The results of a study by researchers at the Rensselaer Polytechnic Institute Center for Biotechnology and Interdisciplinary Studies have shown how a protein called PRC1 acts as a “viscous glue” during mitotic cell division, to help precisely control the speed at which the two sets of DNA are separated as division proceeds. The finding could explain why too much or too little PRC1 disrupts the process and causes genome errors linked to cancer. An overabundance of PRC1 is a telltale sign in many cancer types, including prostate, ovarian, and breast cancer.
“Like a lot of biological processes, it’s a bit of a Goldilocks problem,” said research lead Scott Forth, PhD, an assistant professor of biological sciences. “If you don’t have this protein, you’re in trouble, because the cell fails at division. If you have too much, we think that it gums up the works and holds everything together too much, which may be how this protein is linked to cancer. There’s a sort of sweet spot in healthy cell division, where there’s just the right amount controlling the rates carefully and precisely.” Forth is corresponding author of the team’s published paper in Developmental Cell, which is titled, “The Mitotic Crosslinking Protein PRC1 Acts Like a Mechanical Dashpot to Resist Microtubule Sliding.”
For mitotic cell division, a single cell must copy its DNA into two identical sets, and then rapidly and efficiently pull that DNA apart into two new daughter cells. It’s a physical act, and the cellular structure that does it, the mitotic spindle, is a machine that uses mechanical forces—push, pull, and resistance—to complete the task. At the nitty-gritty level of DNA, motor proteins, and microtubules, this process thus takes its cue from physics.
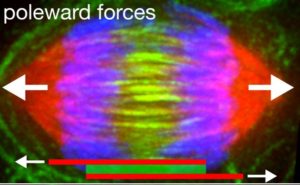
The Forth lab examined the physical forces exerted by components of cellular structures like the mitotic spindle. The spindle is formed when two centrosomes take a position on opposite sides of the two newly created, and hopefully identical, sets of chromosomes massed near the center of the cell. A dense network of microtubules extends from the centrosomes, forming a cage that surrounds and connects the chromosomes. Then the microtubules—aided by millions of proteins and motor proteins—begin to shorten and slide, pulling the chromosomes toward the centrosomes, until the two sets have been separated. “Within the spindle, subsets of microtubules are organized into specialized arrays whose filaments can undergo relative sliding,” the team noted.
Microtubule sliding is driven in part by motor proteins that either crosslink filaments or cluster at microtubule ends to exert active sliding or pulling forces along the filament, the team continued. Non-motor proteins can then crosslink filaments spindle microtubules into higher order arrays such as overlapping bundles. “It has been proposed that crosslinking by such proteins may produce a ‘brake-like’ resistance to filament sliding that acts to balance motor-driven forces, leading to an integration of active and passive forces that sets parameters such as spindle length and filament sliding velocity.”
PRC1 is one of these non-motor mitotic crosslinking proteins in humans. The protein is a long, springy molecule with a head at either end that links two microtubules along their length. Near the center of the mitotic spindle, large quantities of PRC1 link groups of microtubules into bundles. “PRC1 produces a viscous frictional force, a drag that increases with speed,” said Forth. “The friction it produces is similar to that of water—if you try to move your hand through water slowly, you move easily, but if you push your hand fast, the water pushes back hard.”
Forth’s team created a controlled version of the microtubule sliding mechanism in the lab and used an optical trapping technique to measure the frictional force PRC1 exerts between the sliding microtubules. Optical trapping relies on a tightly focused laser beam that attracts an object—in this case, a miniscule polystyrene bead—attached to the microtubule. The researchers use the laser beam to pull on the bead—similar to the ‘tractor beam’ of science fiction—and convert the shift in refracted light as the bead resists the pull of the trap into a direct measure of force.
The investigators also tagged PRC1 with a fluorescent molecule, allowing them to observe its shifting movement and distribution as the microtubules were pulled apart. They used total internal reflection fluorescence microscopy to collect images of the experiment while simultaneously recording the forces. “ … using simultaneous total internal reflection fluorescence (TIRF) microscopy and optical trapping methods, we directly measure the resistive forces generated by ensembles of PRC1 molecules crosslinking microtubule pairs,” the scientists explained.
Forth and his colleagues found that, as more of the protein is added into the system, the microtubules meet more resistance as they move faster. Essentially, PRC1 behaves like a glue holding the cell together. “Surprisingly, we observe that pausing during sliding allows the system to relieve tension and reinitiating sliding results in increased resistive forces, suggesting that upon cessation of relative sliding, PRC1 molecules can rearrange within the overlap to form a higher-order configuration that more strongly resists microtubule separation,” they added. “Our results suggest that PRC1 ensembles act similarly to a mechanical dashpot, producing significant resistance against fast motions but minimal resistance against slow motions, allowing for the integration of diverse motor activities into a single mechanical outcome.”
“This research reveals the inner workings of a fundamental mechanism of biology, providing knowledge that better positions us to defeat cancer,” said Curt Breneman, PhD, dean of the School of Science, who is not one of the study authors. “It’s a carefully and beautifully designed study, the results of which have created a foundation on which future anticancer strategies can be built.”