![The molecular mechanism of vitamin E coupled heteroduplex oligonucleotide molecule within cells. [Tokyo Medical and Dental University]](https://genengnews.com/wp-content/uploads/2018/08/97298_web2391031919-1.jpg)
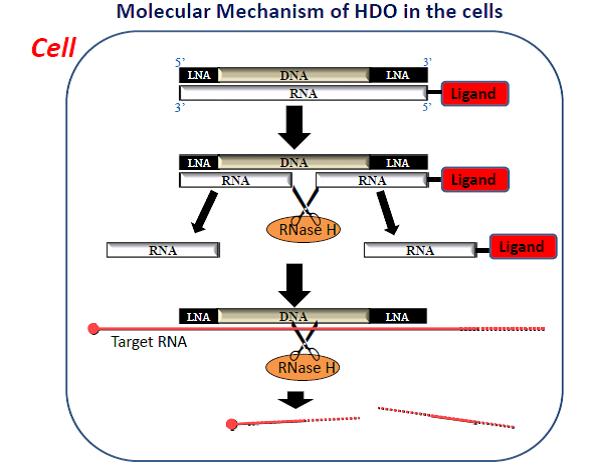
The molecular mechanism of vitamin E coupled heteroduplex oligonucleotide molecule within cells. [Tokyo Medical and Dental University]
Gene silencing is an effective method for turning down or off the aberrant genetic message that leads toward disease phenotypes. Therapeutics that take advantage of this technology are beginning to enter the market. However, opportunities still remain for improving the efficient delivery of nucleic acid drugs to the relevant diseased tissues.
Now, a team of researchers from the Tokyo Medical and Dental University and Osaka University have developed a new oligonucleotide complex, called heteroduplex oligonucleotide (HDO), that they have observed to be significantly potent at reducing the levels of target RNA while simultaneously lowering side effects, such as liver dysfunction.
“Two major types of RNA targeting oligonucleotide drugs are currently being developed as therapeutic platforms for reduction of target gene expression—short interfering RNA (siRNA) and RNase H dependent antisense oligonucleotides (ASO),” explained senior author Takanori Yokota, M.D., Ph.D., professor in the department of neurology and neurological science at Tokyo Medical and Dental University.
Insufficient delivery, poor cellular uptake of oligonucleotides, and their inefficient access to target RNA are major impediments to in vivo silencing methods. Dr. Yokota and his team saw an opportunity to design a better oligo, developing the short DNA/RNA HDO molecule. HDO is composed of DNA/locked nucleic acid gapmer as ASO and its complementary RNA strand.
“HDO has a structure different from double-stranded RNA used for siRNA and single-stranded DNA used for ASO, and different functional molecular mechanisms from siRNA or ASO in the cells,” said Dr. Yokota.
“When α-tocopherol (vitamin E) as drug delivery moiety is conjugated to ASO directly, its silencing effect is reduced because the conjugated lipid interferes with the mechanisms of ASO,” Dr. Yokota continued. “On the other hand, when α-tocopherol is conjugated to cRNA of HDO, an α-tocopherol molecule can improve the delivery of HDO to the liver by five times, making the gapmer DNA strand active by releasing it from HDO through the cleavage of cRNA by cellular nucleases.”
The investigators found that the Toc-HDO was markedly more potent at reducing the target mRNA, in this instance to lower serum LDL levels, when compared to the parental ASO strand. Specifically, the Toc-HDO was 22 times more potent at reducing Apolipoprotein B mRNA than the parent ASO within the liver. Moreover, the pharmacological effects lasted for more than a month with a single injection.
The findings from this study were published recently in Nature Communications through an article entitled “DNA/RNA heteroduplex oligonucleotide for highly efficient gene silencing.”
“A significant improvement in activity is also observed when targeting another gene in the liver,” Dr. Yokota noted. “In addition, the Toc-HDO using another chemically modified nucleic acid instead of LNA in the wing portion of DNA strand also showed a similarly enhanced potency. HDO technique can be applied to any ASO previously reported. Furthermore, the highly potent suppression of the target messenger RNA is observed not only in rodents but also non-human primates.”
These results suggest that DNA/RNA heteroduplex could change the basic technology of oligonucleotide drugs, and it opens up a new horizon for human gene therapy as a novel class of oligonucleotide drugs.
“The high potency of Toc-HDO results in a reduction of liver dysfunction observed in the parent ASO at the same silencing effect,” Dr. Yokota stated, “probably due to much less administered dose of nucleotide. Our team is currently investigating more detailed mechanisms of the HDO and application of ligand-conjugated HDO to other organs, including the brain.”