August 1, 2011 (Vol. 31, No. 14)
Option Enables the Assessment of Bioprocesses in Early Development
While industrial bioprocesses have been operated in fed-batch mode for a long time, the screening of high-producer cell lines and a lot of other development work is still performed in batch mode for a variety of reasons. One downside to fed-batch fermentation is that it requires more instrumentation, e.g., pumps, to realize the substrate feeding.
Another factor that favors batch mode in small scale is that the huge number of clones in screening projects requires researchers to utilize parallel and easy-to-handle cultivation vessels, including shake flasks, which have known limitations.
m2p-labs recently developed a ready-to-use medium to address these shortcomings. The Feed-in-Time (FIT) media series offers enzyme-assisted glucose release to enable fed-batch mode in any fermentation scale. Fed-batch medium is especially useful for small-scale fermentations where simple and easy-to-use bioreactors are preferred. Currently, FIT fed-batch medium is available in synthetic and complex formulations for several cell types such as E. coli, Pichia pastoris, and Hansenula polymorpha.
When performing an aerobic fermentation, the maximum substrate feeding rate must always be adjusted to the maximum oxygen-transfer capacity of the bioreactor, otherwise the culture cannot process the substrate over the aerobic pathway and thus, inevitably, the cells produce anaerobic byproducts such as organic acids.
Shake flasks are generally known as bioreactors with low oxygen transfer capacities. Baffled shake flasks typically provide higher oxygen-transfer capacities but often also exhibit higher heterogeneity of results. Since the baffles are introduced manually into classical Erlenmeyer flasks by glassblowers, these baffled shake flasks are unique and reproducibility is missing.
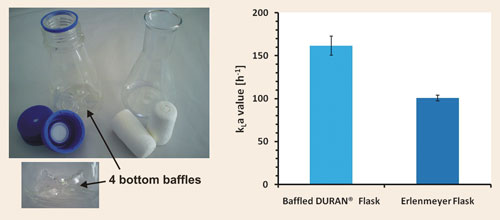
Duran® solved this problem by designing an automated, two-step industrial production process for a new, reproducible baffled shake flask. The baffled Duran flask possesses reproducible geometry with four bottom baffles, which ensures reproducible behavior in respect to oxygen-transfer capacity and culture growth.
Oxygen-Transfer Capacities
We recently undertook a study to determine oxygen-transfer capacities in baffled and classical shake flasks. The oxygen-transfer capacity was determined by applying the sulfite oxidation method.
The basic conditions for determining the oxygen-transfer capacity, here expressed as kLa values, were as follows: 250 mL nominal volume of the shake flasks, 15 mL filling volume, 25 mm shaking diameter, 300 rpm shaking frequency, temperature 28eC, membrane cap for Duran baffled flask, and pulp plug for Erlenmeyer flask.
Under these conditions a kLa value of 160 h-1 ± 7% for the Duran baffled flask and a kLa value of 100 h-1 ± 3% for the Erlenmeyer flask were determined (Figure 1). We were able to show that the Duran baffled flask exceeds the classical Duran Erlenmeyer flask in oxygentransfer capacity by more than 50%, enabling a better aeration for aerobic cultures.
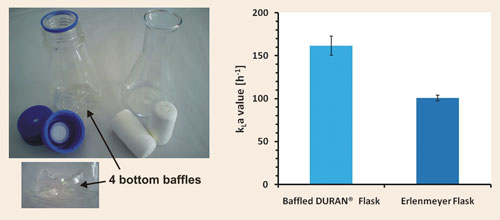
Figure 1. The baffled Duran flask and classical Erlenmeyer flask (left) and the corresponding kLa values determined by the sulfite oxidation method (right).
Functional Enzyme Expression in E.coli
We then evaluated functional enzyme expression in E. coli (enzyme provided by Evocatal). Expression in rich TB media and synthetic WR media containing 20 g/L glucose revealed that cultivation in Duran baffled flasks was better than that in classical Duran Erlenmeyer flasks. In the latter ones the lower oxygen-transfer capacity led to a higher formation of acidic byproducts.
In synthetic WR media, the pH value decreased very fast below a pH of 5, as a result cell growth was inhibited. With Duran baffled flasks, higher cell densities were obtained because of slower and reduced acidification in the culture broth. The metabolization of complex compounds in rich TB media results in rising pH value during cultivation subsequent to consumption of glycerol as the main carbon source. In classical Duran Erlenmeyer flasks a higher rate of acidic byproduct formation can be observed by a slower rate of pH increase.
The better oxygen-transfer conditions exhibited with Duran baffled flasks can also be observed in higher expression of the target enzyme. Activities in WR medium differ by one order of magnitude because cells cultivated in Erlenmeyer flasks are faced with an acidic surrounding, which prevents growth and significant product formation.
Further study was conducted by using FIT fed-batch media (synthetic and complex formulation) both in Duran baffled flasks and Duran Erlenmeyer flasks.
The maximum applicable glucose feeding rate is dependent on the oxygen-transfer capacity of the bioreactor vessel. With this in mind, two concentrations of glucose-releasing enzyme were investigated. As shown in Figure 2, both glucose feeding rates could be realized in Duran baffled flasks. For the higher glucose release rate (1.5% enzyme) in the classical Erlenmeyer flasks, however, the culture was not able to consume the total fed glucose due to limited oxygen in these flasks.
