Scientists in the U.S. have grown mouse skin tissue complete with hair follicles (HFs) directly from mouse pluripotent stem cells (mPSCs). The hairy skin more closely resembles natural mouse skin than existing lab-grown tissue that is constructed by piecing together different cell types. The Indiana University School of Medicine researchers suggest that if hair-growing human skin can be generated using a similar approach, it could provide an important model for studying disease or for evaluating new drugs. “It could be potentially a superior model for testing drugs, or looking at things like the development of skin cancers, within an environment that’s more representative of the in vivo microenvironment,“ comments Karl Koehler, Ph.D., an assistant professor of otolaryngology–head and neck surgery. “And it would allow us to limit the number of animals we use for research.”
Lead author Jiyoon Lee, Ph.D., together with Dr. Koehler and colleagues report on their development in Cell Reports, in a paper entitled “Hair Follicle Development in Mouse Pluripotent Stem Cell-Derived Skin Organoids.”
Skin is composed of two layers, the epidermis and the dermis, and it is the interplay between these two layers during embryonic development that gives rise to HFs, sweat glands, and nails. However, as the authors write, “It is currently unclear how to recapitulate hair follicle induction in pluripotent stem cell cultures for use in basic research studies or in vitro drug testing.”
Current methods for generating skin in vitro involve creating the different types of skin cells from pluripotent skin cells in separate cultures, and then combing the cell types to form a skin-like bilayer, the researchers explain. But the resulting skin models only comprise five or six of the 20 or so skin cell types found in real skin. And none of the existing cultured skin types includes HFs. To generate HFs, embryonic stem cells are needed. “Generation of hair follicles in vitro has only been possible using primary cells isolated from embryonic skin, cultured alone or in a co-culture with stem cell-derived cells, combined with in vivo transplantation,” the authors state.
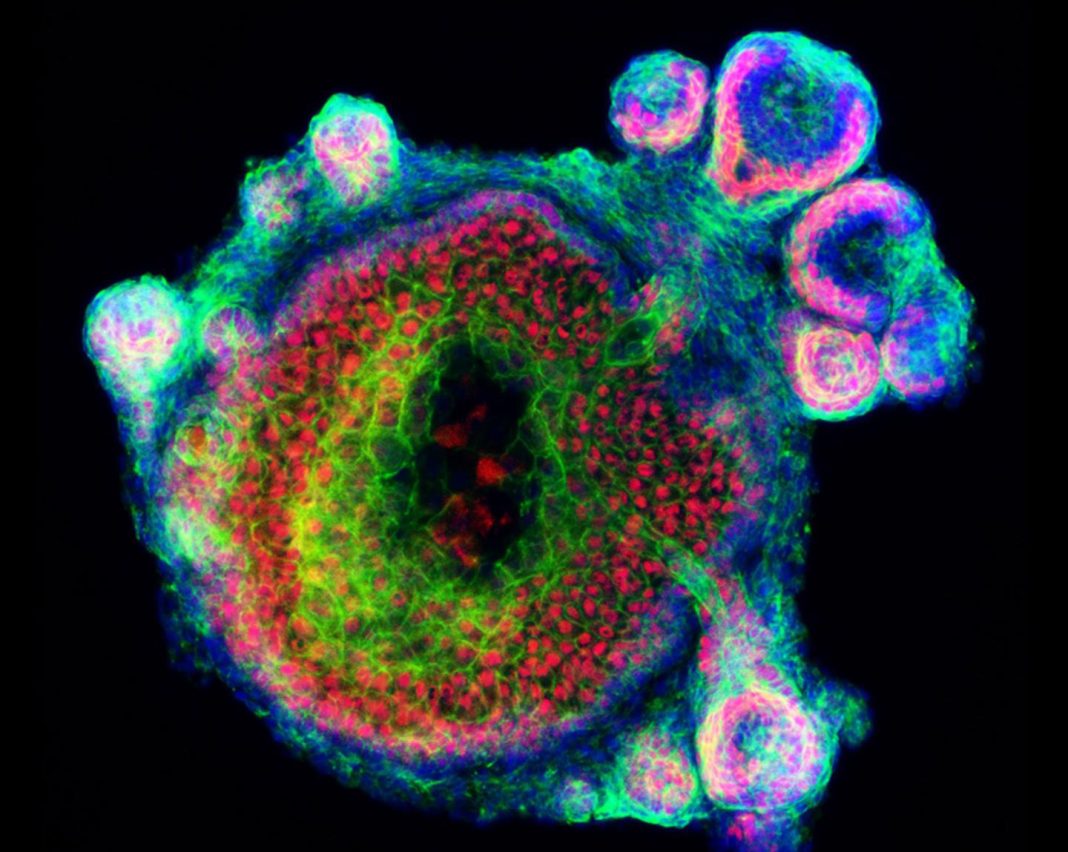
Dr. Koehler’s team had previously developed a mouse stem cell 3D culture system to create organoids that model the inner ear and found that the process also generated skin cells. In their latest studies, the team has shown how mPSCs grown in 3D culture generate skin organoids that comprise both the epidermal and dermal skin layers, and spontaneously produce HFs, in a way that mimics how HFs would normally develop in the mouse embryo skin.
Grown without using embryonic stem cells, the mPSC-derived skin organoid comprised three or four different types of dermal cells and four types of epidermal cells, a combination that more closely mirrored the composition of real mouse skin than previously developed skin tissues, and which developed along similar timelines. “…the mouse skin organoids produced in our culture system recapitulate key features of skin differentiation on a timescale that roughly correlates with normal embryonic development,” the researcher state.
Encouragingly, the HFs growing in the cultured skin contained the key components of naturally deveveloping HFs. “…skin organoids produced in our culture system can generate HFs similar to those seen in late embryonic and early postnatal development, consisting of all major units, including HF matrix, inner root, outer root, and dermal sheath layers.”
The team noticed that the two skin layers needed to grow and interact in a specific way to allow the HFs to develop. The epidermis growing in the culture medium would start to take on the round shape of a cyst, and the dermal cells would then wrap around the cysts. If this process was disrupted, the HFs failed to develop.
“You can see the organoids with your naked eye,” Koehler says. “It looks like a little ball of pocket lint that floats around in the culture medium. The skin develops as a spherical cyst, and then the HFs grow outward in all directions, like dandelion seeds.…One thing we explored in the paper is that if we destroy the organoids and try to put them back together, they don't always generate HFs,” Koehler says. “So, we think that it's very important that the cells develop together at an early stage to properly form skin and HFs.”
The team says its results demonstrate an approach for generating skin from pluripotent stem cells, which induces both the epidermal and dermal cells in a single organoid unit. “Our study demonstrates how these skin organoid structures can be generated de novo, without the use of embryonic tissue…,” the authors write. “We anticipate that this culture system will be useful for studying minimal cellular and microenvironment requirements for HF induction, evaluating HF growth/inhibitory drugs, or modeling skin diseases.”
The researchers are now working to understand how to create a culture system that supports the natural growth cycles of HFs over time, and which allows the skin to shed and regenerate hairs. “Thus, further studies will be necessary to explore ways of increasing the longevity of skin organoid cultures to potentially support in vitro HF cycling,” they conclude.