Scientists in the U.S. and Switzerland have identified three sets of factors that can prompt nerve axons to grow across the damage site of spinal cord injuries (SCI) and re-establish electrical connections with nerves on the other side. The researchers hope that if the results in rodents can be replicated in humans it may lead to new treatments that could regenerate nervous system communication in patients with complete SCI.
“The idea was to deliver a sequence of three very different treatments and test whether the combination could stimulate disconnected axons to regrow across the scar in the injured spinal cord,” explains research lead Michael Sofroniew, Ph.D., a professor of neurobiology at the David Geffen School of Medicine at the University of California, Los Angeles. “Previous studies had tested each of the three treatments separately, but never together. The combination proved to be the key.”
The team, including researchers at Harvard Medical School, and at the Swiss Federal Institute of Technology, reports on its research in Nature, in a paper titled, “Required growth facilitators propel axon regeneration across complete spinal cord injury.”
Spinal cord injuries prevent the brain from sending signals across the site of damage to the neurons below, and this leads to paralysis and the loss of other neurological functions. Prior research has shown that administering molecules such as growth factors to the area around the site of damage can trigger partial axon growth, but not to any significant degree. “Diverse molecules can partially facilitate or attenuate axon growth during development or after injury, but efficient reversal of this regrowth failure remains elusive,” the authors write.
Nerve fibers need three conditions for growth, Dr. Sofroniew suggests. These include the right genetic cues, a substrate rich in the molecules that supports that growth, and a chemoattractant trail that the axons can follow to ensure they are growing in the right direction. These factors are all present in the developing fetus in utero, but after birth these mechanisms become dormant, and genetic cues are switched off.
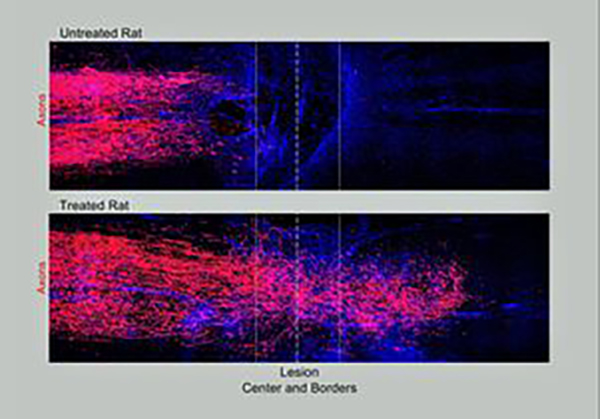
Studies by Dr. Sofroniew’s team aimed to reinstate these three sets of genetic and molecular triggers to promote mature propriospinal neuron axonal growth across complete SCI lesions in rodent models. The injury was designed to affect the animals' hind legs, but not to completely prevent mobility or other functions, such as feeding. The team first used adeno-associated viral (AAV) vectors to deliver genes that would reactivate the intrinsic ability of the propriospinal nerve cells to grow. This technology had been developed and tested previously in the lab of co-author Zhigang He, Ph.D., a neuroscientist at Harvard. Then, at specific time points after spinal cord injury was induced, the team delivered specific growth factors and other molecules to support and promote directional axonal regrowth.
“We reactivated the growth capacity of mature descending propriospinal neurons with osteopontin, insulin-like growth factor 1 and ciliary-derived neurotrophic factor before SCI; induced growth-supportive substrates with fibroblast growth factor 2 and epidermal growth factor; and chemoattracted propriospinal axons with glial-derived neurotrophic factor delivered via spatially and temporally controlled release from biomaterial depots, placed sequentially after SCI,” the authors report.
They witnessed how nerve axons grew across the site of SCI in mice that received all three parts of the treatment at the right time points, whereas there was minimal, if any axonal growth in animals in which one or more factors were missed out. In the triple-treated animals the new axonal growth continued into the neural tissue across from the damage site, forming contacts that expressed synaptic markers. There was also evidence of electrophysiological conduction across the lesion site. “Stimulated, supported, and chemoattracted propriospinal axons regrew a full spinal segment beyond lesion centers, passed well into spared neural tissue, formed terminal-like contacts exhibiting synaptic markers and conveyed a significant return of electrophysiological conduction capacity across lesions,” the researchers state.
“Not only had axons grown robustly through the scar tissue,” Dr. Sofroniew notes, “but many fibers had penetrated into the remaining spinal cord tissue on the other side of the lesion and made new connections with neurons there … When we stimulated the animal's spinal cord with a low electrical current above the injury site, the regrown axons conducted 20% of normal electrical activity below the lesion.”
The treated animals didn’t show improved mobility, but this wasn’t a surprise, Dr. Sofroniew suggests. “We expect that these regrown axons will behave like axons newly grown during development—they do not immediately support coordinated functions. “Much like a newborn must learn to walk, axons that regrow after injury will require training and practice before they can recover function.”
Encouragingly, in a separate set of tests, the same triple-barrelled treatment approach produced equivalent results in rats with severe crush SCI. Again, the animals didn’t recover mobility, but electrophysiological measurements across the lesions in treated animals demonstrated conduction exhibited conduction at about 25% of control levels at 2 mm past lesions, “indicating that propriospinal axon regrowth was associated with a significant return of conduction capacity that correlated with the distance of regrowth past lesions,” the authors write.
They suggest that their findings provide mechanistic insights into why spontaneous axon regrowth fails in adults, and confirm the requirement for all three mechanisms to promote and support axonal growth following SCI. “Our findings, in both mice and rats, strongly support the hypothesis that adult axon regrowth across such lesions fails primarily because of the simultaneous absence or inadequate presence of three types of mechanisms essential for facilitating developmental axon growth: (i) neuron intrinsic growth capacity, (ii) supportive substrate, and (iii) chemoattraction,” they conclude. “Thus, overcoming the failure of axon regrowth across anatomically complete SCI lesions after maturity required the combined sequential reinstatement of several developmentally essential mechanisms that facilitate axon growth … These findings identify a mechanism-based biological repair strategy for complete SCI lesions that could be suitable to use with rehabilitation models designed to augment the functional recovery of remodeling circuits.”