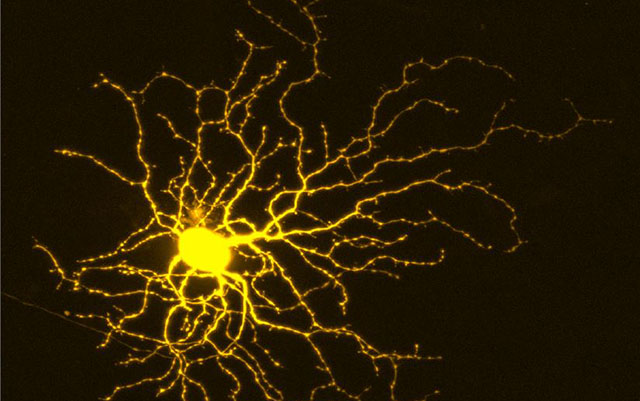
Axon injury often leads to neuron death unless neurons are encouraged to shun the dual leucine zipper kinase (DLK) signaling pathway. As researchers have learned, when this pathway is blocked, neurons more readily survive in models of axon-injuring neurodegenerative disease. But alas, the neurons don’t thrive. That is, they don’t regenerate axons. The problem is, shutting down the DLK pathway does more than prevent cell death. It also inhibits axon regeneration.
According to scientists based at the University of California (UC), San Diego, neurons may benefit from the closure of an alternative route, specifically, the signaling pathway regulated by the germinal cell kinase four (GCK-IV) family of kinases. Blocking GCK-IV kinases, the scientists report, may help neurons survive and thrive even if they are struggling against axon-injuring neurodegenerative diseases. These diseases range from glaucoma to Alzheimer’s disease.
The scientists, led by Derek S. Welsbie, MD, PhD, associate professor of ophthalmology in the Viterbi Family Department of Ophthalmology at Shiley Eye Institute at UC San Diego Health, presented their work December 14 in the Proceedings of the National Academy of Sciences (PNAS), in an article titled, “Inhibition of GCK-IV kinases dissociates cell death and axon regeneration in CNS neurons.”
“[There] are currently no strategies that provide long-term neuroprotection and axonal regeneration after injury,” the article’s authors noted. “[We] identified the GCK-IV family of kinases as targets to maximize neuroprotection while promoting axon regeneration, making it an attractive therapeutic approach for a subset of neurodegenerative diseases.”
The scientists conducted a set of complementary high-throughput screens using a protein kinase inhibitor library in human stem cell–derived retinal ganglion cells (hRGCs). RGCs are neurons that are located near the inner surface of the retina of the eye. They receive visual information from photoreceptors and collectively help transmit that information to the brain.
The first screen involved testing a group of well-studied chemicals to assess their ability to increase the survival of RGCs; the second to measure the ability of chemicals to promote regeneration. After conducting these screens, the scientists determined that “overlapping compounds that promoted both neuroprotection and neurite outgrowth were bioinformatically deconvoluted to identify specific kinases that regulated neuronal death and axon regeneration.”
“[Essentially, we] used a machine learning technique to understand why certain compounds were active while others were not and it identified these key genes,” said Welsbie, who added that the discovery that these genes improved RGC survival was not surprising. “However, you would have predicted that they (like DLK) would have blocked regeneration when inhibited, not promote regeneration. That was definitely a surprise.
“[Our finding] highlights one of the advantages of discovery-based science using high-throughput screening: By testing many agents at once, we can find identify overlooked genes that might not have been thought to play a role.”
Welsbie and colleagues focused their work on RGCs because they are interested in optic neuropathies, such as glaucoma. “Most people think only about glaucoma in terms of ‘eye pressure,’” Welsbie said. But eye pressure is only part of the problem. At its core, glaucoma is a neurodegenerative disease characterized by progressive loss of RGCs and their axons, leading to measurable structural and functional damage to the optic nerve, visual impairment, and blindness.
“Using an adeno-associated virus (AAV) approach, coupled with [CRISPR-based] genome editing, we validated that GCK-IV kinase knockout improves neuronal survival, comparable to that of DLK knockout, while simultaneously promoting axon regeneration,” the authors of the PNAS article detailed. “Finally, we also found that GCK-IV kinase inhibition also prevented the attrition of RGCs in developing retinal organoid cultures without compromising axon outgrowth, addressing a major issue in the field of stem cell-derived retinas.”
Welsbie cautioned that it’s not yet known whether these findings extended beyond RGCs to other neuron types, but he noted that the work suggests strong therapeutic possibilities.
“We basically figured out that there are a set of genes that, when inhibited, allow optic nerve cells to survive and regenerate,” he explained. “Prior to this work, the field knew how to get these cells to survive, but not regenerate. Conversely, there are ways to promote regeneration, but then the survival was rather modest. Of course, for a successful strategy of vision restoration, you need both, and this is a step in that direction.”
“[Our] results,” the authors of the PNAS article concluded, “demonstrate a role for the GCK-IV kinases in dissociating the cell death and axonal outgrowth in neurons, and their druggability provides for therapeutic options for neurodegenerative diseases.”
The modular nature of the AAV/CRISPR system should allow for different permutations of [proregenerative perturbations] to be evaluated in the context of the prosurvival benefit of GCK-IV kinase inhibition. Furthermore, the GCK-IV kinases are druggable targets, which allow for new combinatorial therapeutic possibilities with other proregenerative strategies to maximize survival and the potential for regrowth of axons leading to restored function.”