May 15, 2012 (Vol. 32, No. 10)
Small Molecules Are Easier to Make, but Biologics Offer Powerful Playing Field to Explore
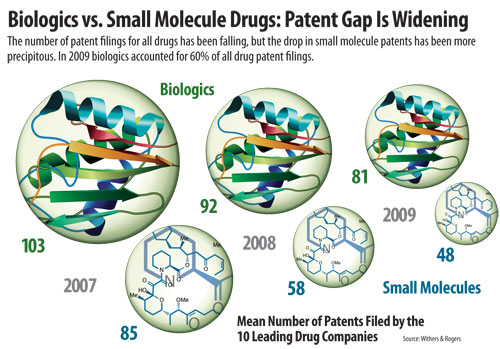
Two studies published in recent weeks along with a pair of studies released over the past two years showcase pharma’s drift toward biologic development and provide evidence of the promise of large molecule drugs. In the most recent report, the U.K. law firm Withers & Rogers found that by 2009, biologics accounted for 60% of the patents filed by the top 10 pharma companies. Abbott had as much as 80% of the patent filings between 2007 and 2009 focused on biologics.
Withers & Rogers noted that the gap between patent filings for biologics and small molecules among the top 10 pharma companies had grown in this timeframe. The number of biologic patent applications in ’09 increased by 14.5% from two years earlier, even though ’09 saw 31.5% fewer overall patent filings than ’07.
This positive trend extends beyond innovation to clinical development and final approval. Studies have come to the conclusion that biologics hold better prospects than traditional small molecules of advancing all the way from the lab to the clinic to the market.
Biologics vs. Small Molecule Drugs
Attrition Rates
Withers & Rogers issued its report almost a month after KMR Group, a biopharma industry consultancy, released findings of its latest Pharmaceutical Benchmarking Forum (PBF). Companies that submitted data for this analysis were Abbott, AstraZeneca, Bayer, Boehringer Ingelheim, Bristol-Myers Squibb, Eli Lilly, GlaxoSmithKline, Johnson & Johnson, Merck Research Labs, Novartis, Pfizer, Roche, and Sanofi.
KMR found that more than 25% of large molecules in Phase II between 2006 and 2010 reached the market compared to about 10% of small molecules. “One of the reasons that small molecules have fared worse is because most of these companies were already very heavily invested in small molecule work. All the low-hanging fruit was found in the last 10 to 15 years,” explains Scott Martin, a consultant with KMR. “Now, they’re turning toward new technologies, the biotech technologies, and therefore, you’re kind of going in a new area, finding what works.”
KMR reports that 12% of biologics were calculated to advance from preclinical to clinical studies, compared with just 2% of small molecule drugs. The biologics percentage clearing each successive clinical review hurdle grows to 17% at Phase I, 27% at Phase II, leaping to 58% at Phase III, and 82% at the registration phase. For small molecule drugs, according to KMR, success was calculated to be 4% at Phase I, 9% at Phase II, 44% at Phase III, and 78% at registration.
The KMR study did, however, find that industry’s large molecule success rates in development phases have declined in recent years. Martin thus adds a cautionary note: “We’re finding it harder as an industry—I’m talking about the broad R&D industry—to get large molecules to approval, even though they do have a higher success rate all the way up the development pipeline. If you look in three years and you ask, ‘Has the small and large molecule success rate started to even out or not?’ if my view is correct, you’re going to see that these large molecule rates are going to come down.”
In looking beyond success in clinical studies to eventual approval, the Tufts Center for the Study of Drug Development found that large molecules outpaced small molecules 32% to 13% between 1993 and 2004. The study covered the top 50 pharma companies and was published during 2010 in Nature Clinical Pharmacology & Therapeutics. The discrepancy may, however, reflect the small share (15%) of large molecule drugs in the sample of 1,738 new drugs (lead indications). For all molecules, large and small, the approval success rate for the entire study period was 19%.
