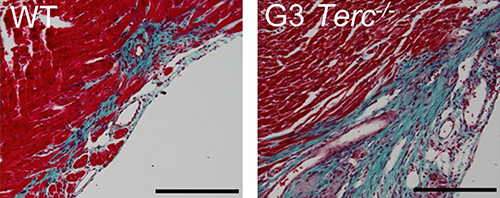
![Telomerase-deficient mice undergo premature telomere shortening, and so, unlike wild-type mice, they are unable to regenerate damaged heart tissue, even at day 1 of age. Twenty-eight days after injury, telomerase-deficient heart tissue (right) has larger fibrotic regions (blue) compared with wild-type myocardium (left). [Aix et al., 2016, The Journal of Cell Biology]](https://genengnews.com/wp-content/uploads/2018/08/May31_2016_CellBiology_Telomerase2392461372-1.jpg)
Telomerase-deficient mice undergo premature telomere shortening, and so, unlike wild-type mice, they are unable to regenerate damaged heart tissue, even at day 1 of age. Twenty-eight days after injury, telomerase-deficient heart tissue (right) has larger fibrotic regions (blue) compared with wild-type myocardium (left). [Aix et al., 2016, The Journal of Cell Biology]
It’s a deadly combination—heart attack and cell cycle arrest. The heart attack damages cardiac tissue, which cannot be repaired because, for unknown reasons, mature cardiomyocytes lack regenerative capacity. It is known that cardiomyocytes withdraw from the cell cycle soon after birth. Additional details, however, have been lacking.
Now, as a result of research from the Spanish National Center for Cardiovascular Research (CNIC), new molecular-level details have emerged: Cardiomyocyte cell-cycle arrest is linked to telomere dysfunction. Essentially, the telomeres that protect the ends of heart muscle cell chromosomes erode rapidly after birth. As a result, heart muscle cells have little ability to proliferate, which means that damaged heart tissue cannot be repaired through the proliferation of new heart muscle cells.
This finding appeared May 30 in The Journal of Cell Biology, in an article entitled “Postnatal Telomere Dysfunction Induces Cardiomyocyte Cell-Cycle Arrest through p21 Activation.” This article describes a team of CNIC researchers led by Ignacio Flores, Ph.D., who examined genetically modified newborn mice to find cardiomyocyte cell-cycle arrest for telling physiological signals.
Newborn mice, like newborn babies, are able to regenerate damaged heart tissue. Their heart muscle cells, or cardiomyocytes, can proliferate and repair the heart in the first week after birth, but this regenerative capacity is lost as the mice grow older and the majority of their cardiomyocytes withdraw from the cell cycle.
Dr. Flores and colleagues wondered whether the cause of this cell-cycle arrest might involve telomeres, repetitive DNA sequences that protect the ends of chromosomes. If telomeres grow too short—due, for example, to a loss of the telomere-extending telomerase enzyme—cells can mistake chromosome ends for segments of damaged DNA, leading to the activation of a checkpoint that arrests the cell cycle.
“We show that telomerase activity and cardiomyocyte telomere length decrease sharply in wild-type mouse hearts after birth, resulting in cardiomyocytes with dysfunctional telomeres and anaphase bridges and positive for the cell-cycle arrest protein p21,” wrote the article’s authors. “We further show that premature telomere dysfunction pushes cardiomyocytes out of the cell cycle.”
Essentially, the CNIC researchers found that the telomeres rapidly eroded in the first week after birth. Also, the team found that this erosion coincided with a decrease in telomerase expression and was accompanied by the activation of the DNA damage response and a cell cycle inhibitor called p21.
“Cardiomyocytes from telomerase-deficient mice with dysfunctional telomeres (G3 Terc−/−) show precocious development of anaphase-bridge formation, p21 up-regulation, and binucleation,” explained the authors of The Journal of Cell Biology article. “In line with these findings, the cardiomyocyte proliferative response after cardiac injury was lost in G3 Terc−/− newborns but rescued in G3 Terc−/−/p21−/− mice.”
Telomerase-deficient mice have shorter telomeres than wild-type animals, and, the researchers discovered, their cardiomyocytes already begin to stop proliferating 1 day after birth. When Dr. Flores and colleagues injured the hearts of 1-day-old mice, telomerase-deficient cardiomyocytes failed to proliferate or regenerate the injured myocardium. In contrast, wild-type cardiomyocytes were able to proliferate and replace the damaged tissue.
They also found that knocking out the cell cycle inhibitor p21 extended the regenerative capacity of cardiomyocytes, allowing 1-week-old p21-deficient mice to repair damaged cardiac tissue much more effectively than week-old wild-type animals.
Maintaining the length of cardiomyocyte telomeres might therefore boost the regenerative capacity of adult cells, improving the recovery of cardiac tissue following a heart attack. “We are now developing telomerase overexpression mouse models to see if we can extend the regenerative window,” said Dr. Flores.