As a result, glucose accumulated in the broth, and cells switched to an overflow metabolism and started to produce acidic byproducts. As a consequence the pH value quickly dropped to a growth-inhibiting value of 5. In addition, productivity was dramatically decreased under these conditions.
In Duran baffled flasks the cells experienced a higher oxygen supply and, therefore, they were able to consume the fed glucose at the same rate as it was released for the higher release rate with 1.5% enzyme. Consequently, this led to faster generation of biomass (=50 OD600 after 24 hours) and higher target enzyme expression.
When comparing enzyme expression in batch and fed-batch mode, the target enzyme exhibited greater activity in fed-batch fermentation, and the proportion of active product to inactive product was improved. This means that the growth-limiting glucose feeding in FIT fed-batch medium improves the overall product formation and also produces more active protein in the soluble fraction.
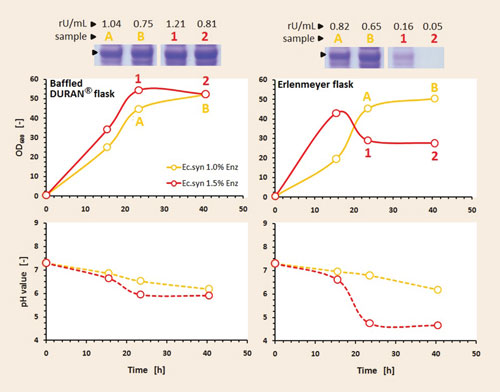
Figure 2. Progression of biomass, pH, and expression of target enzyme in fed-batch mode on synthetic FIT medium in the two studied shake flasks and two glucose release rates: Induction was performed with 100 µM IPTG after 15.5 hrs; activities determined with a specific enzyme activity assay; SDS-PAGE prepared under reducing conditions.
The addition of complex components to the culture medium is normally advantageous for protein expression and cell growth, hence the supplementation of peptones to the FIT fed-batch medium was also studied. A plant peptone was added to the medium at the beginning of the fermentation and also after induction. The results of this experiment in complex FIT fed-batch medium and the influence of the applied shake flask geometry is presented in Figure 3. Again here, two glucose release rates were tested.
The results demonstrated the superiority of a Duran baffled flask over a classical Erlenmeyer flask. Biomass concentrations in the range of OD600 50 could be reached in the Duran baffled flask and very high enzyme expression levels could be found in SDS-PAGE. The Erlenmeyer flask led only to biomass concentrations between 30 and 40. The achieved enzyme expression for the lower glucose release rate (1.0% enzyme) is still remarkable, but the enzyme expression for the higher glucose release rate (1.5% enzyme) is disappointing.
The cells could not consume the total released glucose with the higher feeding rate (1.5% enzyme) due to the limited oxygen supply in the Erlenmeyer flask. Therefore, the cells produced organic acids and the pH value dropped rapidly below pH 5. Under these conditions the E. coli cells were no longer able to produce high levels of enzyme.
The addition of plant peptone to the FIT fed-batch medium showed that enzyme expression can be improved dramatically, which can be seen in SDS-PAGE. The measured enzyme activities confirmed these findings by an increase of 400% in activity (Figure 3 versus Figure 2).
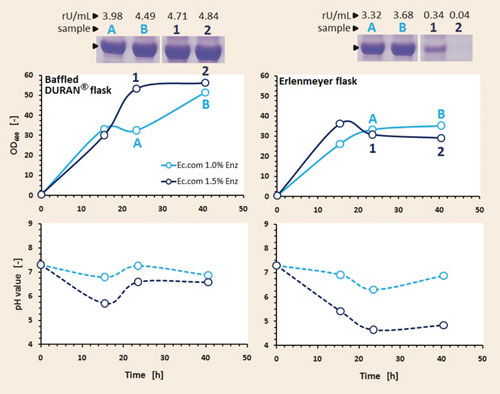
Figure 3. Progression of biomass, pH, and expression of target enzyme in fed-batch mode on complex FIT medium in the two studied shake flasks and two glucose release rates: Induction was performed with 100 µM IPTG after 15.5 hrs and a high concentrated peptone solution was added (10% v/v of cultivation volume) at the same time; activities determined with a specific enzyme activity assay; SDS-PAGE prepared under reducing conditions.
Conclusions
The results of these studies demonstrated clearly that fed-batch mode is superior to a simple batch mode. Addition of a plant peptone to synthetic fed-batch medium improved the enzyme expression and activity. Use of the baffled shake flask from Duran showed that higher kLa values could be measured and that better and more reproducible results in microbial batch and fed-batch fermentations could be achieved as compared to the classical Erlenmeyer flask.
The combination of FIT fed-batch media and Duran baffled shake flasks now allows the study of microbial fed-batch fermentations with a broad range of different linear feeding rates without running into strong oxygen limitations. In conclusion, the efforts for bioprocess development in stirred tank bioreactors can be reduced and more valuable studies can the transferred to the smaller scale. The presented new shake flask and fed-batch medium enables bioprocessors to generate more knowledge at the early stage and thus, promise to speed up bioprocess development in the near future.
Johannes Hemmerich and Pamela Wenk are application scientists at m2p-labs. Tim Lütkepohl is product manager labware at Duran Group. Frank Kensy ([email protected]) is managing director of m2p-labs.