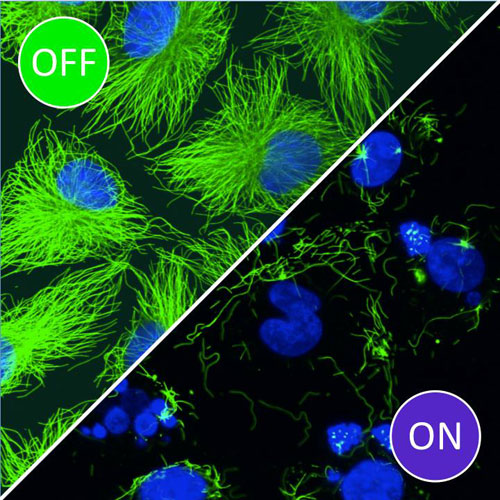
![In cells incubated with photostatins in the dark (OFF), microtubules (green) and nuclei (blue) are unaffected. However, exposure to blue light (ON) prompts disintegration of the microtubules, which leads to cell death. [Dirk Trauner/LMU]](https://genengnews.com/wp-content/uploads/2018/08/PhotstatinsImage1051322386-1.jpg)
In cells incubated with photostatins in the dark (OFF), microtubules (green) and nuclei (blue) are unaffected. However, exposure to blue light (ON) prompts disintegration of the microtubules, which leads to cell death. [Dirk Trauner/LMU]
For most drugs, it is always show time, which is seldom a problem, except if the drugs are mitotic show-stoppers, like the powerful chemotherapeutic Taxol, or the Vinca alkaloids. These drugs interfere with microtubule dynamics, and hence cell division, preventing the proliferation of cancer cells and eventually inducing cell death. Unfortunately, cell death can also be induced in healthy cells that need to divide quickly, such as the specialized cells found in bone marrow and the gastrointestinal tract.
To prevent anticancer drugs from acting on healthy cells, and prematurely bringing the curtain down, drug developers are auditioning photopharmacology agents, molecules that show activity only if they are exposed to light. Photopharmacology agents that would inhibit cell division only where and when needed, as directed by carefully controlled illumination, could help eliminate harmful side-effects.
This approach was explored by scientists based at Ludwig-Maximilians-Universität München (LMU) and the University of Lyon. They developed photoresponsive derivatives of an antimitotic drug that could provide the basis for precisely targeted tumor therapies.
“We have incorporated a light-sensitive molecular switch into a known inhibitor of microtubule polymerization, so that the agent only becomes active when it is exposed to blue light,” said LMU’s Oliver Thorn-Seshold, Ph.D. “Our aim was to modify a microtubule inhibitor in such a way that its action could be limited to a targeted tumor tissue, and we have now achieved this goal by designing a suitable light-sensitive molecular switch for several chemical derivatives of the bioactive agent colchicine.”
The LMU- and Lyon-based researchers detailed their work July 16 in the journal Cell, in an article entitled, “Photoswitchable Inhibitors of Microtubule Dynamics Optically Control Mitosis and Cell Death.” In this article, they refer to their photoswitchable inhibitors, which can be switched either ON or OFF, as photostatins.
“Photostatins modulate microtubule dynamics with a subsecond response time and control mitosis in living organisms with single-cell spatial precision,” the authors wrote. “In longer-term applications in cell culture, photostatins are up to 250 times more cytotoxic when switched on with blue light than when kept in the dark.”
The researchers believe that photostatins would be most likely to find clinical application in the treatment of localized tumors that are easily accessible to light, such as retinoblastoma—the most common pediatric tumor of the eye—and skin cancers.
“However, fiber optics are already in routine use in medical procedures such as endoscopic examination of the gastrointestinal tract. And given the pace of development in LED-based technology, miniaturized and more powerful LEDs that can be implanted in the body—like tiny, blinking pacemakers—are probably not that far off,” noted Dr. Thorn-Seshold. “We hope that our photosensitive drugs will help to stimulate the search for such innovative therapeutic approaches.”
The authors of the cell article also suggested that microtubule-inhibiting photostatins could be useful in advancing cell biology. For microtubules not only play a crucial part in cancer progression, they also play complex roles in the transport of signaling molecules, cargoes, and organelles around cells, as well as in cell movement, migration and repair, and are critical for the organization of embryonic development.
Since photostatins enable exquisitely precise spatial and temporal control over microtubule dynamics—enabling control over individual cells within networks, and with switching times under a second—they open up new ways of exploring how the functions and effects of microtubules are regulated in space and time.
“One could, for instance, arrest the differentiation of a selected cell over a given time period, then remove the brake and observe its recovery and how it integrates into the further development of an organism,” explained LMU’s Dirk Trauner, Ph.D., who co-led the current study. “This promises to give us new insights into the role of various types of progenitor cells during development.”