/MS method yields individual peaks for each of the unknown impurities and enables the determination of their empirical formulas.
With this approach, explains Hirano, drug developers can continue to use established LC separation methods with nonvolatile buffers (as accepted by regulatory bodies such as Japanese Pharmacopeia) in the first dimension, and then introduce an MS-friendly volatile buffer on injection of the fractionated impurity peaks onto the mass spectrometer in the second dimension.
Shimadzu’s 2D-LCMS-IT-TOF system: The pink dotted line region is the first dimension LC system, and the green dotted line region is the second dimension LC/MS system.
Sample-Prep Strategies
A key bottleneck in the LC/MS workflow for quantitative analysis of drug analytes has been sample preparation and the need to remove endogenous components of the sample matrix, such as proteins and lipids, which can interfere with MS detection and cause ion suppression.
Guifeng Jiang, Ph.D., applications manager, chromatography and mass spectrometry division at Thermo Fisher Scientific, describes an online sample-preparation method designed to be simpler and faster than traditional off-line techniques.
The sample-prep procedure requires only 1–2 minutes per sample and can be applied to a 10 µL plasma sample without the need for dilution or protein precipitation. The method is designed for laboratories performing routine bioanalysis using a single quadrupole mass spectrometer. It incorporates turbulent flow chromatography at a high flow rate to remove high molecular weight matrix components through the combined dynamics of diffusion and column chemistry.
Dr. Jiang demonstrates the use of this method with rat and human plasma matrices spiked with six drug standards at various concentrations. Plasma samples are loaded onto the turbulent flow chromatography column, with subsequent transfer of the cleaned-up sample directly onto the LC separation system, and automated elution of the fractions onto the mass spectrometer.
Mass detection on a single quadrupole MS system was performed in selected ion monitoring mode with constant positive/negative polarity switching. The entire process of online sample extraction, LC separation, and MS detection of the six drug compounds required about 10 minutes per sample.
Dr. Jiang presents chromatograms showing comparable results—in terms of peak size, shape, and position, and the absence of confounding peaks—when comparing samples in which the analytes were spiked into water or rat or human plasma. She reports no evidence of matrix-related ion suppression after sample prep, and presents calibration curves showing high linearity in detector response over analyte levels ranging from 12.5 to 12,500 ng/mL.
To minimize interference from endogenous matrix components and their effects on analyte ionization in MS analysis, Sigma-Aldrich developed a one-step sample-prep procedure that simultaneously removes proteins and phospholipids from biological matrices. The technology combines solid-phase extraction (SPE) for protein precipitation with a separation medium that is selective for phospholipids.
Craig Aurand, senior application chemist, describes a study designed to demonstrate the detrimental effect of phospholipid-induced matrix ionization suppression on LC/MS analysis of plasma samples. The study focused on analytical discrimination between propanolol and the 4-hydroxy-propanolol metabolite, and the comparative effects of a sample-prep method using either standard protein precipitation or a hybrid phospholipid extraction method.
For standard SPE, 100 µL plasma samples were combined with 1% formic acid acetonitrile in 96-well vacuum manifold plates that contained a zirconia-coated silica bed (for trapping the phospholipids) above a hydrophobic graded filter. Each well is topped with a Teflon frit. Application of a vacuum pulls the samples through the packed bed, achieving greater than 99% removal of phospholipids, according to Aurand.
Following the hybrid phospholipid extraction sample-prep technique, no phospholipids were observed in the matrix, resulting in a fourfold increased sensitivity for propanolol detection compared to standard protein precipitation. This improved sensitivity is “directly related to the phospholipid-induced ionization effect,” says Aurand.
Mike Chang, application development scientist at Agilent Technologies, points to lipids as being the major component of biological matrices that causes the greatest interference and ionization suppression in MS analysis. A common sample-prep strategy employed upstream of LC/MS is SPE, which mainly removes large proteins.
Chang describes the use of a hydroxylated SPE sorbent in an automated 96-well plate format that is intended to filter out the large proteins and minimize the interactions between the endogenous materials such as lipid and protein molecules in the matrix and the sorbent, thereby removing them from the sample and resulting in reduced ion suppression downstream.
Coating the SPE adsorbent with hydroxide residues instead of commonly used amides makes it less likely that the lipids in the matrix will react with the sorbent surface. Chang presents data showing reduced lipid content in samples treated with hydroxylated SPE and an associated decrease in ion suppression on MS analysis, with good accuracy and linearity in the calibration curves.

Thermo Fisher Scientific reports that its TurboFlow technology used together with LC/MS speeds up the quantitative analysis of drug analytes in biological fluid.
Optimization across the Workflow
“Sample prep is one of the most critical pieces,” says Erin Chambers, principal applications chemist at Waters, adding that the use of sample-prep methods designed to remove isobaric molecules can increase analyte detection on MS analysis and remove background interferences that can negatively impact the ability to achieve pg/mL detection limits.
“This is particularly true when the molecules being studied are synthetic or modified versions of substances that exist in the body,” she says.
For sample prep upstream of LC/MS, Chambers emphasizes the advantages of introducing orthogonal techniques. For example, if the LC separation is being done in a reverse-phase dimension, using sample-prep sorbents based on ion-exchange properties will yield improved selectivity.
A mixed-mode SPE sample-prep technique developed by Waters uses complementary SPE sorbents (a weak cation and a strong anion, for example) to enhance selectivity. This technique includes a final elution step designed to minimize peptide aggregation and adsorption. It is possible to achieve up to 15x concentration without evaporation using the mixed-mode SPE technique, according to Chambers.
Advances in LC have focused primarily on increasing sensitivity and resolution to improve detection limits and enhance the ability to resolve an analyte from the matrix background. Sub-2 micron LC, for example, can provide improved resolution and increased peak capacity, reducing the likelihood of co-elution and, thus, of matrix effects.
Another strategy described by Chambers is the selection of a chromatography medium with wider pores, which facilitates diffusion of large biomolecules, allowing for faster flow rates (without compromising peak shape) and reduced run times.
Selection of a triple quadrupole MS platform for quantification of large biomolecules such as peptides should focus on instruments with a broad enough mass range to accommodate high m/z multiply charged precursors.
Methods for improving MS sensitivity and analyte detection typically focus on the properties of the biological sample and may include strategies to optimize the solubility of peptides. This ensures that the peptides stay in solution throughout the analytical process, minimizes nonspecific binding, helps eliminate ghost peaks caused by carry-over, and improves the linearity of standard curves by eliminating drop-off at the low end, explains Chambers.
To learn about a new technique that leverages mass spec for the discovery of catalytic reactions, click here.