Sponsored content brought to you by
Gene editing is a powerful tool for the development of cell therapies across disciplines. Over the past several years, CRISPR has become the standard tool for gene editing; however, when developing novel therapies, using CRISPR is only part of the process. In order to evaluate the efficacy of a CRISPR-edited therapy, functional proteomics is needed, as CRISPR itself cannot confirm the function of gene edits. Functional single-cell proteomics is uniquely able to determine if cells are functioning as intended in a therapy product, while methods such as flow cytometry and surface phenotyping can only estimate function. IsoPlexis’ single-cell technology detects the highly polyfunctional cell subsets that correlate to in vivo biology in cellular therapies which are typically missed with bulk assays. Identifying these powerful functional T cell drivers can give scientists a leg up when developing complex engineered immune cell therapies.
In cell and gene therapies, IsoPlexis’ polyfunctionality metric can be used in preclinical development to evaluate a therapy’s durability and potency. By combining CRISPR with IsoPlexis’ single-cell secreted proteomics, researchers can engineer therapies for increased efficacy and see how their gene edits translate to in vivo function.
IsoPlexis’ Single-Cell Proteomics Confirms Antitumor Function of CRISPR-Edited NK Cells
In a study published in Cell Stem Cell, IsoPlexis’ Single-Cell Secretome solution identified a correlation between polyfunctional CRISPR-edited NK cells and improved function against leukemia. NK cells are ideal for use in immune therapies due to their ability to kill unhealthy cells without antigen stimulation. However, while the antitumor response of NK cells is powerful, it is short lived due to their poor in vivo lifespan. Researchers Zhu et al. sought to overcome this limitation of NK cells in immune therapies and increase efficacy by developing a sustainable supply of NK cells with improved in vivo persistence and potency. They did so by deriving NK cells from induced pluripotent stem cells (iPSCs) and using CRISPR to engineer improved antitumor function, which was confirmed by IsoPlexis’ single-cell proteomics.
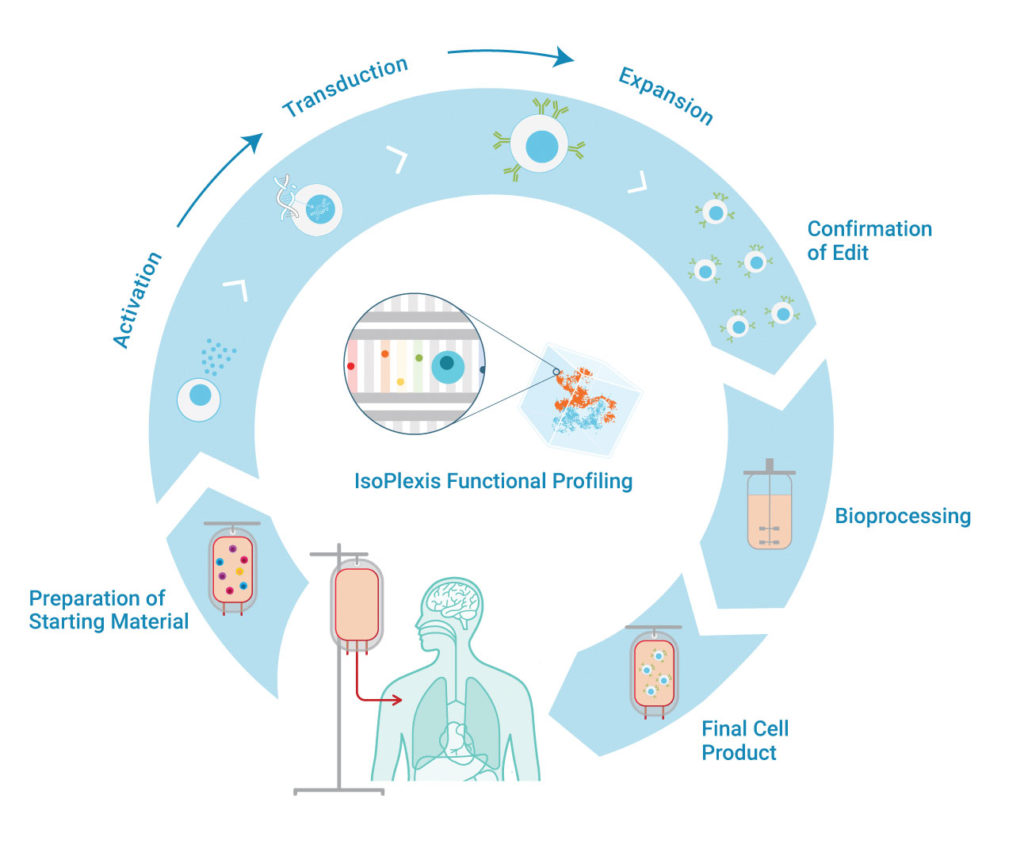
Zhu et al. predicted that deleting CISH, the human gene for cytokine-inducible SH2-containing protein (CIS), would improve the function of the iPSC-NK cells. CIS is a negative regulator of IL-15 signaling with NK cells. IL-15 stimulates NK cell differentiation, proliferation, activation, and survival, but it can cause toxicities in high doses. Therefore, researchers aimed to increase the sensitivity of NK cells to IL-15 by deleting CISH, thereby increasing beneficial effects of low doses of IL-15 while avoiding toxicity. Results showed that the CRISPR-edited cells had increased in vivo persistence compared to wild- type (WT) NK cells. Furthermore, when these cells were tested against acute myeloid leukemia (AML), the CISH-KO iPSC-NK cells demonstrated enhanced antitumor activity. This improved persistence and antitumor function correlated with upregulated polyfunctionality and high functional potency. IsoPlexis’ platform found that approximately 5% of the CISH-KO iPSC-NK cells were polyfunctional, compared to 1–2% of the WT iPSC-NK and peripheral blood NK cells. Furthermore, the polyfunctional strength index (PSI) was increased >10-fold in the CISH-KO iPSC-NK cells.
Shedding Light on Function in Gene Edits
This study highlights how functional single-cell proteomics works in tandem with CRISPR across the development and testing process of cell and gene therapies. The IsoPlexis platform builds upon well-validated ELISA technology, adding highly multiplexed single-cell capabilities (up to 30+ cytokines) which detect functionally heterogenous cells. IsoPlexis’ technology confirms whether engineered and gene-edited cells function as intended after modification and improves researchers’ understanding of how potential therapies will affect patients. The field of research on engineered cell therapies is quickly growing and holds significant promise for the treatment of a wide range of complex diseases. IsoPlexis’ walk-away automated platform helps improve these therapies by detecting the potent cells that other technologies miss, leading to more targeted and effective therapies and accelerating the process of getting these therapies to patients.
Download the eBook, Single-Cell Functional Proteomics: Empowering Translational Genome Editing in Immunotherapy, now.