February 1, 2010 (Vol. 30, No. 3)
Whether Model Is Apt Seems to Depend on Type of Cancer and Individual Patient
The cancer stem cell theory, which holds that a small number of tumorigenic cells are responsible for metastases, applies only to some cancers and possibly to certain patients, according to Sean Morrison, founder of OncoMed, now at the University of Michigan Center for Stem Cell Biologics. Therefore, learning when the cancer stem cell model is valid is a vital step to understanding and, ultimately, treating cancer.
“Not every cancer cell is bad. Some cancer cells differentiate into cells that are benign,” Dr. Morrison emphasized as he opened the cancer stem cell workshop at the American Society for Cell Biology (ASCB) meeting in San Diego in December. “Cancer stem cells have the unique capability of proliferating.”
The stochastic, or clonal evolution, model dictates that, “despite the heterogeneity (in cancer), cells of many different phenotypes are capable of proliferating extensively and forming new tumors,” Dr. Morrison summarized.
The cancer stem cell model, in contrast, says that “cancer stem cells are uniquely capable of proliferating extensively and forming new tumors, and that they go through an aberrant process of differentiation giving rise to phenotypically diverse cancer cells with a limited capacity to divide.” Which of these two theories is correct seems to depend upon the type of cancer.
Researchers are trying to determine whether such tumorigenic potential is within all cancer cells, or whether it is confined to a small number of cells. “If the cancer stem cells are rare,” he said, “the ability to identify these cells is critical to studying them. If the ability to become tumorigenic is common, distinguishing them is less valuable.”
“Studies are showing that a variety of cancers follow the cancer stem cell model, where markers can be identified that distinguish cancer stem cells from nontumorigenic cancer cells,” Dr. Morrison explained. “One of the untested, but underlying assumptions of the cancer stem cell model is that the difference that distinguishes tumorigenetic and nontumorigenetic cells is epigenetic, rather than genetic in nature.”
The clonal evolution model predicts genetic changes that will be deleterious to the cell, so the observation that not every cell is tumorigenetic is consistent with the clonal evolution model. What’s different about the cancer stem cell model is the idea that you can have cancer from only a small minority of the cells, that can differentiate into nontumorigenic cells, he said.
Most of the talk about cancer stem cells is mainly predictions, he cautioned. Conclusions about the fraction of cancers that follow the stem cell model are based primarily upon markers that distinguish tumorigenetic from nontumorigenetic cells. “So, all of the conclusions depend upon the robustness of these markers,” he noted, at least some of which are less robust than initially believed. Additionally, “Do some patients follow the model and others not? Does metastasis arise exclusively from migration of cancer stem cells? That makes intuitive sense, but isn’t tested.”
“We can’t make general conclusions that cancer stems cells are less sensitive to therapy,” Dr. Morrison said. There are several potential explanations for drug resistance. For example, “the apparent resistance may reflect a log kill, in which a therapy eradicates 95 percent of the tumorigenic cells but the remaining five percent proliferate.”
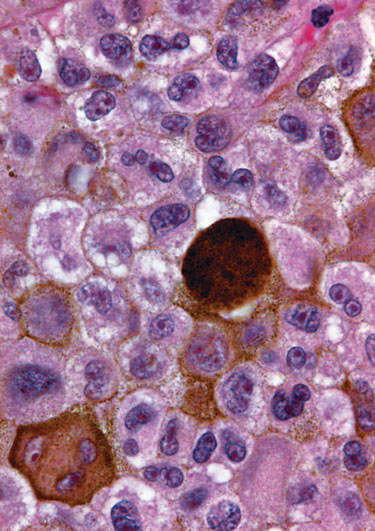
Pigmentation due to melanin deposition in a section of a melanoma stained with hematoxylin and eosin (University of Michigan Center for Stem Cell Biologics)
Disease Transfer
In his own lab, experiments with acute myeloid leukemia and acute lymphoblastic leukemia in mice showed that not every cell proliferates and transfers disease. “Cells that express a phenotype similar to hematopoietic stem cells are 400-fold enriched for leukemigenic activity as compared to the bulk population of bone marrow cells.” That supports the idea, he said, that “not every cell has the capacity to transmit disease, and cells that transfer disease are rare.” A similar study also showed that chronic myeloid leukemia (CML) followed the cancer stem cell model.
In studies of imatinib response against CML using a different mouse model, imatinib shrunk the (enlarged) spleen to normal levels. It did not cure the disease though. In fact, cells from these mice were more capable of transferring disease, showing that the “CML stem cells are orders of magnitude more resistant to imatinib than other cancer cells in the same mice, which is consistent with clinical experience.”
