January 1, 2011 (Vol. 31, No. 1)
2010’s Success Rate Can Be Chalked Up to the Focus on Cutting-Edge Technologies
During 2010, 73% of major clinical and regulatory outcomes were positive. For 2011 we expect a similar figure of 75%—underscoring a robust success rate. We have focused mainly on innovative programs (vs. formulations or me-too drugs) and are encouraged to note the biopharma industry’s progress from increasingly harnessing the fruits of “new science”.
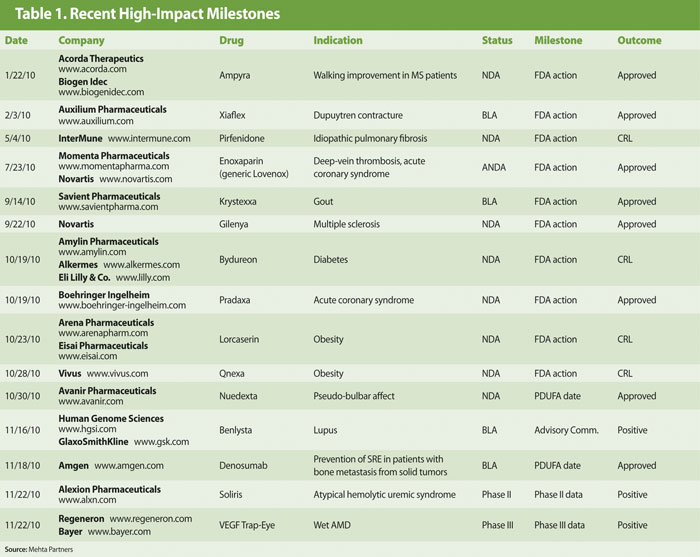
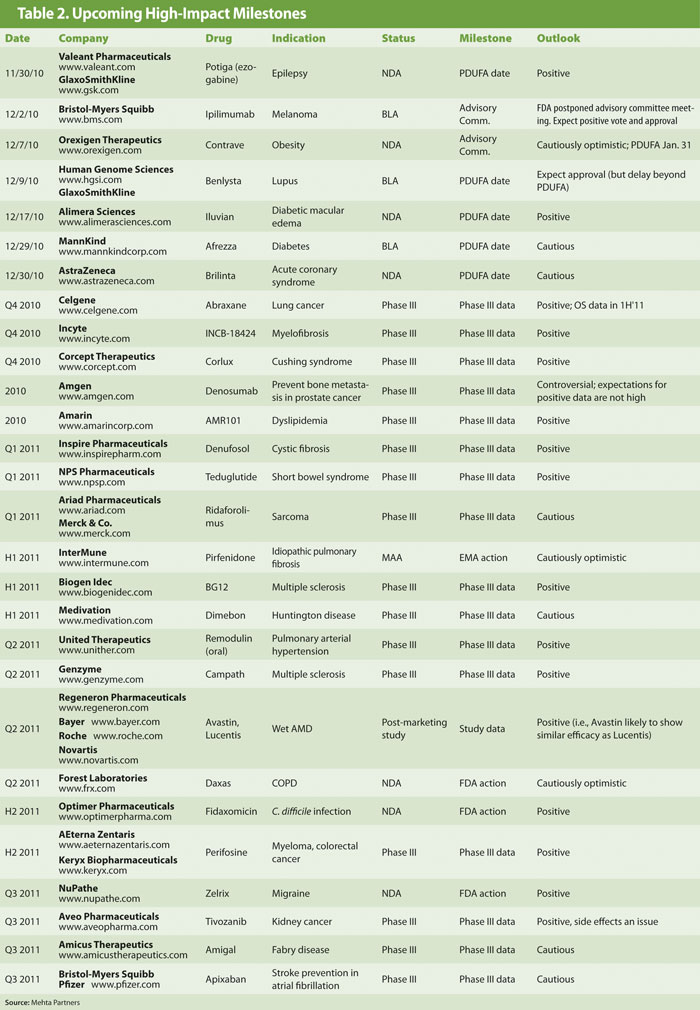
Milestones in Tables 1 and 2 have been selected based on their potential to have a high impact on company valuation and/or an industry-wide impact through addressing high unmet medical needs. Accordingly, we have focused on results of late-stage clinical trials and key regulatory actions.
Of the 15 high-impact events in 2010 (through November 22), 11 were positive with only 4 negative outcomes. Importantly, of the negative developments, three of the four still have a decent chance of eventually becoming successful.
One of the most highly anticipated events earlier in the year was the FDA decision on Intermune’s Pirfenidone for idiopathic pulmonary fibrosis. Despite a positive 9–3 advisory panel vote in March, the FDA issued a Complete Response Letter (CRL) requesting an additional clinical study to support the efficacy claim for Pirfenidone. While the path forward for potential U.S. approval is far from clear, Intermune also has submitted a Marketing Authorization Application to the European Medicines Agency, and there remains some hope that the drug could be approved in Europe—helped by additional data from Japanese studies by Shionogi (www.shionogi.co.jp). This decision is expected in the first half of 2011.
In the popular obesity area, two closely watched drugs were also met with CRLs from the FDA. In case of Arena Pharmaceuticals (www.arenapharm.com) and Eisai’s (www.eisai.com) Lorcaserin, FDA had serious issues with both safety and marginal efficacy, and the fate of the drug seems pretty much sealed for now.
