December 1, 2012 (Vol. 32, No. 21)
A term originally coined by Conrad Hal Waddington in the 1940s to describe the causal relationship between genotypes and phenotypes, epigenetics has witnessed profound transformations as a concept.
The increasing appreciation of the roles that epigenetic changes play in a growing number of medical conditions opened the attractive possibility to therapeutically modulate them, and the approval of the first four epigenetic compounds set the stage for a new era that promises to reshape the therapeutic arena.
“We are pursuing drug discovery in a variety of areas relevant to chromatin structure and function, and this is the key feature that allowed us to establish our industry-leading programs in these areas,” says Jim Audia, Ph.D., CSO at Constellation Pharmaceuticals.
The most advanced of the epigenetic drug discovery efforts at Constellation, the BET (bromodomain and extra-terminal) program, focuses on small molecule inhibitors of the BET family proteins. In mammals, the BET family includes the ubiquitously expressed Brd2, Brd3, and Brd4, and the testis-specific isoform BRDT, which function to facilitate gene activation.
Each of these proteins has two N-terminal bromodomains, which harbor a deep, hydrophobic pocket that recognizes specific acetylated lysine residues on histone tails. “Their very well-formed pocket allows us to use structure-based drug design to find binding sites for small molecule inhibitors,” says Dr. Audia.
While pursuing this work, other researchers discovered that BET proteins selectively regulate the NF-κB pathway in inflammation, prompting Constellation to pursue this mechanism in oncology due to the importance of NF-κB-dependent transcriptional signaling in many malignant tumors.
One of the surprising findings was that BET proteins, particularly Brd2 and Brd4, localize to the transcriptional start sites of the myc oncoprotein, downregulating its expression in a variety of cellular contexts. The possibility to directly displace them by small molecule inhibitors promises opportunities to rapidly and potently suppress myc transcription in an oncology setting.
“We found a relatively broad subset of hematological malignancies that appear to be very sensitive to bromodomain inhibitors, and we are now optimizing our initial prototypic targets to ultimately make them become clinically relevant molecules,” explains Dr. Audia. This work, in addition, promises to elucidate in more detail the biology of the proteins involved in chromatin remodeling.
“We are learning how context-dependent everything is, how subtly different the effects of some of these targets may be depending on the cell type and on the state of the cell. One of our goals, that we think is both possible and advantageous, and perhaps even necessary, is to understand the factors that control this chromatin-based signaling,” reveals Dr. Audia.
Constellation recently reported its collaboration with the Leukemia & Lymphoma Foundation to support preclinical and Phase I programs that explore the therapeutic potential of BET bromodomain inhibitors in hematological malignancies.
“In addition, we have an exclusive collaboration with Genentech, a broad effort that is being driven by the belief that a number of targets across various methyltransferases, demethylases, and bromodomain readers offer the potential for significant therapeutic applications,” says Keith Dionne, Ph.D., president and CEO at Constellation.
The collaboration is not restricted to therapeutic targets in malignant tumors. This reflects the vast body of experimental findings that underscore the key roles that epigenetic modifications play in medical conditions outside the sphere of oncology such as inflammatory, neurodegenerative, and cardiovascular diseases.
“Rapid progress in these areas is driven by a partnership between chromatin biology and chemistry, which work together to understand the specificity of the interaction from the chemistry side, and the therapeutic impact from the biology side,” explains Dr. Dionne.

Structure-based design used to map the entirety of the BET binding pocket and design best-in-class BET inhibitors. [Constellation Pharmaceuticals]
Inhibiting EZH2
“GlaxoSmithKline plans to take EZH2 inhibitors into the clinic in cancer,” says Peter J. Tummino, Ph.D., head of biology, cancer epigenetics discovery performance unit at GlaxoSmithKline (GSK). EZH2, the catalytic component of the polycomb repressive complex 2 (PRC2), represses gene transcription by methylating lysine 27 residues on the histone H3 tails. EZH2 overexpression has been reported in several types of solid tumors while, in contrast, mainly EZH2 point mutations were described in lymphomas.
“We found that these heterozygous mutations are biochemically neomorphic, an aspect that was not obvious when the mutations were discovered,” explains Dr. Tummino.
The presence of these gain-of-function EZH2 mutations pointed toward a desire to therapeutically inhibit the activity of the protein. Previously, scientists at GSK have developed several new reagents that helped conduct two EZH2 high-throughput screens. “We have been able to develop potent and selective small molecule EZH2 inhibitors that inhibit histone H3 lysine 27 trimethylation and possess potent antiproliferative activity specifically in cells with EZH2 activating mutations,” says Dr. Tummino.
Most recently, scientists at GSK described GSK126, a small molecule EZH2 inhibitor that can reactivate silenced PRC2 target genes. GSK126 inhibited the proliferation of diffuse large B-cell lymphoma cell lines harboring a heterozygous Y641 gain-of-function mutation, which is the most frequently encountered EZH2 mutation in this group of malignancies, and inhibited the growth of EZH2 mutant diffuse large B-cell lymphoma mouse xenografts.
