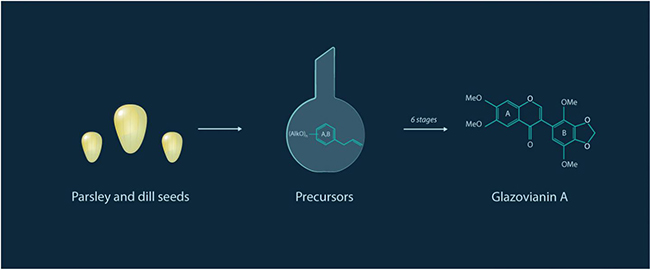
![Basic synthesis of the antimitotic compound glaziovianin A [Moscow Institute of Physics and Technology]](https://genengnews.com/wp-content/uploads/2018/08/118633_web1241711253-1.jpg)
Basic synthesis of the antimitotic compound glaziovianin A [Moscow Institute of Physics and Technology]
Next time you take a bite into that delicious deli pickle or spread out that lovely bright green pesto sauce, give a thought to how some of the main seasonings for these foods can greatly aid in the development of novel anticancer agents. This is exactly what a team of scientists from the Moscow Institute of Physics and Technology (MIPT), the N.D. Zelinsky Institutes of Organic Chemistry, Developmental Biology, and Cell Biophysics have done by proposing an efficient new approach toward the synthesis of potential chemotherapeutic agents—which are based on the extraction of compounds from parsley and dill seeds.
Most chemotherapy regimens target rapidly dividing cells, such as cancer cells, through the use of antimitotic agents that inhibit the growth of these cells by disrupting the process of cell division. Cancer cells divide much more frequently than normal cells and therefore are more susceptible to the effects of antimitotics. Moreover, many antimitotics affect microtubule dynamics, which are critical for cellular mitosis. Microtubules are comprised of a protein called tubulin, which is the binding site for most antimitotics, leading to the disruption of the cell cycle and ultimately cell death.
In the current study, the investigators focused on the synthesis of the potent antimitotic agent glaziovianin A (GVA), which is typically isolated from the leaves of the Brazilian tree Ateleia glazioviana Baill. Previous reports on the synthesis of GVA have described the process as rather laborious, requiring expensive precursors and catalysts. The authors proposed a novel and more efficient six-stage synthesis process (the usual process has nine stages) for GVA. Precursors for the process were derived from the seeds of common plants, namely parsley and dill.
“Both improvement of existing therapies and search for innovative approaches are essential components of a quest to treat cancer,” explained MIPT adjunct professor Alexander Kiselev, Ph.D. “Our combined team developed a simple method of producing GVA and its structural analogs, which inhibit the growth of human tumor cells, using feasible building blocks from nature. Furthermore, evaluation of these novel agents in vivo using our validated sea urchin embryo assays yielded several promising candidates selectively affecting tubulin dynamics.”
The findings from this study were published recently it the Journal of Natural Products in an article entitled “Efficient Synthesis of Glaziovianin A Isoflavone Series from Dill and Parsley Extracts and Their in Vitro/in Vivo Antimitotic Activity.”
In addition to GVA, a number of its structural analogs were synthesized to help find analogs with favorable antimitotic properties. The antitumor activity was tested via two independent methods using the sea urchin embryos and human cancer cells.
The embryos of sea urchins were used to mimic actively dividing tumor cells dependent on tubulin dynamics. The Russian team added test substances to an aqueous medium with the embryos and determined the concentrations at which the rate of division changes and when it comes to a complete stop. Using the embryos, the researchers were able to determine several critical parameters essential for an anticancer molecule. These included the specific antimitotic effect, solubility, overall toxicity, and biomembrane permeability.
Additionally, to further confirm the antitumor effect of active molecules, this was studied with various human cancer cells, e.g., lung carcinoma, melanoma, prostate, breast, colon, and ovarian cancers. The experiments showed that the test substances were effective at limiting the growth of melanoma cells and nontoxic to healthy blood cells used as a control. Detailed structure–activity relationship studies in both assay systems converged on GVA as being the most active antitubulin agent.
“Notably, the target compounds were not cytotoxic against human peripheral blood mononuclear cells up to 10 μM concentration,” the authors wrote. “Phenotypic readouts from the sea urchin assay unequivocally suggest a direct microtubule-destabilizing effect of GVA and its alkoxy derivatives.”