Alternating conformations and switching tasks, a dimer’s oppositely oriented halves relieve the cellular stress that can build up when molecular oxygen is used for power generation. When one half of the dimer acts like a proton pump, the other half converts NADH to NADP+—and vice versa.
Both of these mutually exclusive tasks are necessary to create NADPH, which is used for amino acid biosynthesis and to remove reactive oxygen species. Yet somehow both tasks are accomplished by one enzyme, nicotinamide nucleotide transhydrogenase (TH).
The details of TH’s task juggling ways emerged from structural studies of the ancient enzyme, which is found throughout the animal kingdom as well as in plants and many simpler species. In humans and other higher organisms, TH works within mitochondria, the tiny double-hulled oxygen reactors that help power most cellular processes.
Although oxygen is great for efficient energy production, it has a serious downside: the accumulation of reactive oxygen species. These have been linked with diseases such as diabetes, cancer, and Alzheimer's and Parkinson's diseases, as well as with cell death and aging. To support their use of oxygen, cells have had to evolve defense mechanisms, of which TH is a part.
TH has long been thought to participate in the production of NADPH, which is crucial for defusing oxygen-free radicals. But TH has been hard to study. It has exceptionally loose structural components, and so it is difficult to evaluate using X-ray crystallography. With its structure poorly understood, TH has given up few functional secrets.
Until recently. In the January 9 issue of Science, researchers based at The Scripps Research Institute (TSRI) presented a new structural model for TH, which they used to support speculation about TH’s mechanisms. The details, which appeared in an article entitled, “Division of labor in transhydrogenase by alternating proton translocation and hydride transfer,” included the structure of TH’s transmembrane portion and the way in which TH’s parts relate to the enzyme’s whole.
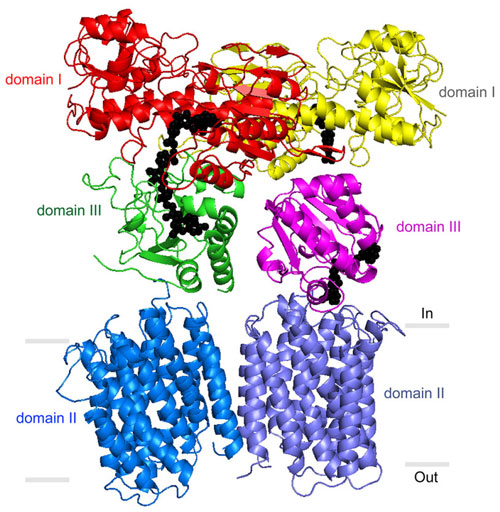
“We present the 2.8 Å crystal structure of the transmembrane proton channel domain of TH from Thermus thermophilus and the 6.9 Å crystal structure of the entire enzyme (holo-TH),” wrote the authors. “The membrane domain crystallized as a symmetric dimer, with each protomer containing a putative proton channel.”
Most interesting were revelations about TH’s “domain III” structures. These appear directly above TH’s transmembrane structure, just inside the mitochondrial matrix. They serve to bind NDPH’s precursor molecule, NADP+, during conversion to NADPH. Structural biologists haven't understood how two such structures could work side by side in the TH dimer and not interfere with each other's activity. The new structural data suggest that these side-by-side structures are highly flexible and always have different orientations.
“Our most striking finding was that the two domain III structures are not symmetric—one of them faces up while the other faces down,” said Josephine H. Leung, first author of the Science article and a graduate student in the laboratory of C. David Stout, Ph.D., an associate professor at TSRI.
One of structures is oriented apparently to catalyze the production of NADPH, while the other is turned toward the membrane, perhaps to facilitate transit of a proton. The new structural model suggests that with each proton transit, the two domain III structures flip and switch their functions. “We suspect that the passage of the proton is what somehow causes this flipping of the domain III structures,” Leung noted.
“Despite its importance, TH has been one of the least studied of mitochondrial enzymes,” stated Dr. Stout. “Our new study helps clear up some mysteries—suggesting how the enzyme structure might harness protons and indicating that its two sides are able to alternate functions, always staying in balance.”