Achieving Specificity
“Our approach differs from the one used by many other epigenetic companies, in that we specifically target individual genes to upregulate,” says Arthur Krieg, M.D., president and CEO at RaNA Therapeutics. Investigators at RaNA achieve this specificity by targeting individual noncoding RNA molecules that bind PRC2 to repress the expression of individual genes.
“Oligonucleotide therapeutics are particularly well suited for this, because we can take advantage of the Watson-Crick base-pairing to specifically target individual genes, and with the knowledge coming out on noncoding RNAs, we can now accomplish this in a highly selective fashion,” notes Dr. Krieg.
One of the target genes that Dr. Krieg and colleagues used to illustrate this proof of concept is erythropoietin. “By using an oligonucleotide to target a noncoding RNA, we were able to prevent PRC2 recruitment to the erythropoietin locus, derepress gene expression, and upregulate erythropoietin expression in vitro and in vivo,” explains Dr. Krieg. Investigators at RaNA are planning to use this approach as they proceed toward clinical development with several therapeutic targets.
“We live in the next frontier in genetics and genomics, and one of the big advances has been the understanding that epigenetics is involved, in an ongoing basis, in regulating the expression of most genes in the genome in a highly selective and tissue-specific manner,” explains Dr. Krieg.
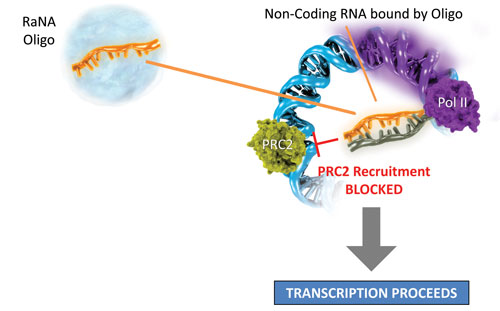
According to RaNA Therapeutics, its oligos interrupt PRC2 recruitment.
Mind Those Histones
“At 4SC, our focus is on the interplay between different modifications that occur on histones,” says Bernd Hentsch, Ph.D., CDO at 4SC. Recent years witnessed two major advancements relevant to histone post-translational modifications. In addition to unveiling the contribution of histone deacetylases to regulate additional key players in tumorigenesis, such as p53 and Hsp90, increasing numbers of studies unveiled the complex interplay that exists among different types of histone post-translational modifications.
“It is therefore important to examine not only direct changes, but the activity of a whole group of modulators of the histone code, and methylation will probably represent the next wave of scientific interest,” says Dr. Hentsch.
Investigators at 4SC recently revealed a clinical setting where modulating histone acetylation could translate into new and much needed therapeutic applications. One of the acute challenges in oncology is the resistance to therapy that often develops, through a process that involves mutations and, therefore, is not reversible. However, approximately two years ago, an early stage during this process was described and became known as drug tolerance.
“Tolerance, in contrast to resistance, occurs by epigenetic changes, and it can be reversed when cells are treated with histone deacetylatse or histone demethylase inhibitors,” explains Dr. Hentsch.
A strategy developed by 4SC scientists is to combine a histone deacetylase inhibitor with an existing therapeutic compound to which the tumor cells are sensitized or re-sensitized.
In a Phase II 4SC-sponsored Shelter clinical trial for hepatocellular carcinoma that enrolled patients after they had developed tumor resistance to sorafenib, the only first-line treatment available, clinicians co-administered the oral pan-histone deacetylase inhibitor resminostat in addition to sorafenib, and reported an overall median eight-month survival—the longest, to date, among second-line therapies for patients with comparable characteristics.
“This puts into perspective the fact that epigenetic alterations can revert drug tolerance to a drug sensitive state, where pre-existing regimens can work again,” notes Dr. Hentsch.
Another epigenetic drug that scientists at 4SC are developing, 4SC-202, is currently in Phase I development. “We have shown that 4SC-202 acts on a broad number of Wnt signaling target molecules, and dysregulation in this pathway is important in a number of malignancies,” says Daniel Vitt, Ph.D., CSO. Additionally, 4SC-202 inhibits protein deacetylation and arrests the cell cycle in the G2/M phase.
“Resminostat and 4SC-202 interfere with truly important oncogenic characteristics, and it is important to appreciate that even though both are inhibitors of histone deacetylases, they consist of very different chemistries and comprise individual and thus differentiating biological properties. We will, therefore, be able to position these drugs individually in different selected cancers and in different populations,” notes Dr. Hentsch.