Melanoma is another model that was thought to follow a cancer stem cell model. It doesn’t, Dr. Morrison reported, based upon transplanting melanoma cells from several patients into NOD/SCID mice. After eight weeks, only a minority of cells had formed tumors. At 28 weeks, however, the ratio of tumorigenic cells had increased nearly eightfold. “One issue, therefore, is that people sometimes don’t run their assays long enough to detect the full spectrum of tumorigenic cells.”
Assay Optimization
As part of this research, Dr. Morrison’s lab decided to optimize the off-the-shelf assay to see whether changes could detect a broader range of cancer cells. “In addition to going longer and detecting a 10-fold increase in tumorigenic cells, if we use more highly immunocompromised mice we get almost a 200-fold increase in the detectable frequency of tumorigenic cells. Co-injection with Matrigel™ (which generally improved cell engraftment and thus increases cell survival without conferring tumorigenicity) yielded up to a 20-fold increase.
“Modifications in xenograft assays combine to dramatically increase the detectable frequency of tumorigenic cells, so that they were actually quite common,” Dr. Morrison said. In a larger study, initial data indicated that one in one million cells has the potential to form a tumor but, by slightly modifying the assay, the ratio changed to one in four.
“Therefore, for the first time in cancer biology, we can study tumorigenesis from single human cancer cells in vivo.” Rather than proving that metastatic melanoma doesn’t follow the cancer stem cell model, it proves that the tumorigenic cells are frequent.
“A separate question is whether the 28 percent of cells in which we can detect tumorigenic capacity are intrinsically different from the nontumorigenic cells that we cannot detect,” Dr. Morrison continued. In his experiments, involving 50 markers, in which 17 that are frequently heterogeneously expressed, the positive and negative fractions resulting in tumorigenesis. “We are unable to detect a hierarchical organization in melanoma, having looked very hard.” If such a marker is eventually found, he suggests it will have a very shallow hierarchy.
Resistance
The second portion of the workshop, led by Franziska Michor, Ph.D., computational biologist at Memorial Sloan-Kettering, addressed therapeutic resistance and the origin of cancer stem cells.
“If you have a drug that kills tumor stem cells, then over time, the tumor stem cell population will die out. If you have drugs that kill tumor cells but leave the cancer stem cells intact, then the tumor cell population will grow back,” she hypothesized.
In investigating the molecular response to imatinib among 68 patients, Dr. Michor found two distinct response slopes—one at 20 days life span and another at 125 days life span during therapy—with two distinct decay rates. The main body of cancer cells was depleted at a rate of 5% per day, while the other cells were depleted by 0.8% per day. This dramatic difference indicates that there are distinct subpopulations of cancer cells.
The idea that distinct subpopulations of cancer cells exists is further supported by a German study of patients who discontinued imatinib therapy after three years. Within one week, the cancer cells rapidly rebounded in 60% of the patients, peaking at levels higher than baseline. Therefore, it appears that cancer stem cells were not depleted by the drug and are, in fact, driving the disease. “There’s something going on that leads to resistance to imatinib,” Dr. Michor said.
Because imatinib is so specific as to be useless if a single base changes, it may be possible to answer that question while exploring the origin of tumorigenic cells. To do so, she turned to a mathematical analysis of the evolution of cancer stem cells, focusing upon identifying the mutation that triggered their drug resistance and considered several theories: that tissue-specific stem cells accumulated all the mutations needed to transform into tumor cells, that progenitors also accumulated the necessary mutations, and that the mutation conferring self-renewal to progenitors first arises in the stem cells without changing their phenotype.
Based upon her work with JAK2V617F mutations, she determined that progenitor cells are the most likely cell of origin for those mutations, which lead to cancer. This finding also may be relevant to other tumor types in tissues organized with a differentiation hierarchy.
Her next step was to identify the predicted first mutation. Using primary glioblastomas, Dr. Michor investigated cells in the subventricular zone of the brain that targets for transfection. Using mathematical modeling of a small number of cell divisions, she found that, “self-renewing transit amplifying cells are the most likely cell of origin for gliomas.”
In general discussions, Dr. Michor broached the idea that “some cancers really seem to follow the cancer stem cell model but are reversibly organized.” The role of microenvironments also was touched upon and should be the subject of additional research. “Tumor microenvironments are very important, but we don’t know how,” Dr. Morrison said. Microenvironments appear to kick off morphogenetic processes, so the markers that often are used become moving targets that, ultimately, are of little value, Dr. Michor added.
