February 15, 2006 (Vol. 26, No. 4)
A New Screening Paradigm in 1,536-Well Microtitre Plates Using Aequorin and LumiLux
Gprotein-coupled receptors (GPCRs) are an important class of receptor in modern drug discovery. This superfamily of receptors is highly tractable for drug discovery purposes because many of these receptors are coupled (via Gaq/11) to transient release of intracellular calcium upon receptor activation. Co-expression of non Gaq/11coupled GPCRs with various promiscuous and/or chimeric G-proteins directs receptor activation toward the same endpoint.
Generic screening strategies based on measurement of intracellular calcium have been employed throughout the industry, forming the basis of most high-throughput screening at this receptor class in recent years. The standard paradigm for these generic intracellular calcium assays has been based around the use of Ca2+-sensitive fluorescent dyes such as Fluo-3 and Fluo-4. These assays involve multiple steps (e.g., cell plating, dye-loading, plate washing), are not easily miniaturized beyond 384-well format, and are prone to compound interference from autofluorescence.
This tutorial describes 384- and 1,536-well format intracellular assays using the PerkinElmer (www.perkinelmer.com) LumiLux cellular screening platform in combination with recombinantly expressed aequorin, a Ca2+-sensitive photoprotein, to address this unmet need.
The LumiLux can measure flash and glow luminescence in a variety of plate formats, including 384-well and 1,536-well plates. An ultracooled, fiber-optic coupled back-thinned CCD camera allows for imaging of all wells across the entire plate. The camera optics are based on fiber optic contact-imaging technology, which results in efficient light gathering and avoids the need for complicated lenses in the optical path. The LumiLux accommodates a 384-well Multi-Position-Dispense (MPD) head with a pipetting volume range of 0.530 L using disposable tips.
With three built-in stackers, LumiLux is a standalone system that performs multiple operations. Concurrent with plate reading, the conveyor and the MPD head can operate simultaneously for parallel processing. In addition, the robotic shuttle provides access into the light-tight enclosure and integration with external devices if required. An on-board cell stirrer maintains cells in a homogeneous suspension that allows aequorin flash luminescence assays to be run continuously with full automated control. The combination of the aequorin suspension assay paradigm and the parallel processing on LumiLux allows for standalone 1,536 screening.
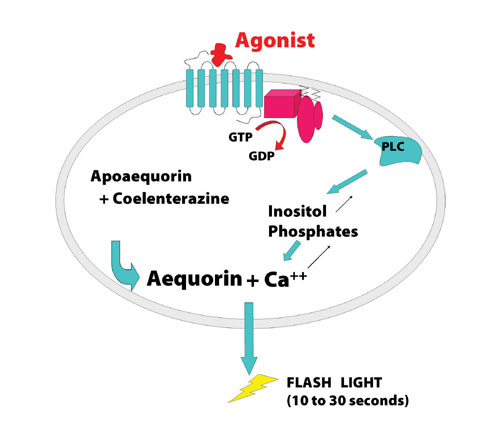
Figure 1: Principles of the aequorin-based method.
Aequorin Protocol
An aequorin protocol provides an alternative for cell-based screening using calcium mobilization (Figure 1). Aequorin assays are easier to automate as they do not require cell wash steps, and optimal assay performance can be achieved at room temperature. Using cells in suspension renders aequorin assays more amenable to automated operation in 1,536-well format with increased throughput and productivity. The use of cell suspensions provides reduction in cell plating, and maintenance costs and sensitivity of the assay afforded by high signal-to-background means fewer cells per well can be used than in fluorescent-based assays.
Assay costs are further reduced using the low-cost reagent coelenterazine (<$.02/well) which has little cellular toxicity and is not subject to efflux. Reduced compound interference arising from auto-fluorescence and toxicity should produce fewer false positives and a cleaner data set.
Chinese Hamster Ovary cells expressing recombinant histamine H1 receptors and mitochondrial-targeted aequorin (CHO-Aeq-H1: EuroScreen) were grown in the following media: DMEM:F12 (1:1), 1% Penicillin-Streptomycin, 2 mM L-glutamine, 10% fetal calf serum (FCS), and 500 g/mL geneticin. Cells were routinely cultured at 37C in an atmosphere of 95% air/5% CO2.
On the day of assay the cells were harvested using versene and resuspended at a density of 5×106 cells/mL in DMEM (without phenol red) supplemented with 0.1% BSA and 15mM HEPES medium. Active aequorin was reconstituted by the addition of 5M coelenterazine h (Invitrogen C 6780) and the cell suspension stirred at 200 rpm on a magnetic stirrer for four hours in the dark at room temperature.
Prior to measurement (384-well format) on LumiLux, the cell suspension was diluted 10-fold and transferred to the integrated cell stirrer assembly. For agonist assays, cell suspension (25 l) was dispensed onto compound (25 l) in a 384-well black, clear-bottom plate (Greiner) with simultaneous imaging of the entire plate for 40 seconds with 10-second baseline prior to cell addition. For antagonist assays, compound and cells were pre-mixed (25 l of each), incubated for 10 minutes, with a final addition (25 l) of agonist (EC80).
The 1,536-well format agonist assays were conducted with 3 l of the cell suspension dispensed onto compound (3 l) in a 1,536-well black, clear bottom LoBase plate (Greiner). For the antagonist assay compound and cells were pre-mixed (3 l of each), incubated for 10 minutes, with a final addition (3 l) of agonist (EC80).
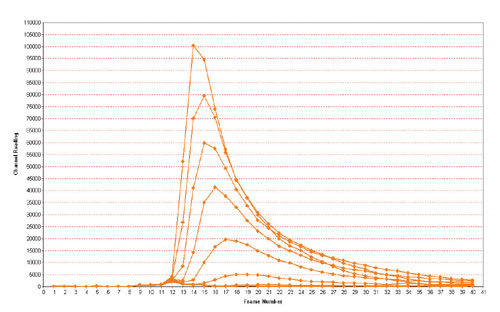
Fig. 2 Kinetic profile of flash luminescence signal from CHO-Aeq-H1 cells stimulated with varying concentrations of histamine from 0.1 nM to 2uM
Data Analysis
Data were analyzed using the PerkinElmer AssayPro (Figures 2 and 3) or GraphPad Prism version 3.02 (Figure 4). Agonist concentration response curves (Figure 3a,c) and antagonist inhibition curves (Figure 3b,d) were fit to the 4-parameter logistic equation [Y = a + (b – a) / (1 + 10^ ((LogEC50 – X) * d ))] to allow calculation of pEC50 and pIC50 values respectively. For comparison of aequorin suspension cell assay data with fluorescence (Fluo-4) assay data using adherent cells, data were expressed as % of positive control.
The kinetic response profile of CHO-Aeq-H1 cells stimulated with various concentrations of histamine is shown in Figure 2. Activation of the H1 receptor induces a transient flash luminescence signal, which is maximal after 58 seconds, returning to baseline levels after 30 seconds.