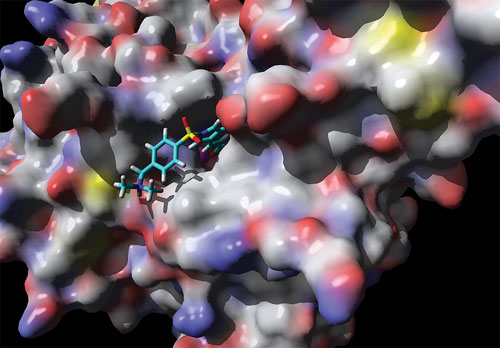
Resminostat (the small chemical molecule illustrated by its molecule structure) is shown binding to an HDAC/histone deacetylase enzyme. DNA expression is regulated by acetylation and de-acetylation. Thus, inhibition of an HDAC enzyme (e.g., through resminostat) will result in a more “open” and more “active” state of the histones and the chromatine structure, which will allow better transcription, according to 4SC.
Colon and Lung Cancer Tests
“The elegance of epigenetic measurements is that they are incredibly simple and very deployable on platforms commonly used in clinical labs and can be performed on blood specimens,” says Noel Doheny, CEO at Epigenomics.
Epigenomics has two products, Epi proColon and Epi proLung, available in several European countries, though neither of which is currently available in the U.S. or Canada. Investigators at Epigenomics are in the process of conducting the required clinical trials and seeking regulatory approval in these markets.
Epi proColon, a noninvasive plasma-based test, relies on detecting septin 9 DNA hypermethylation, a sensitive marker for colorectal cancer. “For this product, the pre-market approval submission in the U.S. is in its later stages,” reveals Doheny. In the U.S., Epigenomics has licensed its septin 9 methylation technology to Quest Diagnostics, ARUP Laboratories, and Companion Dx. “There are tens of thousands of assays that have already been done by these laboratories on patients in the U.S., and the utilization is very encouraging,” says Doheny.
A recent study revealed that this marker shows comparative sensitivities for detecting tumors on the right and left sides of the colon, for which colonoscopy detection rates differ, particularly in the early stages. The test does not require stool samples or invasive procedures. The firm believes that the availability of this test would encourage and facilitate early detection, at a stage when tumors are easier to monitor, treat, and cure.
The second test, Epi proLung, which is based on the detection of the methylated SHOX2 gene and at this time is also marketed exclusively in Europe, addresses one of the challenging aspects of lung cancer—the indeterminate results that are often obtained for cytology specimens. In combination with cytology, the Epi proLung assay can provide an immediate answer, which is crucial during the management of this condition, given the urgency for early therapeutic intervention. Early data has shown that this method may be applied in the future to the blood-based screening of lung cancer.
Networking and Outsourcing
“We are a virtual business and, as such, all our experimental research is outsourced to external labs,” explains Anthony Brown, Ph.D., scientific director at CellCentric.
The main therapeutic area that CellCentric focuses on is cancer. Through a combination of option agreements and consultancies, the company has established contractual relationships with a discovery network comprising over 30 academic laboratories across the world working in the epigenetics field.
Through its access to unpublished and privileged research information from the participating labs, CellCentric has prioritized over 50 unexplored and underexplored epigenetic targets. It is devoting significant efforts on second-generation enzymes, including developing small molecules that target histone-modifying enzymes. “But we have targets all across the epigenetic space; it is the clinical rationale and opportunity that drives our target selection,” notes Dr. Brown.
Compounds at various stages of preclinical and clinical development illustrate the promises of recent advances in epigenetics research, and reflect efforts to exploit distinct epigenetic mechanisms. These strides are catalyzing the shift toward newer generations of targets.
As many epigenetic modifications occur early during disease pathogenesis, their modulation is anticipated to provide, in addition to therapeutic compounds, predictive biomarkers and opportunities for prophylactic interventions, forecasting new perspectives for pharmacology and clinical medicine.
Epigenetics Renaissance
“While the field has been around for a while, recent years have seen a renaissance of epigenetics, partly because we realized that genetics cannot explain everything, and also thanks to the remarkable technological advances that enable us to profile and analyze epigenetic changes in a genome-wide and high-throughput setting,” says Zdenko Herceg, Ph.D., group leader and head of the section of mechanisms or carcinogenesis at the International Agency for Research on Cancer.
Recent years unveiled the importance of epigenetic modifications in shaping the pathogenesis not only of cancer, but that of other diseases as well, and helped advance ideas for developing more efficient preventive and therapeutic strategies. “The main challenge is to identify the drivers of the functionally important changes and distinguish them from the nonfunctional, passenger ones,” says Dr. Herceg.
The therapeutic use of epigenetic markers is an area that has witnessed increasing attention, and it is encouraged by the reversibility of epigenetic modifications and by many pieces of evidence pointing to their early occurrence during disease pathogenesis. “The technological revolution in genomics and epigenomics now allows the development of cost-effective and sensitive assays to detect and measure epigenetic markers and their changes in body fluids,” explains Dr. Herceg.