There are a lot of fundamental questions that remain about cancer stem cells, Dr. Morrison emphasized. “The way we think about cancer stem cells will change dramatically during the next five years. Cancer is so endlessly resourceful.”
Sidebar: Product Highlights from ASCB Meeting
In addition to the scientific presentations, the American Society for Cell Biology (ASCB) conference saw the launch of several new and innovative products. Here are some of the highlights:
Applied Biosystems, part of Life Technologies, introduced the Attune™ acoustic focusing cytometer amid much fanfare. The Attune works with 10 micron fluorescent-labeled beads in a capillary tube in real time, with the ability to slow, stop, and reverse the flow without loss of focus.
Consequently, the Attune offers greater sensitivity, lower fluid consumption, the potential for in-line washes, and a compact size, according to Nicholas Barthelemy, president of cell systems at Life Technologies.
Specifications weren’t released at the introduction, because, according to Barthelemy, “the data is improving as we finalize development.” However, he pointed out that it was about five times faster than other high-end cytometers, capturing 1,213 events per second. It weighs about 60 pounds, doesn’t need a fluidics cart, and has a low fluidics consumption rate of about one liter per day for full-time use, versus about seven liters for other cytometers.
Mayachitra introduced version 1.0 of its imago solution for bio-image management and analysis by showing a highly detailed image automatically stitched together from 444 retinal images. The actual mosaic, which would be 20 inches x 20 inches, provides an overall view that puts multiple small images in context. According to the company, imago handles 5-D images and integrates an interactive segmentation tool that automatically computes properties of individual cells, provides automatic cell counting with result modification capabilities, and offers rich graphical annotation.
Pseudo-colors can be assigned to aid searches, which can be organized by image name, embedded metadata, annotation, user tags, publication, image analysis, image content, or any combination of those parameters. Importantly, it also can perform searches based upon pattern matching or similar shapes using visual information.
Millipore introduced the Scepter™, a handheld, automated cell counter at the ASCB conference. About the size of an automated pipette, it features a sensor zone embedded in the dispensing pad and can report cell count and average cell volume within 20 seconds of inserting the tip into a cell culture sample, according to the company. It also can display a histogram of cell distribution by volume or diameter for an instant snapshot of the health of the culture. It can store up to 72 histograms. The Scepter also features an on-screen menu.
Eric Schulze, a scientist at the Qilong Ying Lab at the University of Southern California, is developing embryonic stem cells and mouse embryonic fibroblasts. He reports that Scepter works well for cells between 5 and 36 microns and is intuitive to use. It will become commercially available during the latter half of the first quarter, 2010.
Nikon introduced the n-SIM and n-Storm super-resolution microscopes, which will be available in early summer. The n-SIM (structured illumination microscopy) provides resolutions of approximately 85 to 100 nm—nearly twice that of conventional microscopes—and offers a time resolution of 0.6 seconds per frame, suitable for live-cell imaging.
The n-Storm (stochastic optical resonance microscopy) reportedly enhances resolution ten times beyond that of conventional microscopes, providing 20–30 nm XY resolution and about 50–60 nm Z resolution. This microscope can switch between high resolution 2-D and 3-D fluorescent images with a flick of a switch.
Platypus introduced the Oris™ Pro Assay system to provide alternatives in cell-migration studies. The Oris Pro replaces the stopper the original Oris used to form the cell-migration zone with a biocompatible gel that dissolves in contact with the cells. The change did not affect cell viability, migration, or cytotoxicity. The primary differences are that the second-generation version uses the biocompatible gel, which reduces handling time and can be automated for liquid handling, the company reported.
Olympus introduced the Cell^TIRF motorized, multicolor illuminating microscope with four channels for simultaneous image acquisition and instant setting and angle confirmation. Wavelengths are optimized to have the same depth. The unit integrates easily with an incubator system.
PhotoMetrics announced a new feature set for the Evolve 512 EMCCD camera, of which Quant-View™ is the most notable. This new quantitation technique counts photos, which are then converted into an analog-to-digital unit in real time. Gain factor calculations and conversions also occur in real time. This information can be used to calculate the number of electrons generated at any pixel per unit measure of time. This provides a standard unit of measurement that can be used to compare or contrast experiments over time or across labs. Each camera includes a certification of performance and the ability to be recalibrated in about five minutes.
imago is an image database and analysis system designed for managing microscopic images. (Gabriel Luna, Retinal Cell Biology Lab, NRI, UCSB)



