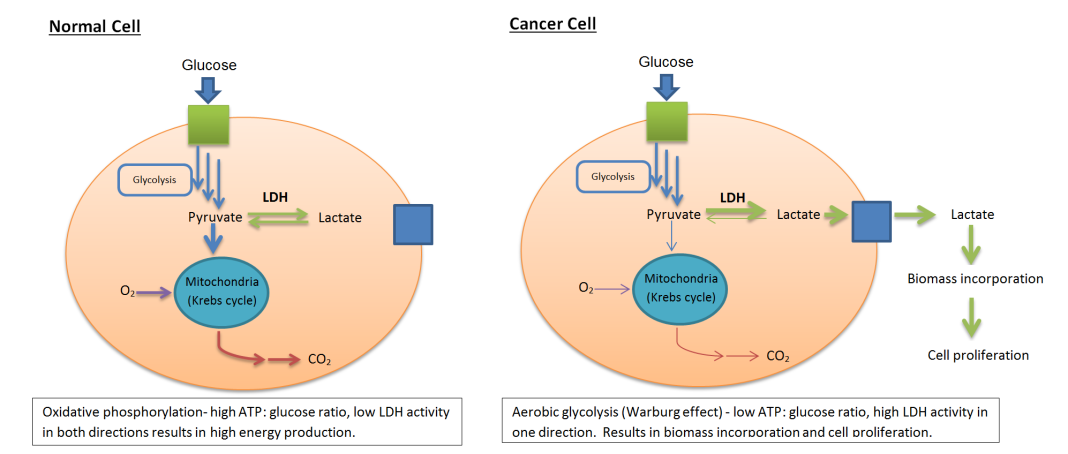
![Schematic ofthe Warburg effect. [WikiCommons]](https://genengnews.com/wp-content/uploads/2018/08/LDH_activity_normal_vs_canceous_cells11152498450-1.png)
Schematic ofthe Warburg effect. [WikiCommons]
For those with a tendency toward the conspiratorial, this is not a takedown about how “Big Sugar” is poisoning the world and increasing cancer rates. Instead, new evidence from a collaborative team of Belgian researchers at the University of Leuven (KU Leuven), the University of Brussels (VUB), and the Vlaams Institute for Biotechnology (VIB) has clarified how the Warburg effect—a phenomenon in which cancer cells rapidly break down sugars—stimulates tumor growth. This discovery provides evidence for a positive correlation between sugar and cancer, which may have far-reaching impacts on tailor-made diets for cancer patients. Findings from the new study were published recently in Nature Communications, in an article entitled “Fructose-1,6-Bisphosphate Couples Glycolytic Flux to Activation of Ras.”
“Our research reveals how the hyperactive sugar consumption of cancerous cells leads to a vicious cycle of continued stimulation of cancer development and growth,” explained senior study author Johan Thevelein, Ph.D., a professor at KU Leuven. “Thus, it is able to explain the correlation between the strength of the Warburg effect and tumor aggressiveness. This link between sugar and cancer has sweeping consequences. Our results provide a foundation for future research in this domain, which can now be performed with a much more precise and relevant focus.”
This study represents a nine-year joint research project conducted by VIB, KU Leuven, and VUB that focused on the observation that tumors convert significantly higher amounts of sugar into lactate compared to healthy tissues—the Warburg effect. As one of the most prominent features of cancer cells, this phenomenon has been extensively studied and even used to detect brain tumors, among other applications. But thus far, it has been unclear whether the effect is merely a symptom of cancer or a cause.
“We now reveal an evolutionarily conserved mechanism linking fermentation to activation of Ras, a major regulator of cell proliferation in yeast and mammalian cells, and prime proto-oncogene product,” the authors wrote. “A yeast mutant (tps1?) with overactive influx of glucose into glycolysis and hyperaccumulation of Fru1,6bisP, shows hyperactivation of Ras, which causes its glucose growth defect by triggering apoptosis. Fru1,6bisP is a potent activator of Ras in permeabilized yeast cells, likely acting through Cdc25. As in yeast, glucose triggers activation of Ras and its downstream targets MEK and ERK in mammalian cells. Biolayer interferometry measurements show that physiological concentrations of Fru1,6bisP stimulate dissociation of the pure Sos1/H-Ras complex. Thermal shift assay confirms direct binding to Sos1, the mammalian ortholog of Cdc25.”
Yeast cell research was essential to the discovery, as these cells contain the same Ras proteins commonly found in tumor cells, which can cause cancer in mutated form. Using yeast as a model organism, the research team examined the connection between Ras activity and the highly active sugar metabolism in yeast.
“We observed in yeast that sugar degradation is linked via the intermediate fructose 1,6-biophosphate to the activation of Ras proteins, which stimulate the multiplication of both yeast and cancer cells,” Dr. Thevelein noted. “It is striking that this mechanism has been conserved throughout the long evolution of yeast cell to human.”
Dr. Thevelein concluded that “the main advantage of using yeast was that our research was not affected by the additional regulatory mechanisms of mammalian cells, which conceal crucial underlying processes. We were thus able to target this process in yeast cells and confirm its presence in mammalian cells. However, the findings are not sufficient to identify the primary cause of the Warburg effect. Further research is needed to find out whether this primary cause is also conserved in yeast cells.”