Vivus’ Qnexa, on the other hand, still has hope as its strong efficacy was never in question and many of the safety concerns raised by the FDA and advisory panel have been subsequently addressed with data from an extension study. We expect Qnexa to be ultimately approved in the second half of 2011.
Bydureon for diabetes from Amylin—and partners Alkermes and Eli Lilly—was expected to be a shoo-in at the FDA, and many were surprised by the setback. The main issue involves the potential for QT prolongation, which the companies were remiss in not having fully addressed prior to NDA submission. Previous studies with Bydureon (and exenatide—the underlying active drug) have not shown any significant association with QT prolongation. We expect Amylin to complete a thorough QT study and refile by the end of 2011, with approval expected in mid-2012.
Of the positive developments in 2010, the following are most notable:
- Approval of enoxaparin, a generic Lovenox from Momenta Pharmaceuticals and Novartis which begins to set forth a pathway for future biosimilar approvals,
- Approval of Novartis’ Gilenya, the first oral drug for MS,
- Positive panel recommendation for Human Genome Sciences and partner GlaxoSmithKline’s Benlysta, a potential new therapy for lupus,
- Amgen’s much-needed pipeline success with Xgeva (denosumab) for fracture prevention.
High-Impact Milestones of 2011
Of the 25 high-impact clinical and regulatory events in 2011, we expect positive outcomes for 16, are cautiously optimistic on 3, and cautious or negative on 6.
We highlight below several small-cap companies with transformative events that stand to significantly impact their valuations.
The obesity story continues with Orexigen Therapeutics and its lead drug Contrave. Like Vivus’ Qnexa, Contrave demonstrated good efficacy (although not as strong as Qnexa), but is likely to face scrutiny on the safety front. An advisory panel was scheduled for December 7 with a PDUFA date of January 31, 2011.
As the last of the obesity players going into an advisory panel, Orexigen has had some time to prepare its defense on the key side effect observations of increased blood pressure and seizure risk (which are well known to be associated with bupropion, one of the approved and widely used components of Contrave). We remain cautiously optimistic.
Amarin, unlike many small biotechs, has chosen to operate in the pharma mode of pursuing cardiovascular blockbusters. The company is conducting two Phase III trials under special protocol assessment in patients with high triglyceride levels. AMR-101 (eicosapentaenoic acid) is reportedly a purer and potentially more potent form of Omega-3 fatty acids including GSK’s Lovaza. The first of the two trials is expected to report results soon. Therapeutic benefits of the Omega-3 class have been well validated by Lovaza (which has sales of over $1 billion).
Inspire Pharmaceuticals will report results of a confirmatory Phase III trial of denufosol in cystic fibrosis in early 2011. Denufosol is a P2Y2 ion-channel regulator with disease-modifying potential. With positive data from a previous study and a more robust subsequent trial design (larger study with a longer duration of therapy, tighter enrollment criteria), we ascribe a high likelihood of success.
NPS Pharmaceuticals is developing Gattex (or teduglutide) for short bowel syndrome and will report Phase III results in early 2011. As with Inspire’s denufosol, this is a second Phase III trial, which builds upon the design and results obtained from the first study—meaningfully enhancing the chances for a successful outcome. A prior trial showed that the lower dose of teduglutide was associated with a significant reduction in need for parenteral nutrition, the primary endpoint.
NPS’ second program, NPSP558 (recombinant parathyroid hormone), also has good chance for success as a hormone-replacement therapy in patients with hypoparathyroidism. An ongoing Phase III trial will report final data by year-end 2011.
The antibiotic space has had its regulatory troubles in the past partly due to shifting goalposts at the FDA. Recently, however, the landscape has improved in this regard, providing the opportunity for safe and efficacious new antibiotics studied in well-designed trials to gain approval. Optimer Pharmaceuticals is in a strong position with two positive trials of fidaxomicin in C. difficile infections. Fidaxomicin has demonstrated high clinical cure rates and superiority over the leading antibiotic, vancomycin, in recurrence, bolstering its chance for approval in the second half of 2011.
High-profile programs from larger companies facing critical milestones in 2011 include:
- Approval of Bristol-Myers Squibb’s (www.bms.com) ipilimumab for melanoma (an advisory panel scheduled for December 2 was postponed; PDUFA date is in March 2011),
- AstraZeneca’s potential blockbuster Brilinta in cardiovascular setting with a PDUFA date on December 16 (outcome remains controversial due to a weaker dataset in the U.S. population),
- Biotech investors will be closely watching for Phase III data for Celgene’s Abraxane in lung cancer, expected before year end. Celgene acquired Abraxis Bioscience in June of this year, and this will be the first important dataset toward justifying the $2.9 billion acquisition cost.
The resilient Biogen Idec, which has investors concerned over the fate of Tysabri due to increasing PML cases and now the launch of Novartis’ Gilenya as the first oral drug for MS, will unveil the results of its own oral MS compound BG12 in the first half of 2011. It is the potential of BG12 in the mid term and Biogen’s hemophilia programs in the longer term (in addition to the M&A possibility) that continues to attract investors with longer-term investment horizons.
Last but not least, the CATT study comparing Avastin and Lucentis in a head-to-head trial in wet AMD is expected to report data in the second quarter of 2011. With implications for multiple companies—in addition to Roche other players are Novartis, Bayer, and Regeneron—most healthcare investors will be watching closely for the data.
Sushant Kumar, Ph.D. ([email protected]), is partner at Mehta Partners. Web: www.mpglobal.com. Mehta Partners and/or its affiliates may hold a position in any security or financial instrument mentioned herein.