Fig. 3A. Concentration response curves for histamine and ATP (384-well, 12,500-cells/well)
Results
Agonist concentration response curves in 384- and 1,536-well format were conducted using histamine and ATP, an endogenous purinergic receptor agonist. The results show (Figure 3a,c) comparable assay pharmacology and assay performance with minimal loss of data quality associated with miniaturization, giving control Z values (2 M histamine) of 0.93 and 0.78 in 384- and 1,536-well formats respectively. Similarly the assay pharmacology and performance (Z range 0.720.83) in both 384- and 1,536-well formats are almost identical with pIC50 values of 7.98 (384-well) and 7.92 (1,536-well) for the antagonist trans-triprolidine (Figure 3b,d).
LumiLux aequorin assays (histamine concentration-response) in 384-well format show comparable pharmacology and improved assay performance (pEC50 = 7.55, Z = 0.91) over the standard fluorescence adherent assay (pEC50 = 7.38, Z= 0.76) using the CHO-Aeq-H1 cell line (Figure 4).

Fig. 3b. Inhibition curve for the antagonist trans-triprolidine (384-well, 12,500-cell/wells)
Conclusion
In conclusion, our data shows that the LumiLux Cellular Screening Platform (Figure 5) with unique cell stirrer can deliver excellent assay performance for simplified suspension cell-based Ca2+ assays, compared to the standard fluorescent dye methodology. The successful miniaturization of this assay to 1,536well format as demonstrated will allow, for the first time, screening groups to run even higher throughput intracellular Ca2+ assays (up to 200 1,536-well plates per day) with increased productivity, reduced screening and labor costs, and the high degree of assay performance demanded by drug discovery.
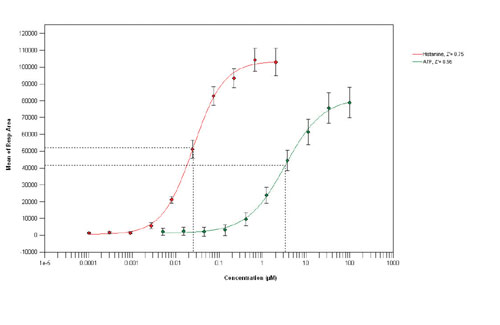
Fig. 3C: Concentration response curves for histamine and ATP (1536-well, 1,500cells/well).
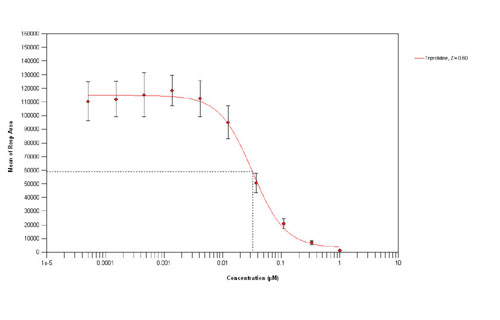
Fig. 3D: Inhibition curve for antagonist trans-triprolidine (1,536-well, 1,500 cells/well.
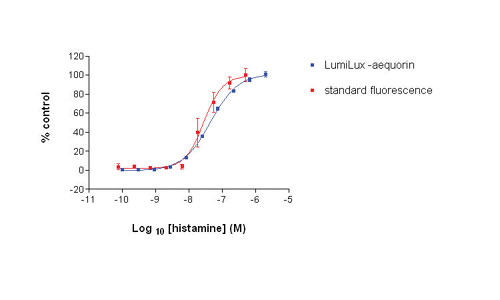
Fig 4: Comparison of 384-well histamine concentration response (%control) for aequorin suspension assay and standard fluorescence adherent cell assay paradigm using CHO-Aeq-H1 cells.

LumiLux Cellular Screening Platform