A few months after the Tufts study was released, a study by the Biotechnology Industry Organization (BIO) and BioMedTracker looked at the success of 4,275 drugs with a total 7,300 indications from 2003 to 2010. Overall success was just 9%–14.5% for lead indications and 3.2% for secondary indications. Most of the drugs studied, though, were small molecule drugs, notes David Thomas, BIO’s director, industry research and analysis.
Overall success rate for lead indications varied from 14% for NMEs to 26% for biologics and 41% for non-NMEs. For secondary indications, success ranged from 3% for NMEs to 7% for biologics to 10% for non-NMEs.
Reasons for Differences
Estimated phase transition probabilities and eventual approval success rates differed significantly by therapeutic class in Tufts’ study. For self-originated drugs, systemic anti-infective drugs enjoyed the greatest approval rate (23.9%), followed by musculoskeletal (20.4%), antineoplastic/immunologic (19.4%), respiratory (9.9%), GI/metabolism (9.4%), cardiovascular (8.7%), and CNS drugs lowest (8.2%).
“The distribution of disease categories does differ between large and small molecules. In particular, large molecule development is more concentrated in oncology and immunology, while small molecule development is more concentrated on cardiovascular and CNS conditions,” says Joseph A. DiMasi, Ph.D., director of economic analysis for Tufts CSDD. “However, differences in therapeutic class focus do not appear to explain the differences in success rates.”
What reasons, known or suspected, can explain that difference? “This is a difficult question to answer definitively, but it may be that large molecule development, in general, is more targeted, and, given their therapeutic focus and functions (e.g., immunologics and replacement therapies), safety issues may be less prevalent,” Dr. DiMasi speculates.
BIO’s Thomas also points out an interesting result of the BIO/BioMedTracker study: Oncology drugs had the lowest success rate at 5.6%, meaning just one in 20 cancer drugs advanced from Phase I all the way to approval. That rate combines the 11% success rate of lead cancer indications and 2% success rate of secondary indications.
Thomas shared a possible reason for the success-rate differences between diseases: With infectious disease, it’s easy to take a blood sample and quickly generate a readout that tells whether or not your drug is working. In oncology and cardiology, developing a drug is more difficult given the outcome studies required, with multiple efficacy endpoints, not to mention the complexity of the diseases; a cancer drug may work against several different enzymes, versus infectious disease, where a simpler drug targeting a single virus may suffice.
Thinking Large
As more biologic drug candidates are discovered and undergo R&D and review, it will be interesting to see how much the gap with small molecule drugs in terms of attrition rates changes. While some narrowing can be expected, the studies to date have all found more biologics advancing through reviews than NMEs.
No wonder then that big pharma is increasingly thinking large, as in molecule, as it scrambles to squeeze more success from the billions it is spending on R&D. Withers & Rogers’ study in particular bolsters the paradigm that big pharma is shifting its development focus from small molecule chemicals to large molecule biologics in recent years.
Among the top 10 pharma firms it surveyed, Novartis had the most patent applications relating to biological drugs published in 2009, with 260—more than twice as many patents for biological drugs compared to small molecules—followed by Johnson & Johnson and Merck & Co. AstraZeneca ranked lowest with just 15 biologicals patents, or 22% of its total. AstraZeneca and Pfizer had less than half of its patents sought for biological in 2009.
There are, of course, far more small molecule drugs on the market. “It is considerably easier to develop and manufacture small molecule drugs. They come with lower R&D costs and there is an established market infrastructure for them. Therefore, the shift to biologics could result in fewer new products making it to market,” Nicholas Jones, Ph.D., partner at Withers & Rogers, explains.
Future studies should examine more closely the type of biologics most apt to achieve regulatory approval—a topic touched on by the Tufts study. KMR’s Martin says that the firm plans to issue another PBF analysis next winter. Research will have to dig deeper to spell out just which type of biologic works best on what disease.



