Doug Auld, Ph.D. Novartis Institutes for BioMedical Research
This literature review shows how researchers used CyTOF mass cytometry to obtain spatial resolution of cell samples.
The integration of mass spectrometry (specifically laser ablation of the sample in combination with inductively coupled plasma mass spectrometry) with flow cytometry instrumentation along with sensitive and rare earth metal labels has enabled multiplexing of up to 32 cell markers (see Assay Drug Dev Technol 2011;9:567 commentary “Flow cytometry goes atomic;” CyTOF system sold by Fluidigm, formerly DVS Sciences). This process employs typical immunocytochemistry techniques, but the antibodies are tagged with rare earth metal isotopes (predominately lanthanides) that act as specific reporters of cellular proteins. Comparison of these rare earth–labeled antibodies to typical fluorescent antibody labels supports that these labels do not affect the specificity or sensitivity of the antibodies. In this article the authors* extend this method to obtain spatial resolution of cell samples.
In the method, the signals from the rare earth reporters following laser ablation of the sample is correlated with the position of the laser spot as it is scanned across the sample with 1 μm resolution. The limit of detection is determined to be ∼500 molecules. The data can then be plotted based on the position of each ion spot for each rare earth reporter, and these images are then overlaid to create a high-dimensional image that can be analyzed (Figure). Measurement of a 0.5 mm × 0.5 mm area at 1 μm resolution takes ∼3.5 h. The system is capable of measuring 100 analytes simultaneously, but only 32 rare earth metal chelates are currently available.
The authors applied this method to measure heterogeneity in breast cancer tumors using formalin-fixed, paraffin-embedded (FFPE) breast cancer samples. A total of 21 FFPE samples were analyzed using 32-plex imaging mass cytometry covering cell markers and phosphoproteins. Differences in expression even within the same tumor sample were noted, and the subpopulations branch points often contained markers used for patient classification. Some exceptions occurred; for example, Her2 was detected and confirmed in one triple-negative case. This high-dimensional imaging should increase our understanding of tumor biology and pathologies.
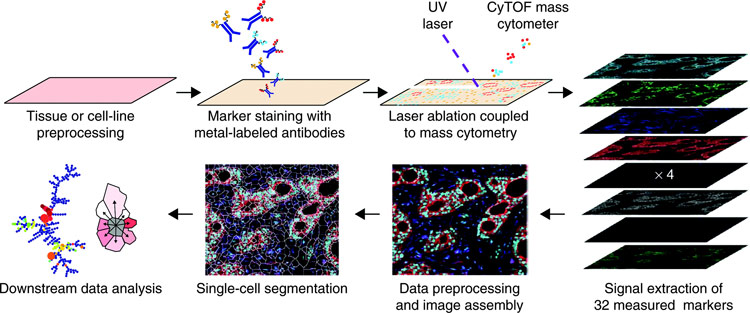
Workflow of imaging mass cytometry.
*Abstract from Nature Methods 2014, Vol. 11: 403–406
Mass cytometry enables high-dimensional, single-cell analysis of cell type and state. In mass cytometry, rare earth metals are used as reporters on antibodies. Analysis of metal abundances using the mass cytometer allows determination of marker expression in individual cells. Mass cytometry has previously been applied only to cell suspensions.
To gain spatial information, we have coupled immunohistochemical and immunocytochemical methods with high-resolution laser ablation to CyTOF mass cytometry. This approach enables the simultaneous imaging of 32 proteins and protein modifications at subcellular resolution; with the availability of additional isotopes, measurement of over 100 markers will be possible. We applied imaging mass cytometry to human breast cancer samples, allowing delineation of cell subpopulations and cell–cell interactions and highlighting tumor heterogeneity.
Imaging mass cytometry complements existing imaging approaches. It will enable basic studies of tissue heterogeneity and function and support the transition of medicine toward individualized molecularly targeted diagnosis and therapies.
Doug Auld, Ph.D., is affiliated with the Novartis Institutes for BioMedical Research.
ASSAY & Drug Development Technologies, published by Mary Ann Liebert, Inc., offers a unique combination of original research and reports on the techniques and tools being used in cutting-edge drug development. The journal includes a "Literature Search and Review" column that identifies published papers of note and discusses their importance. GEN presents here one article that was analyzed in the "Literature Search and Review" column, a paper published in Nature Methods titled "Highly multiplexed imaging of tumor tissues with subcellular resolution by mass cytometry." Authors of the paper are Giesen C, Wang HAO, Schapiro D, Zivanovic N, Jacobs A, Hattendorf B, Schüffler PJ, Grolimund D, Buhmann JM, Brandt S, Zsuzsanna V, Wild PJ, Günther D, and Bodenmiller B.







