Scientists at the National Cancer Institute (NCI) report that in a clinical trial of 42 women with metastatic breast cancer, 28 (or 67 percent) generated an immune reaction against their cancer. The approach, which utilizes tumor-infiltrating lymphocytes (TILs), was used to treat six women, half of whom experienced measurable tumor shrinkage. Results from the trial appeared in an article (“Breast Cancers Are Immunogenic: Immunologic Analyses and a Phase II Pilot Clinical Trial Using Mutation-Reactive Autologous Lymphocytes”) in the Journal of Clinical Oncology.
“Metastatic breast cancer (mBrCa) is most often an incurable disease with only modest responses to available immunotherapies. This study investigates the immunogenicity of somatic mutations in breast cancer and explores the therapeutic efficacy in a pilot trial of mutation-reactive tumor-infiltrating lymphocytes (TILs) in patients with metastatic disease.” write the investigators.
“Forty-two patients with mBrCa refractory to previous lines of treatment underwent surgical resection of a metastatic lesion(s), isolation of TIL cultures, identification of exomic nonsynonymous tumor mutations, and immunologic screening for neoantigen reactivity. Clinically eligible patients with appropriate reactivity were enrolled into one cohort of an ongoing phase II pilot trial of adoptive cell transfer of selected neoantigen-reactive TIL, with a short course of pembrolizumab.
“TILs were isolated and grown in culture from the resected lesions of all 42 patients with mBrCa, and a median number of 112 (range: 6–563) nonsynonymous mutations per patient were identified. Twenty-eight of 42 (67%) patients contained TIL that recognized at least one immunogenic somatic mutation (median: three neoantigens per patient, range: 1–11), and 13 patients demonstrated robust reactivity appropriate for adoptive transfer.
“Eight patients remained clinically eligible for treatment, and six patients were enrolled on a protocol of adoptive cell transfer of enriched neoantigen-specific TIL, in combination with pembrolizumab (≤ 4 doses). Objective tumor regression was noted in three patients, including one complete response (now ongoing over 5.5 years) and two partial responses (6 and 10 months).
“It’s popular dogma that hormone receptor–positive breast cancers are not capable of provoking an immune response and are not susceptible to immunotherapy,” said study leader Steven A. Rosenberg, MD, PhD, chief of the Surgery Branch in NCI’s Center for Cancer Research. “The findings suggest that this form of immunotherapy can be used to treat some people with metastatic breast cancer who have exhausted all other treatment options.”
The immunotherapy approach used in the trial was pioneered in the late 1980s by Rosenberg and his colleagues at NCI. It relies on TILs, T cells that are found in and around the tumor.
TILs can target tumor cells that have neoantigens, that the immune cells recognize. Neoantigens are produced when mutations occur in tumor DNA. Other forms of immunotherapy have been found to be effective in treating cancers (such as melanoma) that have many mutations, and therefore many neoantigens. Its effectiveness in cancers that have fewer neoantigens, such as breast cancer, however, has been less clear, according to the scientists.
The results of the new study come from an ongoing Phase II clinical trial being carried out by Rosenberg and his colleagues. This trial was designed to see if the immunotherapy approach could lead to tumor regressions in people with metastatic epithelial cancers, including breast cancer. In 2018, the researchers showed that one woman with metastatic breast cancer who was treated in this trial had complete tumor shrinkage, known as a complete response.
In the trial, the researchers used whole-genome sequencing to identify mutations in tumor samples from 42 women with metastatic breast cancer whose cancers had progressed despite all other treatments. The researchers then isolated TILs from the tumor samples and, in lab tests, tested their reactivity against neoantigens produced by the different mutations in the tumor.
Twenty-eight women had TILs that recognized at least one neoantigen. Nearly all the neoantigens identified were unique to each patient.
“It’s fascinating that the Achilles’ heel of these cancers can potentially be the very gene mutations that caused the cancer,” said Rosenberg. “Since that 2018 study, we now have information on 42 patients, showing that the majority give rise to immune reactions.”
For the six women treated, the researchers took the reactive TILs and grew them to large numbers in the lab. They then returned the immune cells to each patient via intravenous infusion. All the patients were also given four doses of the immune checkpoint inhibitor pembrolizumab (Keytruda) before the infusion to prevent the newly introduced T cells from becoming inactivated.
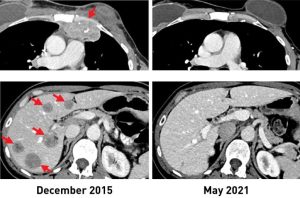
After the treatment, tumors shrank in three of the six women. One is the original woman reported in the 2018 study, who remains cancer free to this day. The other two women had tumor shrinkage of 52% and 69% after six months and 10 months, respectively. However, some disease returned and was surgically removed. Those women now have no evidence of cancer approximately five years and three and a half years, respectively, after their TIL treatment.
The researchers acknowledged that the use of pembrolizumab, which has been approved for some early-stage breast cancers, may raise uncertainties about its influence on the outcome of TIL therapy. However, they said, treatment with such checkpoint inhibitors alone has not led to sustained tumor shrinkage in people with hormone receptor–positive metastatic breast cancer.
Rosenberg said that with the anticipated opening early this year of NCI’s new building devoted to cell-based therapies, he and his colleagues can begin treating more individuals with metastatic breast cancer as part of the ongoing clinical trial. He noted that this new immunotherapy approach could potentially be used for people with other types of cancer as well.